211 5 b Pill: white, oval, 11mm
The pill with imprint 211 5 b (White, Oval, 11mm) has been identified as Norethindrone Acetate 5 mg and is used for Abnormal Uterine Bleeding, Endometriosis, Amenorrhea, and Birth Control. It belongs to the drug classes contraceptives, Progestogens and is not a controlled substance.
Images for 211 5 b


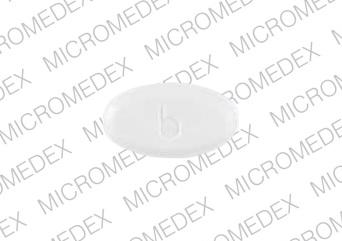
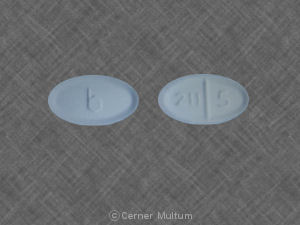
Norethindrone Acetate
- Imprint
- 211 5 b
- Strength
- 5 mg
- Color
- White
- Size
- 11.00 mm
- Shape
- Oval
- Availability
- Prescription only
- Drug Class
- Contraceptives, Progestogens
- Pregnancy Category
- X - Not for use in pregnancy
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Teva Pharmaceuticals USA
- Inactive Ingredients
-
lactose anhydrous,
magnesium stearate,
microcrystalline cellulose
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 00555-0211 | Barr Laboratories, Inc. |
| 54868-4829 | Physicians Total Care Inc. (repackager) |
See also:
More about norethindrone
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (1,579)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: contraceptives
- Breastfeeding
Patient resources
- Norethindrone drug information
- Norethindrone (Contraceptive)
- Norethindrone (Hormone Replacement Therapy)
Other brands
Gallifrey, Errin, Ortho Micronor, Camila, ... +4 more
Professional resources
Other brands
Aygestin, Gallifrey, Errin, Jencycla, ... +16 more
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
