Praluent: Package Insert / Prescribing Info
Package insert / product label
Generic name: alirocumab
Dosage form: injection, solution
Drug class: PCSK9 inhibitors
Medically reviewed by Drugs.com. Last updated on Sep 17, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
PRALUENT® (alirocumab) injection, for subcutaneous use
Initial U.S. Approval: 2015
Indications and Usage for Praluent
PRALUENT is a proprotein convertase subtilisin kexin type 9 (PCSK9) inhibitor indicated:
- To reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with established cardiovascular disease. (1)
- As adjunct to diet, alone or in combination with other low-density lipoprotein cholesterol (LDL-C)-lowering therapies, in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH), to reduce LDL-C. (1)
- As an adjunct to other LDL-C-lowering therapies in adult patients with homozygous familial hypercholesterolemia (HoFH) to reduce LDL-C. (1)
- As an adjunct to diet and other LDL-C-lowering therapies in pediatric patients aged 8 years and older with HeFH to reduce LDL-C. (1)
Praluent Dosage and Administration
- In adults with established cardiovascular disease or with primary hyperlipidemia, including HeFH (2.1):
- The recommended starting dosage of PRALUENT is either 75 mg once every 2 weeks or 300 mg once every 4 weeks administered subcutaneously.
- For patients receiving PRALUENT 300 mg every 4 weeks, measure LDL-C just prior to the next scheduled dosage, because LDL-C can vary between dosages in some patients.
- If the LDL-C response is inadequate, the dosage may be adjusted 150 mg subcutaneously every 2 weeks.
- In adults with HeFH undergoing LDL apheresis or in adults with HoFH (2.1):
- The recommended dosage of PRALUENT is 150 mg once every 2 weeks administered subcutaneously.
- PRALUENT can be administered without regard to the timing of LDL apheresis.
- In pediatric patients with HeFH (2.2):
- The recommended dosage of PRALUENT for patients with a body weight less than 50 kg is 150 mg once every 4 weeks administered subcutaneously.
- The recommended dosage of PRALUENT for patients with a body weight of 50 kg or more is 300 mg once every 4 weeks administered subcutaneously.
- If the LDL-C response is inadequate, the dosage may be adjusted for patients with a body weight less than 50 kg to 75 mg subcutaneously once every 2 weeks or for patients with a body weight of 50 kg or more to 150 mg subcutaneously once every 2 weeks.
- Assess LDL-C when clinically appropriate. The LDL-lowering effect of PRALUENT may be measured as early as 4 weeks after initiation. (2.1)
- Administer PRALUENT subcutaneously into areas of the thigh, abdomen, or upper arm that are not tender, bruised, red, or indurated. Rotate injection sites for each administration. (2.4)
- To administer the 300 mg dosage, give two 150 mg PRALUENT injections consecutively at two different injection sites. (2.4)
Dosage Forms and Strengths
Injection: 75 mg/mL or 150 mg/mL in a single-dose pre-filled pen. (3)
Contraindications
History of a serious hypersensitivity reaction to alirocumab or any of the excipients in PRALUENT. (4)
Warnings and Precautions
Hypersensitivity reactions: hypersensitivity vasculitis, angioedema, and other hypersensitivity reactions requiring hospitalization, have been reported with PRALUENT treatment. If signs or symptoms of serious hypersensitivity reactions occur, discontinue treatment with PRALUENT, treat according to the standard of care, and monitor until signs and symptoms resolve. (5.1)
Adverse Reactions/Side Effects
Common (>5% of patients treated with PRALUENT and more frequently than placebo) adverse reactions in adults with: Primary hyperlipidemia: injection site reactions, and influenza. (6) Established cardiovascular disease: myalgia. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Regeneron at 1-844-734-6643 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2024
Full Prescribing Information
1. Indications and Usage for Praluent
PRALUENT® is indicated:
- To reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with established cardiovascular disease.
- As an adjunct to diet, alone or in combination with other low density lipoprotein cholesterol (LDL-C)-lowering therapies, in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH), to reduce LDL-C.
- As an adjunct to other LDL-C-lowering therapies in adult patients with homozygous familial hypercholesterolemia (HoFH) to reduce LDL-C.
- As an adjunct to diet and other LDL-C-lowering therapies in pediatric patients aged 8 years and older with HeFH to reduce LDL-C.
2. Praluent Dosage and Administration
2.1 Recommended Dosage in Adults
- Established cardiovascular disease or with primary hyperlipidemia, including HeFH:
- The recommended starting dosage of PRALUENT is either 75 mg once every 2 weeks or 300 mg once every 4 weeks administered subcutaneously [see Dosage and Administration (2.4)].
- For patients receiving PRALUENT 300 mg every 4 weeks, measure LDL-C just prior to the next scheduled dose, because LDL-C can vary between doses in some patients [see Clinical Studies (14)].
- If the LDL-C response is inadequate, the dosage may be adjusted to 150 mg subcutaneously every 2 weeks.
- HeFH undergoing LDL apheresis or with HoFH:
- The recommended dosage of PRALUENT is 150 mg once every 2 weeks administered subcutaneously [see Dosage and Administration (2.4)].
- PRALUENT can be administered without regard to the timing of LDL apheresis.
- Assess LDL-C when clinically appropriate. The LDL-lowering effect of PRALUENT may be measured as early as 4 weeks after initiation.
2.2 Recommended Dosage in Pediatric Patients aged 8 years and older with HeFH
-
The recommended dosage of PRALUENT for patients with a body weight less than 50 kg is 150 mg once every 4 weeks administered subcutaneously [see Dosage and Administration (2.4)].
- If the LDL-C lowering response is inadequate, the dosage may be adjusted to 75 mg subcutaneously once every 2 weeks [see Dosage and Administration (2.4)].
-
The recommended dosage of PRALUENT for patients with a body weight of 50 kg or more is 300 mg once every 4 weeks administered subcutaneously [see Dosage and Administration (2.4)].
- If the LDL-C lowering response is inadequate, the dosage may be adjusted to 150 mg subcutaneously once every 2 weeks [see Dosage and Administration (2.4)].
- Assess LDL-C when clinically appropriate. The LDL-lowering effect of PRALUENT may be measured as early as 4 weeks after initiation.
2.3 Missed Doses
If a dose is missed:
- Within 7 days from the missed dose, instruct the patient to administer PRALUENT and resume the patient's original schedule.
- More than 7 days after the missed dose:
- For every 2-week dosage, instruct the patient to wait until the next dose on the original schedule.
- For every 4-week dosage, instruct the patient to administer the dose and start a new schedule based on this date.
2.4 Important Administration Instructions
- Train patients and/or caregivers on how to prepare and administer PRALUENT, according to the Instructions for Use and instruct them to read and follow the Instructions for Use each time they use PRALUENT.
- In children aged 12 to 17 years, it is recommended that PRALUENT be given by or under the supervision of an adult. In children aged 8 to 11 years, PRALUENT should be given by a caregiver.
- Prior to use, allow PRALUENT to warm to room temperature for 30 to 40 minutes if PRALUENT has been refrigerated [see How Supplied/Storage and Handling (16)].
- Visually inspect PRALUENT prior to administration. PRALUENT is a clear, colorless to pale yellow solution. Do not use if the solution is cloudy, discolored, or contains particles.
- Administer PRALUENT subcutaneously into areas of the thigh, abdomen, or upper arm that are not tender, bruised, red, or indurated. Rotate injection sites for each administration.
- To administer the 300 mg dose, give two 150 mg PRALUENT injections consecutively at two different injection sites.
3. Dosage Forms and Strengths
PRALUENT injection is a clear, colorless to pale yellow solution available as follows:
- 75 mg/mL single-dose pre-filled pen
- 150 mg/mL single-dose pre-filled pen
4. Contraindications
PRALUENT is contraindicated in patients with a history of a serious hypersensitivity reaction to alirocumab or any of the excipients in PRALUENT. Hypersensitivity vasculitis, angioedema, and hypersensitivity reactions requiring hospitalization have occurred [see Warnings and Precautions (5.1)].
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including hypersensitivity vasculitis, angioedema, and other hypersensitivity reactions requiring hospitalization, have been reported with PRALUENT treatment. If signs or symptoms of serious hypersensitivity reactions occur, discontinue treatment with PRALUENT, treat according to the standard of care, and monitor until signs and symptoms resolve. PRALUENT is contraindicated in patients with a history of a serious hypersensitivity reaction to alirocumab or any excipient in PRALUENT [see Contraindications (4)].
6. Adverse Reactions/Side Effects
The following adverse reactions are also discussed in the other sections of the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Reactions in Adults with Primary Hyperlipidemia
The data in Table 1 are derived from 9 primary hyperlipidemia placebo-controlled trials that included 2,476 adult patients treated with PRALUENT 75 mg and/or 150 mg every 2 weeks, including 2,135 exposed for 6 months and 1,999 exposed for more than 1 year (median treatment duration of 65 weeks). The mean age of the population was 59 years, 40% of the population were female, 90% were White, 4% were Black or African American, 3% were Asian, and 3% other races; 6% identified as Hispanic or Latino ethnicity.
Adverse reactions reported in at least 2% of PRALUENT-treated patients, and more frequently than in placebo-treated patients, are shown in Table 1.
| Adverse Reactions | Placebo (N=1,276) % | PRALUENT*
(N=2,476) % |
|---|---|---|
| Injection site reactions† | 5 | 7 |
| Influenza | 5 | 6 |
| Diarrhea | 4 | 5 |
| Myalgia | 3 | 4 |
| Muscle spasms | 2 | 3 |
| Contusion | 1 | 2 |
Adverse reactions led to discontinuation of treatment in 5.3% of patients treated with PRALUENT and 5.1% of patients treated with placebo. The most common adverse reactions leading to treatment discontinuation in patients treated with PRALUENT were allergic reactions (0.6% versus 0.2% for PRALUENT and placebo, respectively) and elevated liver enzymes (0.3% versus <0.1%).
In an analysis of ezetimibe-controlled trials in which 864 patients were exposed to PRALUENT for a median of 27 weeks and 618 patients were exposed to ezetimibe for a median of 24 weeks, the types and frequencies of common adverse reactions were similar to those listed above.
Adverse Reactions in a Cardiovascular Outcomes Trial in Adults
In a cardiovascular outcomes trial in which 9,451 patients were exposed to PRALUENT for a median of 31 months and 9,443 patients were exposed to placebo for a median of 32 months, common adverse reactions (greater than 5% of patients treated with PRALUENT and occurring more frequently than placebo) included myalgia (6% PRALUENT, 5% placebo).
Adverse Reactions in Pediatric Patients with HeFH
In a 24-week placebo-controlled clinical trial in which 101 pediatric patients aged 8 to 17 years with HeFH were exposed to PRALUENT and 52 pediatric patients with HeFH were exposed to placebo [see Clinical Studies (14.3)], the safety profile of PRALUENT observed in this population was consistent with the safety profile observed in adults with HeFH.
Other Adverse Reactions
Local Injection Site Reactions
In a pool of placebo-controlled trials evaluating PRALUENT 75 mg and/or 150 mg administered every 2 weeks in adults, local injection site reactions including erythema/redness, itching, swelling, and pain/tenderness were reported more frequently in patients treated with PRALUENT (7.2% versus 5.1% for PRALUENT and placebo, respectively). Few patients discontinued treatment because of these reactions (0.2% versus 0.4% for PRALUENT and placebo, respectively), but patients receiving PRALUENT had a greater number of injection site reactions, had more reports of associated symptoms, and had reactions of longer average duration than patients receiving placebo.
In a 48-week placebo-controlled trial evaluating PRALUENT 300 mg every 4 weeks and 75 mg every 2 weeks in adults, in which all patients received an injection of drug or placebo every 2 weeks, local injection site reactions were reported more frequently in patients treated with PRALUENT 300 mg every 4 weeks as compared to those receiving PRALUENT 75 mg every 2 weeks or placebo (16.6%, 9.6%, and 7.9%, respectively). Three patients (0.7%) treated with PRALUENT 300 mg every 4 weeks discontinued treatment due to local injection site reactions versus no patients (0%) in the other 2 treatment groups.
In a cardiovascular outcomes trial in adults, local injection site reactions were reported in 3.8% of patients treated with PRALUENT versus 2.1% patients treated with placebo, and led to permanent discontinuation in 26 patients (0.3%) versus 3 patients (<0.1%), respectively.
In the trial of pediatric patients with HeFH, local injection site reactions were reported in 5% of patients treated with PRALUENT versus 0% patients treated with placebo; no patients discontinued treatment due to injection site reactions.
Hypersensitivity Reactions in Adults
Hypersensitivity reactions were reported more frequently in adult patients treated with PRALUENT than in those treated with placebo (8.6% versus 7.8%). The most common hypersensitivity reaction was pruritus (1.1% versus 0.4% for PRALUENT and placebo, respectively). The proportion of patients who discontinued treatment due to allergic reactions was higher among those treated with PRALUENT (0.6% versus 0.2%).
Serious allergic reactions, such as hypersensitivity, nummular eczema, and hypersensitivity vasculitis were reported in patients using PRALUENT in controlled clinical trials.
Liver Enzyme Abnormalities in Adults
In the primary hyperlipidemia trials in adults, liver-related disorders (primarily related to abnormalities in liver enzymes) were reported in 2.5% of patients treated with PRALUENT and 1.8% of patients treated with placebo, leading to treatment discontinuation in 0.4% and 0.2% of patients, respectively. Increases in serum transaminases to greater than 3 times the upper limit of normal occurred in 1.7% of patients treated with PRALUENT and 1.4% of patients treated with placebo.
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval use of PRALUENT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypersensitivity reactions: Angioedema
- Influenza-like illness
Related/similar drugs
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Available data from clinical trials and postmarketing reports on PRALUENT use in pregnant women are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes. In animal reproduction studies, there were no effects on embryo-fetal development when rats were subcutaneously administered alirocumab during organogenesis at dose exposures up to 12-fold the exposure at the maximum recommended human dose of 150 mg every two weeks. In monkeys, suppression of the humoral immune response was observed in infant monkeys when alirocumab was dosed during organogenesis to parturition at dose exposures 13-fold the exposure at the maximum recommended human dose of 150 mg every two weeks. No additional effects on pregnancy or neonatal/infant development were observed at dose exposures up to 81-fold the maximum recommended human dose of 150 mg every two weeks. Measurable alirocumab serum concentrations were observed in the infant monkeys at birth at comparable levels to maternal serum, indicating that alirocumab, like other IgG antibodies, crosses the placental barrier. Monoclonal antibodies are transported across the placenta in increasing amounts especially near term; therefore, alirocumab has the potential to be transmitted from the mother to the developing fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%-4% and 15%-20%, respectively.
There is a pregnancy safety study for PRALUENT. If PRALUENT is administered during pregnancy, healthcare providers should report PRALUENT exposure by contacting Regeneron at 1-844-734-6643.
Data
Animal data
In Sprague Dawley rats, no effects on embryo-fetal development were observed when alirocumab was dosed at up to 75 mg/kg/dose by the subcutaneous route on gestation days 6 and 12 at exposures 12-fold the maximum recommended human dose of 150 mg every two weeks, based on serum AUC.
In cynomolgus monkeys, suppression of the humoral immune response to keyhole limpet hemocyanin (KLH) antigen was observed in infant monkeys at 4 to 6 months of age when alirocumab was dosed during organogenesis to parturition at 15 mg/kg/week and 75 mg/kg/week by the subcutaneous route, corresponding to 13-fold and 81-fold the human exposure at the maximum recommended human dose of 150 mg every two weeks, based on serum AUC. The lowest dose tested in the monkey resulted in humoral immune suppression; therefore, it is unknown if this effect would be observed at clinical exposure. No study designed to challenge the immune system of infant monkeys was conducted. No additional embryo-fetal, prenatal or postnatal effects were observed in infant monkeys, and no maternal effects were observed, when alirocumab was dosed at up to 75 mg/kg/week by the subcutaneous route, corresponding to maternal exposure of 81-fold the exposure at the maximum recommended human dose of 150 mg every two weeks, based on serum AUC.
8.2 Lactation
Risk Summary
There is no information regarding the presence of alirocumab in human milk, the effects on the breastfed infant, or the effects on milk production. The development and health benefits of breastfeeding should be considered along with the mother's clinical need for PRALUENT and any potential adverse effects on the breastfed infant from PRALUENT or from the underlying maternal condition. Human IgG is present in human milk, but published data suggest that breast milk IgG antibodies do not enter the neonatal and infant circulation in substantial amounts.
8.4 Pediatric Use
The safety and effectiveness of PRALUENT as an adjunct to diet and other LDL-C-lowering therapies for the treatment of HeFH have been established in pediatric patients aged 8 years and older. Use of PRALUENT for this indication is based on data from a 24-week, randomized, placebo-controlled, double-blind trial in pediatric patients with HeFH. In the trial, 101 patients received PRALUENT and 52 patients received placebo; 26 patients (17%) were 8 to 9 years of age. This indication is supported by evidence from controlled trials in adults [see Adverse Reactions (6.1) and Clinical Studies (14.3)].
The safety and effectiveness of PRALUENT have not been established in pediatric patients with HeFH who are younger than 8 years of age or in pediatric patients with other types of hyperlipidemia.
8.5 Geriatric Use
In controlled trials, 3663 patients treated with PRALUENT were ≥65 years of age and 734 patients treated with PRALUENT were ≥75 years of age. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
No dose adjustment is needed for patients with mild or moderately impaired renal function. No data are available in patients with severe renal impairment [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dose adjustment is needed for patients with mild or moderate hepatic impairment. No data are available in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)].
11. Praluent Description
Alirocumab is a human monoclonal antibody (IgG1 isotype) that targets proprotein convertase subtilisin kexin type 9 (PCSK9). Alirocumab is a PCSK9 inhibitor produced by recombinant DNA technology in Chinese Hamster Ovary cell suspension culture. Alirocumab consists of two disulfide-linked human heavy chains, each covalently linked through a disulfide bond to a human kappa light chain. A single N-linked glycosylation site is located in each heavy chain within the CH2 domain of the Fc constant region of the molecule. The variable domains of the heavy and light chains combine to form the PCSK9 binding site within the antibody. Alirocumab has an approximate molecular weight of 146 kDa.
PRALUENT is a sterile, preservative-free, clear, colorless to pale yellow solution for subcutaneous use. PRALUENT 75 mg/mL or 150 mg/mL solution for subcutaneous injection in a single-dose pre-filled pen is supplied in a siliconized 1 mL Type-1 clear glass syringe.
Each 75 mg/mL pre-filled pen contains 75 mg alirocumab, histidine (8 mM), polysorbate 20 (0.1 mg), sucrose (100 mg), and Water for Injection USP, to pH 6.0.
Each 150 mg/mL pre-filled pen contains 150 mg alirocumab, histidine (6 mM), polysorbate 20 (0.1 mg), sucrose (100 mg), and Water for Injection USP, to pH 6.0.
12. Praluent - Clinical Pharmacology
12.1 Mechanism of Action
Alirocumab is a human monoclonal antibody that binds to proprotein convertase subtilisin kexin type 9 (PCSK9). PCSK9 binds to the low-density lipoprotein (LDL) receptors (LDLR) on the surface of hepatocytes to promote LDLR degradation within the liver. By inhibiting the binding of PCSK9 to LDLR, alirocumab increases the number of LDLRs available to clear LDL, thereby lowering LDL-C levels.
12.2 Pharmacodynamics
Alirocumab reduced free PCSK9 in a concentration-dependent manner. Following a single subcutaneous administration of alirocumab 75 or 150 mg, maximal suppression of free PCSK9 occurred within 4 to 8 hours. Free PCSK9 concentrations returned to baseline when alirocumab concentrations decreased below the limit of quantitation.
12.3 Pharmacokinetics
Absorption
After subcutaneous administration of 75 mg to 300 mg alirocumab, median times to maximum serum concentrations (tmax) were 3-7 days. The pharmacokinetics of alirocumab after single subcutaneous administration of 75 mg into the abdomen, upper arm, or thigh were similar. The absolute bioavailability of alirocumab after subcutaneous administration was about 85% as determined by population pharmacokinetics analysis. A slightly greater than dose proportional increase was observed, with a 2.1-fold to 2.7-fold increase in total alirocumab concentrations for a 2-fold increase in dose from 75 mg every 2 weeks to 150 mg every 2 weeks. Monthly dose normalized exposure with 300 mg every 4 weeks treatment was similar to that of 150 mg every 2 weeks. Steady state was reached after 2 to 3 doses with an accumulation ratio up to a maximum of about 2-fold.
Distribution
Following intravenous administration, the volume of distribution was about 0.04 to 0.05 L/kg indicating that alirocumab is distributed primarily in the circulatory system.
Elimination
Specific metabolism studies were not conducted, because alirocumab is a protein. Alirocumab is expected to degrade to small peptides and individual amino acids. In clinical studies where alirocumab was administered in combination with atorvastatin or rosuvastatin, no relevant changes in statin concentrations were observed in the presence of repeated administration of alirocumab, indicating that cytochrome P450 enzymes (mainly CYP3A4 and CYP2C9) and transporter proteins such as P-gp and OATP were not affected by alirocumab.
Two elimination phases were observed for alirocumab. At low concentrations, the elimination is predominately through saturable binding to target (PCSK9), while at higher concentrations the elimination of alirocumab is largely through a non-saturable proteolytic pathway.
Based on a population pharmacokinetic analysis, the median apparent half-life of alirocumab at steady state was 17 to 20 days in patients receiving alirocumab at subcutaneous doses of 75 mg every 2 weeks or 150 mg every 2 weeks.
Specific Populations
A population pharmacokinetic analysis was conducted on data from 2799 patients. Age, body weight, gender, race, and creatinine clearance were found not to significantly influence alirocumab pharmacokinetics.
Pediatric Patients
The pharmacokinetics of alirocumab were evaluated in 140 pediatric patients aged 8 to 17 years with HeFH. Steady-state concentrations were reached at or before week 8 (first PK sampling during repeated dosing) with recommended dosing regimen [see Dosage and Administration (2.2)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the trials described below with the incidence of anti-drug antibodies in other trials, including those of PRALUENT or of other alirocumab products.
In adult patients with cardiovascular disease (Trial 1), the incidence of anti-alirocumab antibody (ADA) formation was 5.5% (504/9,091) in patients treated with PRALUENT 75 mg and/or 150 mg every 2 weeks for up to 5 years (with a median treatment exposure of 31 months). Neutralizing antibody (NAb) responses were observed in 0.5% (43/9,091) of all patients treated with PRALUENT. Of the patients who developed ADA, 8.5% (43/504) tested positive for NAb.
- While reductions in LDL-C were generally comparable in patients with or without ADA, including NAbs, some adult patients treated with PRALUENT with persistent or neutralizing antibodies experienced attenuation in LDL-C efficacy.
- Adult patients who developed ADA had a higher incidence of injection site reactions compared to patients without ADA (7.5% vs 3.6%) [see Adverse Reactions (6.1)].
In a pool of placebo-controlled and active-controlled trials of adult patients treated with PRALUENT 75 mg and/or 150 mg every 2 weeks as well as in a separate clinical trial of patients treated with PRALUENT 75 mg every 2 weeks or 300 mg every 4 weeks (including some patients with dose adjustment to 150 mg every 2 weeks), during the treatment period ranging from 6 to 24 months, the incidence of detecting ADA was 4.8% (147/3,033) and NAb was 1.2% (36/3,033), which was similar to the results from the trial described above.
In pediatric patients aged 8 to 17 years with HeFH (Trial 12), the incidence of ADA for patients treated with PRALUENT was 3% (3/98) with a median treatment exposure of 24 weeks in patients receiving PRALUENT once every 2 weeks and 23 weeks in patients receiving PRALUENT once every 4 weeks with an optional up-titration. Of the 3 pediatric patients who developed ADA, no one tested positive for NAb.
Because of the low occurrence of ADA and the small number of pediatric patients enrolled, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of PRALUENT in pediatric patients is unknown.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with alirocumab. The mutagenic potential of alirocumab has not been evaluated; however, monoclonal antibodies are not expected to alter DNA or chromosomes.
There were no adverse effects on surrogate markers of fertility (e.g., estrous cyclicity, testicular volume, ejaculate volume, sperm motility, or total sperm count per ejaculate) in a 6-month chronic toxicology study in sexually-mature monkeys subcutaneously administered at 5, 15, and 75 mg/kg/week at systemic exposures up to 103-fold the 150 mg every two weeks subcutaneous clinical dose based on serum AUC. In addition, there were no adverse alirocumab-related anatomic pathology or histopathology findings in reproductive tissues in rat or monkey toxicology studies at systemic exposures up to 11-fold and 103-fold respectively, in the 6-month studies, compared to clinical systemic exposure following a 150 mg every two weeks dose, based on serum AUC.
13.2 Animal Toxicology and/or Pharmacology
During a 13-week toxicology study of 75 mg/kg once weekly alirocumab in combination with 40 mg/kg once daily atorvastatin in adult monkeys, there were no effects of PRALUENT on the humoral immune response to keyhole limpet hemocyanin (KLH) after one to two months at exposures 100-fold greater than the exposure at the maximum recommended human dose of 150 mg every two weeks, based on AUC.
14. Clinical Studies
14.1 Cardiovascular Outcome Trial in Adult Patients with Established Cardiovascular Disease
Trial 1 (ODYSSEY OUTCOMES, NTC01663402) was a multicenter, double-blind, placebo-controlled trial in 18,924 adult patients (9,462 PRALUENT; 9,462 placebo) followed for up to 5 years. Patients had an acute coronary syndrome (ACS) event 4 to 52 weeks prior to randomization and were treated with a lipid-modifying therapy (LMT) regimen that was statin-intensive (defined as atorvastatin 40 or 80 mg, or rosuvastatin 20 or 40 mg) or at maximally tolerated dose of a statin, with or without other LMT. Patients were randomized to receive either PRALUENT 75 mg or placebo once every two weeks.
At month 2, if additional LDL-C lowering was required based on pre-specified LDL-C criteria (LDL-C ≥50 mg/dL), PRALUENT was adjusted to 150 mg every 2 weeks. For patients who had their dose adjusted to 150 mg every 2 weeks and who had two consecutive LDL-C values below 25 mg/dL, down-titration from 150 mg every 2 weeks to 75 mg every 2 weeks was performed. Patients on 75 mg every 2 weeks who had two consecutive LDL-C values below 15 mg/dL were switched to placebo in a blinded fashion. Approximately 2,615 (27.7%) of 9,451 patients treated with PRALUENT required dose adjustment to 150 mg every 2 weeks. Of these 2,615 patients, 805 (30.8%) were down-titrated to 75 mg every 2 weeks. Overall, 730 (7.7%) of 9,451 patients switched to placebo.
A total of 99.5% of patients were followed for survival until the end of the trial. The median follow-up duration was 33 months.
Baseline Disease and Demographic Characteristics
The mean age at baseline was 59 years (range 39-92), with 25% female, and 27% at least 65 years old. The trial population was 79% White, 3% Black or African American and 13% Asian; 17% identified as Hispanic or Latino ethnicity. The index ACS event was a myocardial infarction in 83% of patients and unstable angina in 17% of patients. Prior to the index ACS event, 19% had prior myocardial infarction and 23% had coronary revascularization procedures (CABG/PCI). Selected additional baseline risk factors included hypertension (65%), diabetes mellitus (25%), New York Heart Association class I or II congestive heart failure (15%), and eGFR <60 mL/min/1.73 m2 (13%). Most patients (89%) were receiving statin-intensive therapy with or without other LMT at randomization. The mean LDL-C value at baseline was 92.4 mg/dL.
Endpoint Results
PRALUENT significantly reduced the risk for the primary composite endpoint (time to first occurrence of coronary heart disease death, non-fatal myocardial infarction, fatal and non-fatal ischemic stroke, or unstable angina requiring hospitalization: p=0.0003). The results are presented in Table 2.
| Endpoint | PRALUENT N=9,462 | Placebo N=9,462 | Hazard Ratio (95% CI)* |
||
|---|---|---|---|---|---|
| n (%) | Incidence Rate per 100 Patient Years (95% CI) | n (%) | Incidence Rate per 100 Patient Years (95% CI) |
||
|
|||||
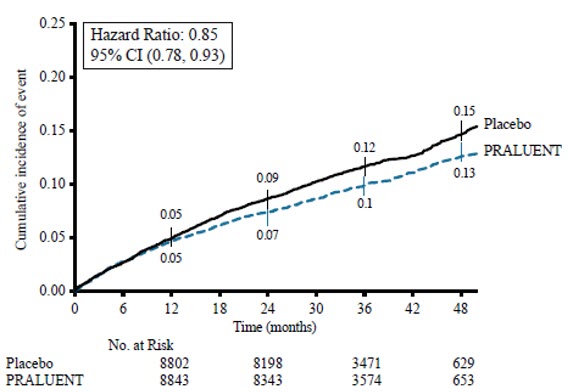
| Primary composite endpoint† | 903 (9.5%) | 3.5 (3.3 to 3.8) | 1052 (11.1%) | 4.2 (3.9 to 4.4) | 0.85 (0.78, 0.93) |
| Components of the Primary Composite Endpoint‡ | |||||
| CHD death | 205 (2.2%) | 0.8 (0.7 to 0.9) | 222 (2.3%) | 0.8 (0.7 to 0.9) | 0.92 (0.76, 1.11) |
| Non-fatal MI§ | 626 (6.6%) | 2.4 (2.2 to 2.6) | 722 (7.6%) | 2.8 (2.6 to 3.0) | 0.86 (0.77, 0.96) |
| Fatal or non-fatal ischemic stroke§ | 111 (1.2%) | 0.4 (0.3 to 0.5) | 152 (1.6%) | 0.6 (0.5 to 0.7) | 0.73 (0.57, 0.93) |
| Unstable angina requiring hospitalization§ | 37 (0.4%) | 0.1 (0.1 to 0.2) | 60 (0.6%) | 0.2 (0.2 to 0.3) | 0.61 (0.41, 0.92) |
| Mortality Endpoint (not statistically significant per pre-specified method to control for type I error) | |||||
| All-cause mortality | 334 (3.5%) | 1.2 (1.1 to 1.4) | 392 (4.1%) | 1.5 (1.3 to 1.6) | 0.85 (0.73, 0.98) |
The Kaplan-Meier estimates of the cumulative incidence of the primary endpoint over time is presented in Figure 1.
Figure 1: Primary Composite Endpoint Cumulative Incidence over 4 Years in ODYSSEY OUTCOMES
14.2 Clinical Trials in Adult Patients with Primary Hyperlipidemia (including HeFH) and HoFH
Primary Hyperlipidemia
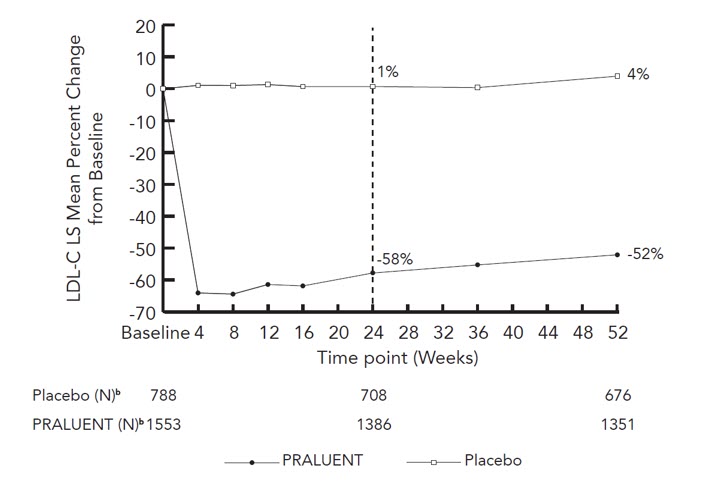
Trial 2 (ODYSSEY LONG TERM, NCT01507831) was a multicenter, double-blind, placebo-controlled trial that randomly assigned 1,553 adult patients to PRALUENT 150 mg every 2 weeks and 788 adult patients to placebo. All patients were taking maximally tolerated doses of statins with or without other lipid-modifying therapy and required additional LDL-C reduction.
Baseline Disease and Demographic Characteristics
The mean age was 61 years (range 18-89), 38% were female, 93% were White, 3% were Black or African American, and 5% identified as Hispanic or Latino ethnicity. The average LDL-C at baseline was 122 mg/dL.
Endpoint Results
At week 24, the treatment difference between PRALUENT and placebo in mean LDL-C percent change was -58% (95% CI: -61%, -56%; p-value: ˂0.0001).
The proportion of patients who prematurely discontinued trial drug prior to the 24-week primary endpoint was 8% among those treated with PRALUENT and 8% among those treated with placebo.
For additional results see Table 3 and Figure 2.
| Treatment Group | LDL-C | Total-C | Non-HDL-C | Apo B |
|---|---|---|---|---|
| Week 24 (Mean Percent Change from Baseline) | ||||
| Placebo (n=788) | 1 | 0 | 1 | 1 |
| PRALUENT 150 mg (n=1,553) | -58 | -36 | -49 | -50 |
| Difference from placebo (LS Mean) (95% CI) | -58 (-61, -56) | -36 (-37, -34) | -50 (-52, -47) | -51 (-53, -48) |
| a The means were estimated based on all randomized patients, with multiple imputation of missing data taking into account treatment adherence |
| b Number of patients with observed data |
| Figure 2: Mean Percent Change from Baseline in LDL-C Over 52 Weeks in Adult Patients on Maximally Tolerated Statin Treated with PRALUENT 150 mg Every 2 Weeks and Placebo Every 2 Weeks (ODYSSEY LONG TERM)a |
|
|
Trial 3 (ODYSSEY COMBO I, NCT01644175) was a multicenter, double-blind, placebo-controlled trial that randomly assigned 209 adult patients to PRALUENT and 107 adult patients to placebo. Patients were taking maximally tolerated doses of statins with or without other lipid-modifying therapy and required additional LDL-C reduction.
Baseline Disease and Demographic Characteristics
The mean age was 63 years (range 39-87), 34% were female, 82% were White, 16% were Black or African American, and 11% were Hispanic or Latino. Mean baseline LDL-C was 102 mg/dL.
Endpoint Results
At week 12, the mean percent change from baseline in LDL-C was -45% with PRALUENT compared to 1% with placebo, and the treatment difference between PRALUENT 75 mg every 2 weeks and placebo in mean LDL-C percent change was -46% (95% CI: -53%, -39%).
At week 12, if additional LDL-C lowering was required based on pre-specified LDL-C criteria, PRALUENT was up-titrated to 150 mg every 2 weeks for the remainder of the trial. The dose was up-titrated to 150 mg every 2 weeks in 32 (17%) of 191 patients treated with PRALUENT for at least 12 weeks. At week 24, the mean percent change from baseline in LDL-C was -44% with PRALUENT and -2% with placebo, and the treatment difference between PRALUENT and placebo in mean LDL-C percent change was -43% (95% CI: -50%, -35%; p-value: <0.0001).
The proportion of patients who prematurely discontinued trial drug prior to the 24-week primary endpoint was 11% among those treated with PRALUENT and 12% among those treated with placebo.
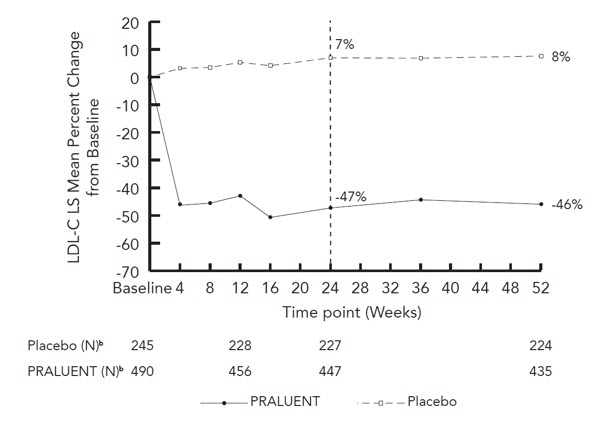
Trials 4 (ODYSSEY FH I, NCT01623115) and 5 (ODYSSEY FH II, NCT01709500) were multicenter, double-blind, placebo-controlled trials that, combined, randomly assigned 490 adult patients to PRALUENT and 245 adult patients to placebo. The trials were similar with regard to both design and eligibility criteria. All patients had HeFH, were taking a maximally tolerated dose of statin with or without other lipid-modifying therapy, and required additional LDL-C reduction. The diagnosis of HeFH was made either by genotyping or clinical criteria ("definite FH" using either the Simon Broome or WHO/Dutch Lipid Network criteria).
Baseline Disease and Demographic Characteristics
The mean age was 52 years (range 20-87), 45% were female, 94% were White, 1% were Black or African American, and 3% identified as Hispanic or Latino ethnicity. The average LDL-C at baseline was 141 mg/dL.
Endpoint Results
At week 12, the treatment difference between PRALUENT 75 mg every 2 weeks and placebo in mean LDL-C percent change was -48% (95% CI: -52%, -44%).
At week 12, if additional LDL-C lowering was required based on pre-specified LDL-C criteria, PRALUENT was up-titrated to 150 mg every 2 weeks for the remainder of the trials. The dose was up-titrated to 150 mg every 2 weeks in 196 (42%) of 469 patients treated with PRALUENT for at least 12 weeks. At week 24, the mean treatment difference between PRALUENT and placebo in mean LDL-C percent change from baseline was -54% (95% CI: -59%, -50%; p-value: <0.0001). The LDL-C-lowering effect was sustained to week 52.
Considering both trials together, the proportion of patients who prematurely discontinued trial drug prior to the 24-week primary endpoint was 6% among those treated with PRALUENT and 4% among those treated with placebo.
For additional results see Table 4 and Figure 3.
| Treatment Group | LDL-C | Total-C | Non-HDL-C | Apo B |
|---|---|---|---|---|
|
||||
| Week 12 (Mean Percent Change from Baseline) | ||||
| Placebo (n=245) | 5 | 4 | 5 | 2 |
| PRALUENT 75 mg (n=490) | -43 | -27 | -38 | -34 |
| Difference from placebo (LS Mean) (95% CI) | -48 (-52, -44) | -31 (-34, -28) | -42 (-46, -39) | -36 (-39, -33) |
| Week 24 (Mean Percent Change from Baseline) | ||||
| Placebo (n=245) | 7 | 5 | 7 | 2 |
| PRALUENT 75 mg/150 mg‡ (n=490) | -47 | -30 | -42 | -40 |
| Difference from placebo (LS Mean) (95% CI) | -54 (-59, -50) | -36 (-39, -33) | -49 (-53, -45) | -42 (-45, -39) |
| a The means were estimated based on all randomized patients, with multiple imputation of missing data taking into account treatment adherence |
| b Number of patients with observed data |
| Figure 3: Mean Percent Change from Baseline in LDL-C Over 52 Weeks in Adult Patients with HeFH on Maximally Tolerated Statin Treated with PRALUENT 75/150 mg Every 2 Weeks and Placebo every 2 weeks (ODYSSEY FH I and FH II Pooled)a |
|
|
Trial 6 (ODYSSEY HIGH FH, NCT01617655) was a multicenter, double-blind, placebo-controlled trial that randomly assigned 72 adult patients to PRALUENT 150 mg every 2 weeks and 35 adult patients to placebo. Patients had HeFH with a baseline LDL-C ≥160 mg/dL while taking a maximally tolerated dose of statin with or without other lipid-modifying therapy.
Baseline Disease and Demographic Characteristics
The mean age was 51 years (range 18-80), 47% were female, 88% were White, 2% were Black or African American, and 6% identified as Hispanic or Latino ethnicity. The average LDL-C at baseline was 198 mg/dL.
Endpoint Results
At week 24, the mean percent change from baseline in LDL-C was -43% with PRALUENT and -7% with placebo, and the treatment difference between PRALUENT and placebo in mean LDL-C percent change was -36% (95% CI: -49%, -24%; p-value: <0.0001).
The proportion of patients who discontinued trial drug prior to the 24-week primary endpoint was 10% among those treated with PRALUENT and 0% among those treated with placebo.
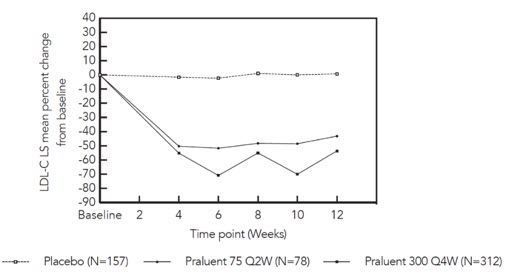
Trial 7 (ODYSSEY CHOICE I, NCT01926782) was a multicenter, double-blind, placebo-controlled trial that randomly assigned 458 adult patients with primary hyperlipidemia to PRALUENT 300 mg every 4 weeks, 115 adult patients to PRALUENT 75 mg every 2 weeks, and 230 adult patients to placebo. Patients were stratified based on whether or not they were treated concomitantly with statin.
Baseline Disease and Demographic Characteristics
The mean age was 61 years (range 21-88), 42% were female, 87% were White, 11% were Black or African American, and 3% identified as Hispanic or Latino ethnicity.
Endpoint Results
In the cohort of patients on background statin, the mean LDL-C at baseline was 113 mg/dL. At week 12, the treatment difference between PRALUENT 300 mg every 4 weeks and placebo in mean percent change in LDL-C from baseline was -54% (97.5% CI: -61%, -48%), and the treatment difference between PRALUENT 75 mg every 2 weeks and placebo in mean percent change in LDL-C was -44% (97.5% CI: -53%, -35%) (Figure 4).
| Figure 4: Mean Percent Change from Baseline in LDL-C up to Week 12 in Adult Patients on Concomitant Statin Treated with PRALUENT 75 mg Every 2 Weeks, PRALUENT 300 mg Every 4 Weeks or Placebo* |
|---|
|
|
|
At week 12, if additional LDL-C lowering was required based on pre-specified LDL-C criteria, PRALUENT was adjusted to 150 mg every 2 weeks for the remainder of the trial. The dose was adjusted to 150 mg every 2 weeks in approximately 20% of patients treated with PRALUENT 75 mg every 2 weeks or 300 mg every 4 weeks for at least 12 weeks.
At week 24, the treatment difference between initial assignment to PRALUENT 300 mg every 4 weeks and placebo in mean percent change in LDL-C from baseline was -56% (97.5% CI: -62%, -49%; p-value: <0.0001), and the treatment difference between initial assignment to PRALUENT 75 mg every 2 weeks and placebo in mean percent change in LDL-C from baseline was -48% (97.5% CI: -57%, -39%).
In the cohort of patients not treated with a concomitant statin, the mean LDL-C at baseline was 142 mg/dL. The treatment difference between PRALUENT and placebo were similar to the cohort of patients treated with a concomitant statin.
The proportion of patients who discontinued trial drug prior to the 24-week primary endpoint was 12% among those treated with PRALUENT 300 mg every 4 weeks, 14% among those treated with PRALUENT 75 mg every 2 weeks, and 15% among those treated with placebo.
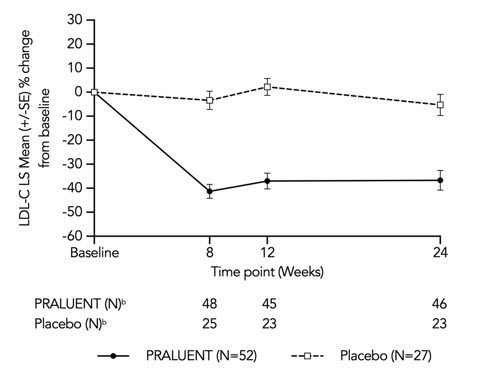
Trial 8 (ODYSSEY ESCAPE, NCT02326220) was a multicenter, double-blind, placebo-controlled trial that randomly assigned adult patients with HeFH who were undergoing LDL apheresis to PRALUENT 150 mg every 2 weeks (N=41) or placebo (N=21). Patients were treated in combination with their usual LDL apheresis schedule for 6 weeks.
Baseline Disease and Demographic Characteristics
The mean age was 59 years (range 27-79), 42% were female, 97% were White, 3% were Black or African American, and 0% identified as Hispanic or Latino ethnicity. The mean LDL-C at baseline, measured before the apheresis procedure, was 181 mg/dL.
Endpoint Results
At week 6, the mean percent change from baseline in pre-apheresis LDL-C was -53% in patients in the PRALUENT group compared to 1% in patients who received placebo.
The proportion of patients who discontinued study drug prior to the 6-week endpoint was 2% among those treated with PRALUENT 150 mg every 2 weeks and 5% among those treated with placebo.
Trial 9 (ODYSSEY COMBO II, NCT01644188) was a multicenter, double-blind, ezetimibe-controlled trial that randomly assigned 479 adult patients to PRALUENT 75 mg every 2 weeks/150 mg every 2 weeks and 241 adult patients to ezetimibe 10 mg/day. Patients were taking a maximally tolerated dose of a statin and required additional LDL-C reduction.
Baseline Disease and Demographic Characteristics
The mean age was 62 years (range 29-88), 26% were female, 85% were White, 4% were Black or African American, and 3% identified as Hispanic or Latino ethnicity. Mean baseline LDL-C was 107 mg/dL.
Endpoint Results
At week 12, the mean percent change from baseline in LDL-C was -50% with PRALUENT compared to -22% with ezetimibe, and the treatment difference between PRALUENT and ezetimibe in mean LDL-C percent change was -28% (95% CI: -32%, -23%).
At week 12, if additional LDL-C lowering was required based on pre-specified LDL-C criteria, PRALUENT was up-titrated to 150 mg every 2 weeks for the remainder of the trial. The dose was up-titrated to 150 mg every 2 weeks in 82 (18%) of 446 patients treated with PRALUENT for at least 12 weeks. At week 24, the mean percent change from baseline in LDL-C was -48% with PRALUENT and -20% with ezetimibe, and the treatment difference between PRALUENT and ezetimibe in mean LDL-C percent change was -28% (95% CI: -33%, -23%; p-value: <0.0001).
The proportion of patients who prematurely discontinued trial drug prior to the 24-week primary endpoint was 9% among those treated with PRALUENT and 10% among those treated with ezetimibe.
Trial 10 (ODYSSEY MONO, NCT01644474) was a multicenter, double-blind, ezetimibe-controlled trial in adult patients with a moderate CV risk, not taking statins or other lipid-modifying therapies, and a baseline LDL-C between 100 mg/dL to 190 mg/dL that randomly assigned 52 patients to PRALUENT 75 mg every 2 weeks and 51 patients to ezetimibe 10 mg/day.
Baseline Disease and Demographic Characteristics
The mean age was 60 years (range 45-72), 47% were female, 90% were White and 10% were Black or African American, and 1% identified as Hispanic or Latino ethnicity. Mean baseline LDL-C was 140 mg/dL.
Endpoint Results
At week 12, the mean percent change from baseline in LDL-C was -48% with PRALUENT compared to -19% with ezetimibe, and the treatment difference between PRALUENT 75 mg every 2 weeks and ezetimibe in mean LDL-C percent change was -29% (95% CI: -37%, -22%). At week 12, if additional LDL-C lowering was required based on pre-specified LDL-C criteria, PRALUENT was up-titrated to 150 mg every 2 weeks for the remainder of the trial. The dose was up-titrated to 150 mg every 2 weeks in 14 (30%) of 46 patients treated with PRALUENT for at least 12 weeks. At week 24, the mean percent change from baseline in LDL-C was -45% with PRALUENT and -14% with ezetimibe, and the treatment difference between PRALUENT and ezetimibe in mean LDL-C percent change was -31% (95% CI: -40%, -22%; p-value: <0.0001).
The proportion of patients who prematurely discontinued trial drug prior to the 24-week endpoint was 15% among those treated with PRALUENT and 14% among those treated with ezetimibe.
Adult Patients with HoFH
Trial 11 (ODYSSEY HoFH, NCT03156621) was a multicenter, double-blind, placebo-controlled trial that randomly assigned 45 adult patients to PRALUENT 150 mg every 2 weeks and 24 adult patients to placebo. Patients were taking maximally tolerated doses of statins with or without other lipid-lowering therapy and required additional LDL-C reduction. Randomization was stratified by LDL apheresis treatment status. The diagnosis of HoFH was made by either clinical diagnosis, which included a history of an untreated total cholesterol concentration >500 mg/dL together with either xanthoma before 10 years of age or with a history of total cholesterol >250 mg in both parents, or by genetic testing.
Baseline Disease and Demographic Characteristics
The mean age was 43 years (range 19-81), 51% were female, 78% were White, 3% were Black or African American, 17% were Asian, and 3% identified as Hispanic or Latino ethnicity. Mean baseline LDL-C was 283 mg/dL with 97% on statins, 72% on ezetimibe, and 14% on lomitapide.
Endpoint Results
At week 12, the treatment difference between PRALUENT and placebo in mean LDL-C percent change from baseline was -36% (95% CI: -51% to -20%; p <0.0001) (see Figure 5). For the effect of PRALUENT on lipid parameters as compared to placebo, see Table 5. No patient discontinued from the trial prior to the 12-week primary endpoint.
Patients with two LDL-receptor negative alleles (little to no residual function) had a minimal to absent response to PRALUENT.
Figure 5: LS Mean Percent Change from Baseline in LDL-C Over 12 Weeks in Adult Patients with HoFH (ODYSSEY HoFH)
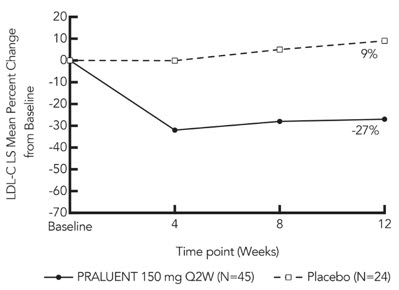
| Treatment Group | LDL-C | Apo B | Non-HDL-C | Total Cholesterol |
|---|---|---|---|---|
| Placebo (n=24) | 9 | 7 | 8 | 7 |
| PRALUENT 150 mg every 2 weeks (n=45) | -27 | -23 | -25 | -20 |
| Difference from placebo (LS Mean) (95% CI) | -36 (-51, -20) | -30 (-42, -17) | -33 (-48, -18) | -27 (-39, -14) |
14.3 Clinical Trials in Pediatric Patients with HeFH
Trial 12 (EFC14643, NCT03510884) was a randomized, multicenter, placebo controlled, double blind, 24 week trial in 153 pediatric patients aged 8 to 17 years with HeFH. Patients were on a low-fat diet and receiving background lipid-lowering therapy.
Patients were randomized in a 2:1 ratio to receive PRALUENT or placebo. In the PRALUENT group dosed every 2 weeks, 49 patients received a dose of 40 mg for body weight less than 50 kg or 75 mg for body weight 50 kg or more. The 40 mg dosage every 2 weeks is not approved [see Dosage and Administration (2.2)]. In the PRALUENT group dosed every 4 weeks, 52 patients received a dose of 150 mg for body weight less than 50 kg or 300 mg for body weight 50 kg or more. Dose adjustment of PRALUENT to 75 mg every 2 weeks for body weight less than 50 kg or 150 mg every 2 weeks for body weight 50 kg or more occurred at week 12 in patients with LDL-C ≥110 mg/dL.
Baseline Disease and Demographic Characteristics
The diagnosis of HeFH was made based on criteria from Simon Broome Register Group (1991) or by genetic testing. The mean age was 13 years (range: 8 to 17 years); 57% female; 82% White, 2% Black or African American, 10% American Indian or Alaska Native, and <1% not reported; 18% Hispanic/Latino ethnicity. Mean body weight was 53 kg. The mean LDL-C at baseline was 174 mg/dL; Of the patients receiving PRALUENT once every 2 weeks with an optional up-titration, 99% were on statins and 7% were on ezetimibe at baseline. Of the patients receiving PRALUENT once every 4 weeks with an optional up-titration, 91% were on statins and 20% were on ezetimibe at baseline.
Endpoint Results
At week 24 in the group receiving treatment every 4 weeks, the treatment difference between the PRALUENT and placebo groups in LS mean LDL-C percent change from baseline was -31.4% (97.5% CI: -45.0 to -17.9; p<0.0001) (see Table 6 and Figure 6). For the effect of PRALUENT on lipid parameters as compared to placebo see Table 6.
| a A pattern-mixture approach was used with multiple imputation of missing post-treatment values based on a patient's own baseline value and multiple imputation of missing on-treatment values based on a model including available on-treatment values |
| b Number of patients with observed data |
| Figure 6: LDL-C LS Mean Percent Change from Baseline Over Time Through Week 24 in Pediatric Patients (aged 8 to 17 years) with HeFH Treated with PRALUENT Every 4 Weeks or Placeboa |
|
|
| Treatment Group* | LDL-C | Apo B | Non-HDL-C | Total Cholesterol |
|---|---|---|---|---|
| PRALUENT once every 4 weeks (150 mg for body weight less than 50 kg or 300 mg for body weight 50 kg or more) † | ||||
|
||||
| LS Mean: Placebo (n=27) | -5.2 | -3.3 | -4.1 | -4.4 |
| LS Mean: PRALUENT (n=52) | -36.6 | -33.0 | -34.2 | -26.7 |
| LS Mean Difference from Placebo‡ (97.5% CI) | -31.4 (-45.0, -17.9) | -29.7 (-41.1, -18.2) | -30.1 (-43.0, -17.2) | -22.3 (-33.0, -11.6) |
| PRALUENT once every 2 weeks (40 mg for body weight less than 50 kg or 75 mg for body weight 50 kg or more)§,¶ | ||||
| LS Mean: Placebo (n=25) | 9.7 | 10.4 | 9.7 | 7.4 |
| LS Mean: PRALUENT (n=49) | -31.9 | -25.7 | -29.5 | -22.4 |
| LS Mean Difference from Placebo‡ (97.5% CI) | -41.7 (-54.2, -29.1) | -36.2 (-45.8, -26.5) | -39.2 (-50.6, -27.8) | -29.8 (-38.7, -21.0) |
16. How is Praluent supplied
PRALUENT injection is a clear, colorless to pale yellow solution, supplied as follows:
| Pack Size | 75 mg/mL Pre-filled Pen | 150 mg/mL Pre-filled Pen |
|---|---|---|
| Pack of 1 pen | NDC 0024-5901-01 | NDC 0024-5902-01 |
| Pack of 2 pens | NDC 0024-5901-02 NDC 72733-5901-2 | NDC 0024-5902-02 NDC 72733-5902-2 |
The needle shield is not made with natural rubber latex.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Pregnancy
Advise women who are exposed to PRALUENT during pregnancy that there is a pregnancy safety study that monitors pregnancy outcomes. Encourage these patients to report their pregnancy to Regeneron at 1 844-734-6643 [see Use in Specific Populations (8.1)].
Hypersensitivity Reactions
Inform patients that serious hypersensitivity reactions (e.g., angioedema) have been reported in patients treated with PRALUENT. Advise patients on the symptoms of hypersensitivity reactions and instruct them to discontinue PRALUENT and seek medical attention promptly, if such symptoms occur.
Administration
Provide guidance to patients and caregivers on proper subcutaneous injection technique and how to use the pre-filled pen. Inform patients that it may take up to 20 seconds to inject PRALUENT. Inform patients the pre-filled pen should be allowed to warm to room temperature for 30 to 40 minutes prior to use if refrigerated.
Manufactured by:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
U.S. License # 1752
Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807)
and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
PRALUENT is a registered trademark of Sanofi
© 2024 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: March 2024 | |||
| Patient Information PRALUENT® (PRAHL-u-ent) (alirocumab) injection, for subcutaneous use |
||||
| What is PRALUENT?
PRALUENT is an injectable prescription medicine used:
|
||||
| Who should not use PRALUENT?
Do not use PRALUENT if you are allergic to alirocumab or to any of the ingredients in PRALUENT. See the end of this leaflet for a complete list of ingredients in PRALUENT. |
||||
| What should I tell my healthcare provider before using PRALUENT?
Before you start using PRALUENT, tell your healthcare provider about all of your medical conditions, including allergies, and if you:
Tell your healthcare provider or pharmacist about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
||||
How should I use PRALUENT?
|
||||
| What are the possible side effects of PRALUENT?
PRALUENT can cause serious side effects, including:
|
||||
|
|
|
||
| The common side effects of PRALUENT include: | ||||
|
|
|||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of PRALUENT. Ask your healthcare provider or pharmacist for more information. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
| General information about the safe and effective use of PRALUENT.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PRALUENT for a condition for which it was not prescribed. Do not give PRALUENT to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information summarizes the most important information about PRALUENT. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about PRALUENT that is written for health professionals. For more information about PRALUENT, go to www.PRALUENT.com or call 1-844-PRALUENT (1-844-772-5836). |
||||
| What are the ingredients in PRALUENT? | ||||
|
|
|||
| Manufactured by: sanofi-aventis U.S. LLC, Bridgewater, NJ 08807; A SANOFI COMPANY, U.S. License # 1752; Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) / PRALUENT is a registered trademark of Sanofi / ©2024 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC | ||||
Instructions For Use
PRALUENT® (PRAHL-u-ent)
(alirocumab)
Injection, for Subcutaneous Injection
Single-Dose Pre-Filled Pen (75 mg/mL)
| Important Information | |
|---|---|
|
|
Storage of PRALUENT
|
|
Keep this leaflet. If you have questions, ask your healthcare provider or call 1-844-PRALUENT (1-844-772-5836).
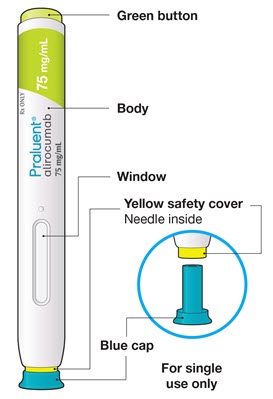
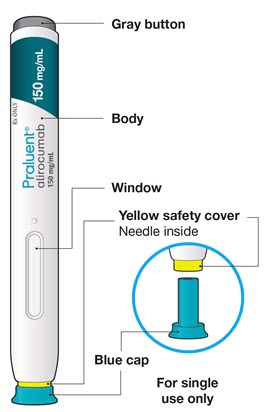
The parts of the PRALUENT pen are shown in this picture.
Step A: Getting ready for your injection.
Before you start you will need:
- the PRALUENT pen
- 1 alcohol wipe
- 1 cotton ball or gauze
- a sharps container or a puncture-resistant container (see Step B8)
A1: Look at the label on the pen.
- Check that you have the correct product and the correct dose.
- Check the expiration date (EXP): do not use if this date has passed.

A2: Look at the window.
- Check the liquid is clear, colorless to pale yellow and free from particles (see Figure A).
- You may see air bubbles. This is normal.
- Do not use if the window appears solid yellow (see Figure B).
- Do not use this medicine if the solution is discolored or cloudy, or if it contains visible flakes or particles.
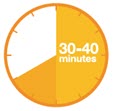
A3: Let the pen warm up at room temperature for 30 to 40 minutes.
- This is important for administering the entire dose and helps minimize discomfort.
- Take PRALUENT out of the refrigerator to warm up before using.
- Do not heat the pen, let it warm up on its own.
- Do not put the pen back in the refrigerator.
A4: Prepare the injection site.
- Wash your hands with soap and water and dry with a towel.
- Clean skin in the injection area with an alcohol wipe.
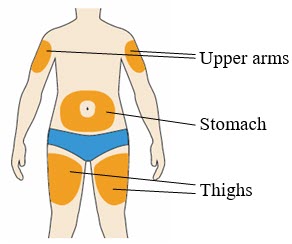
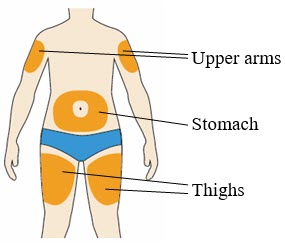
- You can inject into your (see picture):
- thighs
- stomach (except for the 2 inch area around your belly button)
- upper arms
- You can stand or sit to give yourself an injection.
Important:
- Change (rotate) your injection site each time you give yourself an injection. If you need to use the same injection site, make sure it is not the same spot on the site you used last time.
- Do not inject into areas where the skin is tender, bruised, hard, or red. Do not inject PRALUENT into areas with visible veins, scars or stretch marks.
Step B: How to give your injection.
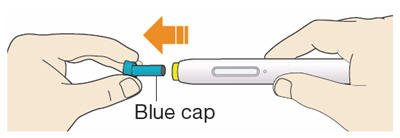
B1: After completing all steps in "Step A: Getting ready for your injection", pull off the blue cap.
- Do not pull off the cap until you are ready to inject.
- Do not put the blue cap back on.
B2: Hold the PRALUENT pen like this.
- Do not touch the yellow safety cover.
- Make sure you can see the window.
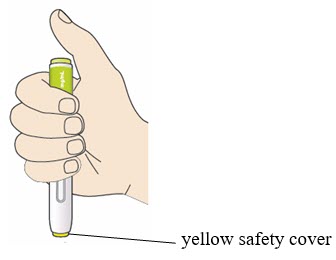
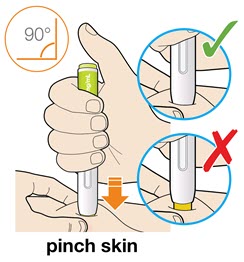
B3: Press the yellow safety cover on your skin at roughly a 90° angle.
- For children younger than 12 years of age, pinching the skin before and during the injection is required.
- In adults and children aged 12 years and older, pinching of skin may be required to make the injection site firm.
- Press and firmly hold the pen against your body until the yellow safety cover is no longer visible. The pen will not work if the yellow safety cover is not depressed fully.
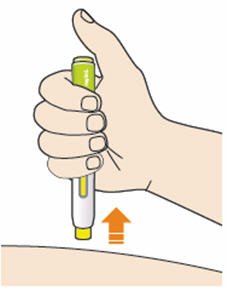
B4: Push and immediately release the green button with your thumb.
- You will hear a click. Your injection has now started.
- The window will start to turn yellow.
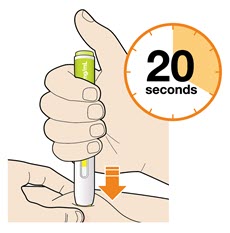
B5: Keep holding the pen against your skin after releasing the button.
- The injection may take up to 20 seconds.
B6: Check the window has turned yellow, before removing the pen.
- Do not remove the pen until the entire window has turned yellow.
- Your injection is complete when the window has turned completely yellow, you may hear a second click.
- If the window does not turn completely yellow, call 1-844-772-5836 for help. Do not give yourself a second dose without speaking to your healthcare provider.

B7: Pull pen away from your skin.
- Do not rub the skin after the injection.
- If you see any blood, press a cotton ball or gauze on the site until the bleeding stops.
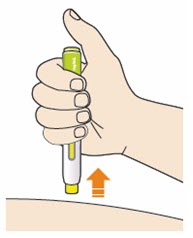
B8: Throw away (Discard) pen and cap.
- Do not put the blue cap back on.
- Throw away pen and cap in a puncture-resistant container immediately after they have been used.
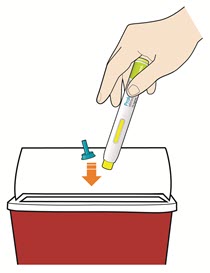
Disposing of used pens:
- Put your used pens in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) pens and caps in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Keep PRALUENT and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
U.S. License # 1752
Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807)
and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
PRALUENT is a registered trademark of Sanofi
©2024 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC
Revised: March 2024
Instructions For Use
PRALUENT® (PRAHL-u-ent)
(alirocumab)
Injection, for Subcutaneous Injection
Single-Dose Pre-Filled Pen (150 mg/mL)
| Important Information | |
|---|---|
|
|
Storage of PRALUENT
|
|
Keep this leaflet. If you have questions, ask your healthcare provider or call 1-844-PRALUENT (1-844-772-5836).
The parts of the PRALUENT pen are shown in this picture.
Step A: Getting ready for your injection.
Before you start you will need:
- the PRALUENT pen
- 1 alcohol wipe
- 1 cotton ball or gauze
- a sharps container or a puncture-resistant container (see Step B8)
A1: Look at the label on the pen.
- Check that you have the correct product and the correct dose.
- Check the expiration date (EXP): do not use if this date has passed.

A2: Look at the window.
- Check the liquid is clear, colorless to pale yellow and free from particles (see Figure A).
- You may see air bubbles. This is normal.
- Do not use if the window appears solid yellow (see Figure B).
- Do not use this medicine if the solution is discolored or cloudy, or if it contains visible flakes or particles.
A3: Let the pen warm up at room temperature for 30 to 40 minutes.
- This is important for administering the entire dose and helps minimize discomfort.
- Take PRALUENT out of the refrigerator to warm up before using.
- Do not heat the pen, let it warm up on its own.
- Do not put the pen back in the refrigerator.
A4: Prepare the injection site.
- Wash your hands with soap and water and dry with a towel.
- Clean skin in the injection area with an alcohol wipe.
- You can inject into your (see picture):
- thighs
- stomach (except for the 2 inch area around your belly button)
- upper arms
- You can stand or sit to give yourself an injection.
Important:
- Change (rotate) your injection site each time you give yourself an injection. If you need to use the same injection site, make sure it is not the same spot on the site you used last time.
- Do not inject into areas where the skin is tender, bruised, hard, or red. Do not inject PRALUENT into areas with visible veins, scars or stretch marks.
Step B: How to give your injection.
B1: After completing all steps in "Step A: Getting ready for your injection", pull off the blue cap.
- Do not pull off the cap until you are ready to inject.
- Do not put the blue cap back on.
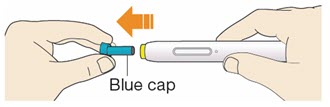
B2: Hold the PRALUENT pen like this.
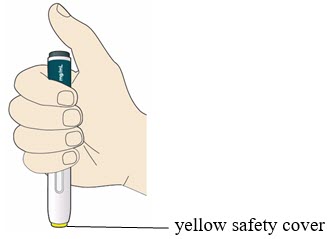
- Do not touch the yellow safety cover.
- Make sure you can see the window.
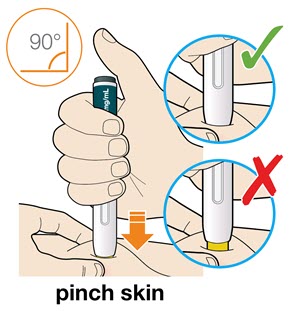
B3: Press the yellow safety cover on your skin at roughly a 90° angle.
- For children younger than 12 years of age, pinching the skin before and during the injection is required.
- In adults and children aged 12 years and older, pinching of skin may be required to make the injection site firm.
- Press and firmly hold the pen against your body until the yellow safety cover is no longer visible. The pen will not work if the yellow safety cover is not depressed fully.
B4: Push and immediately release the gray button with your thumb.
- You will hear a click. Your injection has now started.
- The window will start to turn yellow.
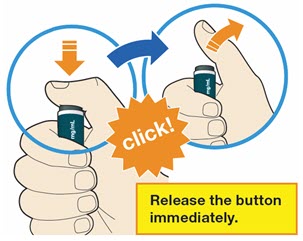
B5: Keep holding the pen against your skin after releasing the button.
- The injection may take up to 20 seconds.
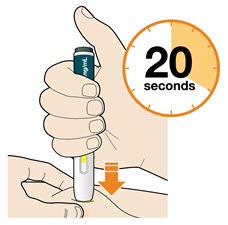
B6: Check the window has turned yellow, before removing the pen.
- Do not remove the pen until the entire window has turned yellow.
- Your injection is complete when the window has turned completely yellow, you may hear a second click.
- If the window does not turn completely yellow, call 1-844-772-5836 for help. Do not give yourself a second dose without speaking to your healthcare provider.

B7: Pull pen away from your skin.
- Do not rub the skin after the injection.
- If you see any blood, press a cotton ball or gauze on the site until the bleeding stops.
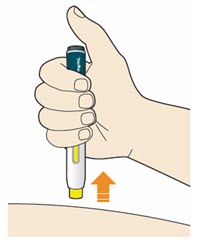
B8: Throw away (Discard) pen and cap.
- Do not put the blue cap back on.
- Throw away pen and cap in a puncture-resistant container immediately after they have been used.
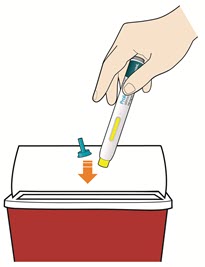
Disposing of used pens:
- Put your used pens in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) pens and caps in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Keep PRALUENT and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
U.S. License # 1752
Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807)
and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
PRALUENT is a registered trademark of Sanofi
©2024 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC
Revised: March 2024
PRINCIPAL DISPLAY PANEL - 75 mg/mL Pen Carton
NDC 0024-5901-01
Rx ONLY
Praluent®
alirocumab
Injection
75 mg/mL
One Pre-filled Pen
For subcutaneous injection only. Single-dose.
Carton contains: One single-dose pre-filled pen, the Package Insert,
Patient Information, and Instructions for Use.
75 mg/mL
↓
OPEN
HERE
REGENERON
SANOFI
PRINCIPAL DISPLAY PANEL - 150 mg/mL Pen Carton
NDC 0024-5902-01
Rx ONLY
Praluent®
alirocumab
Injection
150 mg/mL
One Pre-filled Pen
For subcutaneous injection only. Single-dose.
Carton contains: One single-dose pre-filled pen, the Package Insert,
Patient Information, and Instructions for Use.
150 mg/mL
↓
OPEN
HERE
REGENERON
SANOFI
PRINCIPAL DISPLAY PANEL - 75 mg/mL Syringe Carton
NDC 0024-5903-01
Rx ONLY
Praluent®
alirocumab
Injection
75 mg/mL
One Pre-filled Syringe
For subcutaneous injection only. Single-dose.
Carton contains: One single-dose pre-filled syringe,
the Package Insert, Patient Information, and Instructions for Use.
75 mg/mL
▶ OPEN
REGENERON
SANOFI
PRINCIPAL DISPLAY PANEL - 150 mg/mL SyringeCarton
NDC 0024-5904-01
Rx ONLY
Praluent®
alirocumab
Injection
150 mg/mL
One Pre-filled Syringe
For subcutaneous injection only. Single-dose.
Carton contains: One single-dose pre-filled syringe,
the Package Insert, Patient Information, and Instructions for Use.
150 mg/mL
▶ OPEN
REGENERON
SANOFI
| PRALUENT
alirocumab injection, solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| PRALUENT
alirocumab injection, solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| PRALUENT
alirocumab injection, solution |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PRALUENT
alirocumab injection, solution |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Sanofi-Aventis U.S. LLC (824676584) |
| Registrant - Regeneron Pharmaceuticals, Inc. (194873139) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Regeneron Pharmaceuticals, Inc. | 945589711 | ANALYSIS(0024-5901, 0024-5902, 0024-5903, 0024-5904) , API MANUFACTURE(0024-5901, 0024-5902, 0024-5903, 0024-5904) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sanofi Winthrop Industrie | 297730488 | ANALYSIS(0024-5903, 0024-5904) , LABEL(0024-5903, 0024-5904) , MANUFACTURE(0024-5903, 0024-5904) , PACK(0024-5903, 0024-5904) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sanofi Pasteur Inc. | 086723285 | MANUFACTURE(0024-5903, 0024-5904) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Regn Ireland DAC | 986063166 | ANALYSIS(0024-5901, 0024-5902, 0024-5903, 0024-5904) , API MANUFACTURE(0024-5901, 0024-5902, 0024-5903, 0024-5904) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sanofi-Aventis Deutschland GmbH | 313218430 | ANALYSIS(0024-5901, 0024-5902) , LABEL(0024-5901, 0024-5902) , MANUFACTURE(0024-5901, 0024-5902) , PACK(0024-5901, 0024-5902) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Genzyme Ireland Limited | 985127419 | ANALYSIS(0024-5901, 0024-5902) , LABEL(0024-5901, 0024-5902) , MANUFACTURE(0024-5901, 0024-5902) , PACK(0024-5901, 0024-5902) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sanofi Winthrop Industrie | 280366666 | ANALYSIS(0024-5901, 0024-5902, 0024-5903, 0024-5904) , API MANUFACTURE(0024-5901, 0024-5902, 0024-5903, 0024-5904) | |
Biological Products Related to Praluent
Find detailed information on biosimilars for this medication.
Frequently asked questions
- Praluent vs Repatha: What's the difference?
- What are PCSK9 Inhibitors and how do they work?
- How does Praluent work?
- How do you administer a Praluent injection?
- Does Praluent (alirocumab) lower triglycerides?
More about Praluent (alirocumab)
- Check interactions
- Compare alternatives
- Reviews (306)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- FDA approval history
- Drug class: PCSK9 inhibitors
- Breastfeeding
- En español