Corlanor: Package Insert / Prescribing Info
Package insert / product label
Generic name: ivabradine hydrochloride
Dosage form: tablet, film coated
Drug class: Miscellaneous cardiovascular agents
Medically reviewed by Drugs.com. Last updated on Jun 9, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
- Medication Guide
Highlights of Prescribing Information
CORLANOR® (ivabradine) tablets, for oral use
CORLANOR® (ivabradine) oral solution
Initial U.S. Approval: 2015
Indications and Usage for Corlanor
Corlanor (ivabradine) is a hyperpolarization-activated cyclic nucleotide-gated channel blocker indicated:
- To reduce the risk of hospitalization for worsening heart failure in adult patients with stable, symptomatic chronic heart failure with reduced left ventricular ejection fraction. (1.1)
- For the treatment of stable symptomatic heart failure due to dilated cardiomyopathy in pediatric patients ages 6 months and older. (1.2)
Corlanor Dosage and Administration
Adult and pediatric patients greater than 40 kg
- Starting dose is 2.5 (pediatrics and vulnerable adults) or 5 mg twice daily with food. After 2 weeks of treatment, adjust dose based on heart rate. The maximum dose is 7.5 mg twice daily. (2.1)
Pediatric patients less than 40 kg
- Starting dose is 0.05 mg/kg twice daily with food. Adjust dose at two-week intervals by 0.05 mg/kg based on heart rate. Maximum dose is 0.2 mg/kg (patients 6 months to less than 1 year old) or 0.3 mg/kg (patients 1 year old and older), up to a total of 7.5 mg twice daily.
Contraindications
- Acute decompensated heart failure (4)
- Clinically significant hypotension (4)
- Sick sinus syndrome, sinoatrial block or 3rd degree AV block, unless a functioning demand pacemaker is present (4)
- Clinically significant bradycardia (4)
- Severe hepatic impairment (4)
- Heart rate maintained exclusively by the pacemaker (4)
- In combination with strong cytochrome CYP3A4 inhibitors (4)
Warnings and Precautions
Adverse Reactions/Side Effects
Most common adverse reactions occurring in ≥ 1% of patients are bradycardia, hypertension, atrial fibrillation and luminous phenomena (phosphenes). (6)
To report SUSPECTED ADVERSE REACTIONS, contact Amgen Medical Information at 1-800-772-6436 (1-800-77-AMGEN) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Use In Specific Populations
- Lactation: Breastfeeding not recommended. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 8/2021
Full Prescribing Information
1. Indications and Usage for Corlanor
1.1 Heart Failure in Adult Patients
Corlanor is indicated to reduce the risk of hospitalization for worsening heart failure in adult patients with stable, symptomatic chronic heart failure with left ventricular ejection fraction ≤ 35%, who are in sinus rhythm with resting heart rate ≥ 70 beats per minute and either are on maximally tolerated doses of beta-blockers or have a contraindication to beta-blocker use.
2. Corlanor Dosage and Administration
2.1 Adults
The recommended starting dose of Corlanor is 5 mg twice daily with food. Assess patient after two weeks and adjust dose to achieve a resting heart rate between 50 and 60 beats per minute (bpm) as shown in Table 1. Thereafter, adjust dose as needed based on resting heart rate and tolerability. The maximum dose is 7.5 mg twice daily. In adult patients unable to swallow tablets, Corlanor oral solution can be used [see Clinical Pharmacology (12.3)].
In patients with a history of conduction defects or other patients in whom bradycardia could lead to hemodynamic compromise, initiate therapy at 2.5 mg twice daily before increasing the dose based on heart rate [see Warnings and Precautions (5.3)].
| Heart Rate | Dose Adjustment |
| > 60 bpm | Increase dose by 2.5 mg (given twice daily) up to a maximum dose of 7.5 mg twice daily |
| 50-60 bpm | Maintain dose |
| < 50 bpm or signs and symptoms of bradycardia | Decrease dose by 2.5 mg (given twice daily); if current dose is 2.5 mg twice daily, discontinue therapy* |
*[see Warnings and Precautions (5.3)]
2.2 Pediatric Patients
Recommended Dosage
Pediatric Patients 6 Months of Age and Older Weighing Less than 40 kg (Oral Solution)
The recommended starting dose of Corlanor oral solution in pediatric patients 6 months of age and older and weighing less than 40 kg is 0.05 mg/kg twice daily with food. Assess patient at two-week intervals and adjust dose by 0.05 mg/kg to target a heart rate (HR) reduction of at least 20%, based on tolerability. The maximum dose is 0.2 mg/kg twice daily for patients 6 months to less than 1 year old, and 0.3 mg/kg twice daily for patients 1 year old and older, up to a total of 7.5 mg twice daily.
If a dose of Corlanor is missed or spit out, do not give another dose to make up for the missed or spit out dose. Give the next dose at the usual time.
Pediatric Patients Weighing 40 kg and Greater (Tablets)
The recommended starting dose of Corlanor tablets in pediatric patients weighing more than 40 kg is 2.5 mg twice daily with food. Assess patient at two-week intervals and adjust dose by 2.5 mg to target a heart rate (HR) reduction of at least 20%, based on tolerability. The maximum dose is 7.5 mg twice daily. In patients unable to swallow tablets, Corlanor oral solution can be used.
Dose Reduction for Bradycardia
If bradycardia develops, reduce the dose to the previous titration step. In patients who develop bradycardia at the recommended initial dosage, consider reducing the dosage to 0.02 mg/kg twice daily.
Oral Solution Preparation and Administration
To administer Corlanor oral solution, empty the entire contents of the ampule(s) into a medication cup. With a calibrated oral syringe, measure the prescribed dose of Corlanor from the medication cup and administer the drug orally.
Corlanor oral solution is sterile and preservative-free. Discard the unused oral solution. Do not store or reuse any oral solution left in either the medication cup or an ampule.
3. Dosage Forms and Strengths
Corlanor Tablets
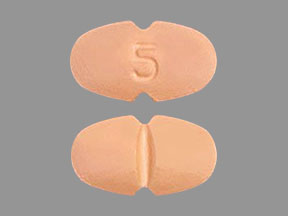
5 mg: salmon-colored, oval-shaped, film-coated tablet, functionally scored on both edges, debossed with “5” on one face and bisected on the other face. The tablet is scored and can be divided into equal halves to provide a 2.5 mg dose.
7.5 mg: salmon-colored, triangular-shaped, film-coated tablet debossed with “7.5” on one face and plain on the other face.
Corlanor Oral Solution
Corlanor 5 mg/5 mL (1 mg/mL) oral solution is a colorless liquid in an opaque, plastic, ampule containing 5 mg of Corlanor in 5 mL of liquid.
4. Contraindications
Corlanor is contraindicated in patients with:
• Acute decompensated heart failure
• Clinically significant hypotension
• Sick sinus syndrome, sinoatrial block or 3rd degree AV block, unless a functioning demand pacemaker is present
• Clinically significant bradycardia [see Warnings and Precautions (5.3)]
• Severe hepatic impairment [see Use in Specific Populations (8.6)]
• Pacemaker dependence (heart rate maintained exclusively by the pacemaker) [see Drug Interactions (7.3)]
• Concomitant use of strong cytochrome P450 3A4 (CYP3A4) inhibitors [see Drug Interactions (7.1)]
5. Warnings and Precautions
5.1 Fetal Toxicity
Corlanor may cause fetal toxicity when administered to a pregnant woman based on findings in animal studies. Embryo-fetal toxicity and cardiac teratogenic effects were observed in fetuses of pregnant rats treated during organogenesis at exposures 1 to 3 times the human exposures (AUC0-24hr) at the maximum recommended human dose (MRHD) [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception when taking Corlanor [see Use in Specific Populations (8.3)].
5.2 Atrial Fibrillation
Corlanor increases the risk of atrial fibrillation. In the Systolic Heart Failure Treatment with the If Inhibitor Ivabradine Trial (SHIFT), the rate of atrial fibrillation was 5.0% per patient-year in patients treated with Corlanor and 3.9% per patient-year in patients treated with placebo [see Clinical Studies (14)]. Regularly monitor cardiac rhythm. Discontinue Corlanor if atrial fibrillation develops.
5.3 Bradycardia and Conduction Disturbances
Adult Patients
Bradycardia, sinus arrest, and heart block have occurred with Corlanor. The rate of bradycardia was 6.0% per patient-year in patients treated with Corlanor (2.7% symptomatic; 3.4% asymptomatic) and 1.3% per patient-year in patients treated with placebo. Risk factors for bradycardia include sinus node dysfunction, conduction defects (e.g., 1st or 2nd degree atrioventricular block, bundle branch block), ventricular dyssynchrony, and use of other negative chronotropes (e.g., digoxin, diltiazem, verapamil, amiodarone). Bradycardia may increase the risk of QT prolongation which may lead to severe ventricular arrhythmias, including torsade de pointes, especially in patients with risk factors such as use of QTc prolonging drugs [see Adverse Reactions (6.2)].
Concurrent use of verapamil or diltiazem will increase Corlanor exposure, may themselves contribute to heart rate lowering, and should be avoided [see Clinical Pharmacology (12.3)]. Avoid use of Corlanor in patients with 2nd degree atrioventricular block unless a functioning demand pacemaker is present [see Contraindications (4)].
Pediatric Patients
Bradycardia and first-degree heart block were observed in pediatric patients treated with Corlanor. Asymptomatic and symptomatic bradycardia were observed in 6.8% and 4.1% of pediatric patients treated with Corlanor, respectively. In the placebo treatment arm, 2.4% of pediatric patients had asymptomatic bradycardia, but none had symptomatic bradycardia. Bradycardia was managed through dose titration but did not result in study drug discontinuation [see Dosage and Administration (2.2)].
6. Adverse Reactions/Side Effects
Clinically significant adverse reactions that appear in other sections of the labeling include:
- Atrial Fibrillation [see Warnings and Precautions (5.2)]
- Bradycardia and Conduction Disturbances [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adult Patients with Heart Failure
In SHIFT, safety was evaluated in 3,260 patients treated with Corlanor and 3,278 patients given placebo. The median duration of Corlanor exposure was 21.5 months.
The most common adverse drug reactions in the SHIFT trial are shown in Table 2 [see Warnings and Precautions (5.2), (5.3)].
| Ivabradine
N = 3,260 | Placebo
N = 3,278 |
|
| Bradycardia | 10% | 2.2% |
| Hypertension, blood pressure increased | 8.9% | 7.8% |
| Atrial fibrillation | 8.3% | 6.6% |
| Phosphenes, visual brightness | 2.8% | 0.5% |
Luminous Phenomena (Phosphenes)
Phosphenes are phenomena described as a transiently enhanced brightness in a limited area of the visual field, halos, image decomposition (stroboscopic or kaleidoscopic effects), colored bright lights, or multiple images (retinal persistency). Phosphenes are usually triggered by sudden variations in light intensity. Corlanor can cause phosphenes, thought to be mediated through Corlanor’s effects on retinal photoreceptors [see Clinical Pharmacology (12.1)]. Onset is generally within the first 2 months of treatment, after which they may occur repeatedly. Phosphenes were generally reported to be of mild to moderate intensity and led to treatment discontinuation in < 1% of patients; most resolved during or after treatment.
Pediatric Patients with Heart Failure
The safety of Corlanor in pediatric patients 6 months to less than 18 years of age is based on a clinical trial [see Clinical Studies (14.2)] in symptomatic heart failure patients with dilated cardiomyopathy and elevated heart rate. This trial provides experience in 73 patients treated with Corlanor for a median duration of 397 days, and 42 patients given placebo. Bradycardia (symptomatic and asymptomatic) occurred at rates similar to those in adults. Phosphenes were observed in pediatric patients treated with Corlanor.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or establish a causal relationship to drug exposure.
The following adverse reactions have been identified in adults during post-approval use of Corlanor: syncope, hypotension, torsade de pointes, ventricular fibrillation, ventricular tachycardia, angioedema, erythema, rash, pruritus, urticaria, vertigo, and diplopia, and visual impairment.
Related/similar drugs
7. Drug Interactions
7.1 Cytochrome P450-Based Interactions
Corlanor is primarily metabolized by CYP3A4. Concomitant use of CYP3A4 inhibitors increases ivabradine plasma concentrations and use of CYP3A4 inducers decreases them. Increased plasma concentrations may exacerbate bradycardia and conduction disturbances.
The concomitant use of strong CYP3A4 inhibitors is contraindicated [see Contraindications (4) and Clinical Pharmacology (12.3)]. Examples of strong CYP3A4 inhibitors include azole antifungals (e.g., itraconazole), macrolide antibiotics (e.g., clarithromycin, telithromycin), HIV protease inhibitors (e.g., nelfinavir), and nefazodone.
Avoid concomitant use of moderate CYP3A4 inhibitors when using Corlanor. Examples of moderate CYP3A4 inhibitors include diltiazem, verapamil, and grapefruit juice [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
Avoid concomitant use of CYP3A4 inducers when using Corlanor. Examples of CYP3A4 inducers include St. John’s wort, rifampicin, barbiturates, and phenytoin [see Clinical Pharmacology (12.3)].
7.2 Negative Chronotropes
Most patients receiving Corlanor will also be treated with a beta-blocker. The risk of bradycardia increases with concomitant administration of drugs that slow heart rate (e.g., digoxin, amiodarone, beta-blockers). Monitor heart rate in patients taking Corlanor with other negative chronotropes.
7.3 Pacemakers in Adults
Corlanor dosing is based on heart rate reduction, targeting a heart rate of 50 to 60 beats per minute in adults [see Dosage and Administration (2.1)]. Patients with demand pacemakers set to a rate ≥ 60 beats per minute cannot achieve a target heart rate < 60 beats per minute, and these patients were excluded from clinical trials [see Clinical Studies (14.1)]. The use of Corlanor is not recommended in patients with demand pacemakers set to rates ≥ 60 beats per minute.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Based on findings in animals, Corlanor may cause fetal harm when administered to a pregnant woman. There are no adequate and well-controlled studies of Corlanor in pregnant women to inform any drug-associated risks. In animal reproduction studies, oral administration of ivabradine to pregnant rats during organogenesis at a dosage providing 1 to 3 times the human exposure (AUC0-24hr) at the MRHD resulted in embryo-fetal toxicity and teratogenicity manifested as abnormal shape of the heart, interventricular septal defect, and complex anomalies of primary arteries. Increased post-natal mortality was associated with these teratogenic effects in rats. In pregnant rabbits, increased post-implantation loss was noted at an exposure (AUC0-24hr) 5 times the human exposure at the MRHD. Lower doses were not tested in rabbits. The background risk of major birth defects for the indicated population is unknown. The estimated background risk of major birth defects in the U.S. general population is 2 to 4%, however, and the estimated risk of miscarriage is 15 to 20% in clinically recognized pregnancies. Advise a pregnant woman of the potential risk to the fetus.
Clinical Considerations
Disease-associated Maternal and/or Embryo-fetal Risk
Stroke volume and heart rate increase during pregnancy, increasing cardiac output, especially during the first trimester. Pregnant patients with left ventricular ejection fraction less than 35% on maximally tolerated doses of beta-blockers may be particularly heart rate dependent for augmenting cardiac output. Therefore, pregnant patients who are started on Corlanor, especially during the first trimester, should be followed closely for destabilization of their congestive heart failure that could result from heart rate slowing.
Monitor pregnant women with chronic heart failure in 3rd trimester of pregnancy for preterm birth.
Data
Animal Data
In pregnant rats, oral administration of ivabradine during the period of organogenesis (gestation day 6-15) at doses of 2.3, 4.6, 9.3, or 19 mg/kg/day resulted in fetal toxicity and teratogenic effects. Increased intrauterine and post-natal mortality and cardiac malformations were observed at doses ≥ 2.3 mg/kg/day (equivalent to the human exposure at the MRHD based on AUC0-24hr). Teratogenic effects including interventricular septal defect and complex anomalies of major arteries were observed at doses ≥ 4.6 mg/kg/day (approximately 3 times the human exposure at the MRHD based on AUC0-24hr).
In pregnant rabbits, oral administration of ivabradine during the period of organogenesis (gestation day 6-18) at doses of 7, 14, or 28 mg/kg/day resulted in fetal toxicity and teratogenicity. Treatment with all doses ≥ 7 mg/kg/day (equivalent to the human exposure at the MRHD based on AUC0-24hr) caused an increase in post-implantation loss. At the high dose of 28 mg/kg/day (approximately 15 times the human exposure at the MRHD based on AUC0-24hr), reduced fetal and placental weights were observed, and evidence of teratogenicity (ectrodactylia observed in 2 of 148 fetuses from 2 of 18 litters) was demonstrated.
In the pre- and post-natal study, pregnant rats received oral administration of ivabradine at doses of 2.5, 7, or 20 mg/kg/day from gestation day 6 to lactation day 20. Increased post-natal mortality associated with cardiac teratogenic findings was observed in the F1 pups delivered by dams treated at the high dose (approximately 15 times the human exposure at the MRHD based on AUC0-24hr).
8.2 Lactation
Risk Summary
There is no information regarding the presence of ivabradine in human milk, the effects of ivabradine on the breastfed infant, or the effects of the drug on milk production. Animal studies have shown, however, that ivabradine is present in rat milk [see Data]. Because of the potential risk to breastfed infants from exposure to Corlanor, breastfeeding is not recommended.
Data
Lactating rats received daily oral doses of [14C]-ivabradine (7 mg/kg) on post-parturition days 10 to 14; milk and maternal plasma were collected at 0.5 and 2.5 hours post-dose on day 14. The ratios of total radioactivity associated with [14C]-ivabradine or its metabolites in milk vs. plasma were 1.5 and 1.8, respectively, indicating that ivabradine is transferred to milk after oral administration.
8.3 Females and Males of Reproductive Potential
Contraception
Females
Corlanor may cause fetal harm, based on animal data. Advise females of reproductive potential to use effective contraception during Corlanor treatment [see Use in Specific Populations (8.1)].
8.4 Pediatric Use
The safety and effectiveness of Corlanor have been established in pediatric patients (age 6 months to less than 18 years old) and are supported by pharmacokinetic and pharmacodynamic trials and evidence from adequate and well-controlled trials of Corlanor in adult patients. The pediatric study included 116 patients in the following age groups: 17 patients in the 6 months to less than 12 months age group, 36 patients in the 1 year to less than 3 years age group, and 63 patients in the 3 years to less than 18 years age group [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3) and Clinical Studies (14.2)].
The safety and efficacy of Corlanor have not been established in patients less than 6 months of age.
Animal Data
Ivabradine given orally to juvenile rats from post-natal day (PND) 7 until PND 77 at 7.5, 15 and 30 mg/kg/day did not affect post-natal (pre-weaning) development and reproductive performance (post-weaning development). Similar to adult animals, ivabradine decreased heart rate dose-dependently, and increased heart weights at the highest dose administered. Ivabradine also decreased white blood cell counts (lymphocytes) at the highest dose administered. The decrease in white blood cell counts partially reversed within a 3-week recovery period. Exposures in male and female rats at the No Observed Adverse Effect Level (NOAEL) of 7.5 mg/kg/day, was approximately 3 and 8 times, respectively, steady state exposure associated with the highest received maintenance doses across age groups in pediatric patients (based on AUCs).
8.5 Geriatric Use
No pharmacokinetic differences have been observed in elderly (≥ 65 years) or very elderly (≥ 75 years) patients compared to the overall population. However, Corlanor has only been studied in a limited number of patients ≥ 75 years of age.
8.6 Hepatic Impairment
No dose adjustment is required in patients with mild or moderate hepatic impairment. Corlanor is contraindicated in patients with severe hepatic impairment (Child-Pugh C) as it has not been studied in this population and an increase in systemic exposure is anticipated [see Contraindications (4) and Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No dosage adjustment is required for patients with creatinine clearance 15 to 60 mL/min. No data are available for patients with creatinine clearance below 15 mL/min [see Clinical Pharmacology (12.3)].
10. Overdosage
Overdose may lead to severe and prolonged bradycardia. In the event of bradycardia with poor hemodynamic tolerance, temporary cardiac pacing may be required. Supportive treatment, including intravenous (IV) fluids, atropine, and intravenous beta-stimulating agents such as isoproterenol, may be considered.
11. Corlanor Description
Corlanor (ivabradine) tablets and oral solution contains ivabradine as the active pharmaceutical ingredient. Ivabradine is a hyperpolarization-activated cyclic nucleotide-gated channel blocker that reduces the spontaneous pacemaker activity of the cardiac sinus node by selectively inhibiting the If current, resulting in heart rate reduction with no effect on ventricular repolarization and no effects on myocardial contractility.
The chemical name for ivabradine hydrochloride is 3-(3-{[((7S)-3,4-Dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl)methyl] methyl amino} propyl)-1,3,4,5-tetrahydro-7,8-dimethoxy-2H-3-benzazepin-2-one, hydrochloride. The molecular formula is C27H36N2O5∙HCl, and the molecular weight (free base + HCl) is 505.1 (468.6 + 36.5). The chemical structure of ivabradine is shown in Figure 1.
Figure 1. Chemical Structure of Ivabradine
Tablets
Corlanor tablets are supplied in 5 mg and 7.5 mg tablets for oral administration. The tablets contain 5 mg and 7.5 mg of ivabradine, as the active ingredient, equivalent to 5.39 mg and 8.09 mg of ivabradine hydrochloride, respectively. The tablets contain the following inactive ingredients: colloidal silicon dioxide, glycerol, hypromellose, lactose monohydrate, magnesium stearate, maize starch, maltodextrin, polyethylene glycol 6000, red iron oxide, titanium dioxide, and yellow iron oxide.
Oral Solution
Corlanor 5 mg/5 mL (1 mg/mL) oral solution is formulated as a sterile, preservative-free, colorless solution for oral administration. Each 5 mL ampule contains 5 mg of ivabradine, equivalent to 5.39 mg ivabradine hydrochloride, as the active ingredient. The solution contains the following inactive ingredients: maltitol and water.
12. Corlanor - Clinical Pharmacology
12.1 Mechanism of Action
Corlanor blocks the hyperpolarization-activated cyclic nucleotide-gated (HCN) channel responsible for the cardiac pacemaker If current, which regulates heart rate. In clinical electrophysiology studies, the cardiac effects were most pronounced in the sinoatrial (SA) node, but prolongation of the AH interval has occurred as has PR interval prolongation. There was no effect on ventricular repolarization and no effects on myocardial contractility [see Clinical Pharmacology (12.2)].
Corlanor can also inhibit the retinal current Ih. Ih is involved in curtailing retinal responses to bright light stimuli. Under triggering circumstances (e.g., rapid changes in luminosity), partial inhibition of Ih by Corlanor may underlie the luminous phenomena experienced by patients. Luminous phenomena (phosphenes) are described as a transient enhanced brightness in a limited area of the visual field [see Adverse Reactions (6.1)].
12.2 Pharmacodynamics
Corlanor causes a dose-dependent reduction in heart rate. The size of the effect is dependent on the baseline heart rate (i.e., greater heart rate reduction occurs in patients with higher baseline heart rate). At recommended doses, heart rate reduction is approximately 10 bpm at rest and during exercise. Analysis of heart rate reduction vs. dose indicates a plateau effect at doses > 20 mg twice daily. In a study of patients with preexisting conduction system disease (first- or second-degree AV block or left or right bundle branch block) requiring electrophysiologic study, IV ivabradine (0.20 mg/kg) administration slowed the overall heart rate by approximately 15 bpm, increased the PR interval (29 msec), and increased the AH interval (27 msec).
Corlanor does not have negative inotropic effects. Ivabradine increases the uncorrected QT interval with heart rate slowing but does not cause rate-corrected prolongation of QT.
12.3 Pharmacokinetics
The peak concentration (Cmax) and area under the plasma concentration time curve (AUC) are similar for ivabradine and S 18982 between oral solution and tablets for the same dose.
Absorption and Bioavailability
Following oral administration, peak plasma ivabradine concentrations are reached in approximately 1 hour under fasting conditions. The absolute oral bioavailability of ivabradine is approximately 40% because of first-pass elimination in the gut and liver.
Food delays absorption by approximately 1 hour and increases plasma exposure by 20% to 40%. Corlanor should be taken with food [see Dosage and Administration (2)].
Ivabradine is approximately 70% plasma protein bound, and the volume of distribution at steady state is approximately 100 L.
Metabolism and Excretion
The pharmacokinetics of ivabradine are linear over an oral dose range of 0.5 mg to 24 mg. Ivabradine is extensively metabolized in the liver and intestines by CYP3A4-mediated oxidation. The major metabolite is the N-desmethylated derivative (S 18982), which is equipotent to ivabradine and circulates at concentrations approximately 40% that of ivabradine. The N-desmethylated derivative is also metabolized by CYP3A4. Ivabradine plasma levels decline with a distribution half-life of 2 hours and an effective half-life of approximately 6 hours.
The total clearance of ivabradine is 24 L/h, and renal clearance is approximately 4.2 L/h, with ~ 4% of an oral dose excreted unchanged in urine. The excretion of metabolites occurs to a similar extent via feces and urine.
Drug Interactions
The effects of coadministered drugs (CYP3A4 inhibitors, substrates, inducers, and other concomitantly administered drugs) on the pharmacokinetics of Corlanor were studied in several single- and multiple-dose studies. Pharmacokinetic measures indicating the magnitude of these interactions are presented in Figure 2.
Figure 2. Impact of Coadministered Drugs on the Pharmacokinetics of Corlanor
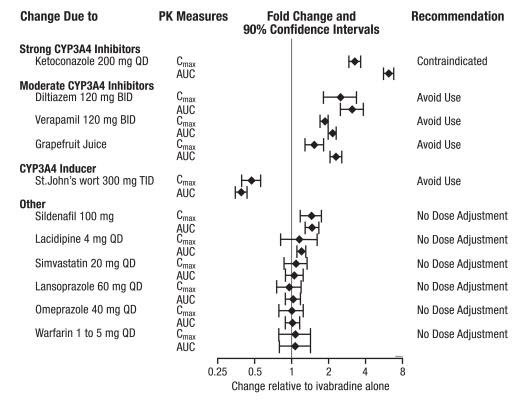
Digoxin exposure did not change when concomitantly administered with ivabradine. No dose adjustment is required when ivabradine is concomitantly administered with digoxin.
Effect of Ivabradine on Metformin Pharmacokinetics
Ivabradine, dosed at 10 mg twice daily to steady state, did not affect the pharmacokinetics of metformin (an organic cation transporter [OCT2] sensitive substrate). The geometric mean (90% confidence interval [CI]) ratios of Cmax and AUCinf of metformin, with and without ivabradine were 0.98 [0.83–1.15] and 1.02 [0.86–1.22], respectively. No dose adjustment is required for metformin when administered with Corlanor.
Specific Populations
Age
No pharmacokinetic differences (AUC or Cmax) have been observed between elderly (≥ 65 years) or very elderly (≥ 75 years) patients and the overall patient population [see Use in Specific Populations (8.5)].
Hepatic Impairment
In patients with mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment, the pharmacokinetics of Corlanor were similar to that in patients with normal hepatic function. No data are available in patients with severe hepatic impairment (Child-Pugh C) [see Contraindications (4)].
Renal Impairment
Renal impairment (creatinine clearance from 15 to 60 mL/min) has minimal effect on the pharmacokinetics of Corlanor. No data are available for patients with creatinine clearance below 15 mL/min.
Pediatrics
The pharmacokinetics of ivabradine and the major metabolite S 18982 were evaluated in 70 pediatric patients with dilated cardiomyopathy and symptomatic chronic heart failure aged from 6 months to less than 18 years of age given ivabradine twice daily [see Clinical Studies (14.2)]. Ivabradine and S 18982 metabolite exposures were measured following weight-based dosing and dose titration designed to reach a dose that enables a 20% heart rate reduction without inducing bradycardia and/or signs or symptoms related to bradycardia. Analyses showed the relationship between exposure and heart rate reduction was similar across pediatric age groups and adults. Steady state exposures of ivabradine and S 18982 following maintenance doses in pediatric patients are similar to exposures achieved in adult heart failure patients given 5 mg BID.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There was no evidence of carcinogenicity when mice and rats received ivabradine up to 104 weeks by dietary administration. High doses in these studies were associated with mean ivabradine exposures of at least 37 times higher than the human exposure (AUC0-24hr) at the MRHD.
Ivabradine tested negative in the following assays: bacterial reverse mutation (Ames) assay, in vivo bone marrow micronucleus assay in both mouse and rat, in vivo chromosomal aberration assay in rats, and in vivo unscheduled DNA synthesis assay in rats. Results of the in vitro chromosomal aberration assay were equivocal at concentrations approximately 1,500 times the human Cmax at the MRHD. Ivabradine tested positive in the mouse lymphoma assays and in vitro unscheduled DNA synthesis assay in rat hepatocytes at concentrations greater than 1,500 times the human Cmax at the MRHD.
Reproduction toxicity studies in animals demonstrated that ivabradine did not affect fertility in male or female rats at exposures 46 to 133 times the human exposure (AUC0-24hr) at the MRHD.
13.2 Animal Toxicology and/or Pharmacology
Reversible changes in retinal function were observed in dogs administered oral ivabradine at total doses of 2, 7, or 24 mg/kg/day (approximately 0.6 to 50 times the human exposure at the MRHD based on AUC0-24hr) for 52 weeks. Retinal function assessed by electroretinography demonstrated reductions in cone system responses, which reversed within a week post-dosing, and were not associated with damage to ocular structures as evaluated by light microscopy. These data are consistent with the pharmacological effect of ivabradine related to its interaction with hyperpolarization-activated Ih currents in the retina, which share homology with the cardiac pacemaker If current.
14. Clinical Studies
14.1 Heart Failure in Adult Patients
SHIFT
The Systolic Heart Failure Treatment with the If Inhibitor Ivabradine Trial (SHIFT) was a randomized, double-blind trial comparing Corlanor and placebo in 6,558 adult patients with stable New York Heart Association (NYHA) class II to IV heart failure, left ventricular ejection fraction ≤ 35%, and resting heart rate ≥ 70 bpm. Patients had to have been clinically stable for at least 4 weeks on an optimized and stable clinical regimen, which included maximally tolerated doses of beta-blockers and, in most cases, ACE inhibitors or ARBs, spironolactone, and diuretics, with fluid retention and symptoms of congestion minimized. Patients had to have been hospitalized for heart failure within 12 months prior to study entry.
The underlying cause of CHF was coronary artery disease in 68% of patients. At baseline, approximately 49% of randomized patients were NYHA class II, 50% were NYHA class III, and 2% were NYHA class IV. The mean left ventricular ejection fraction was 29%. All patients were initiated on Corlanor 5 mg (or matching placebo) twice daily and the dose was increased to 7.5 mg twice daily or decreased to 2.5 mg twice daily to maintain the resting heart rate between 50 and 60 bpm, as tolerated. The primary endpoint was a composite of the first occurrence of either hospitalization for worsening heart failure or cardiovascular death.
Most patients (89%) were taking beta-blockers, with 26% on guideline-defined target daily doses. The main reasons for not receiving the target beta-blocker doses at baseline were hypotension (45% of patients not at target), fatigue (32%), dyspnea (14%), dizziness (12%), history of cardiac decompensation (9%), and bradycardia (6%). For the 11% of patients not receiving any beta-blocker at baseline, the main reasons were chronic obstructive pulmonary disease, hypotension, and asthma. Most patients were also taking ACE inhibitors and/or angiotensin II antagonists (91%), diuretics (83%), and anti-aldosterone agents (60%). Few patients had an implantable cardioverter-defibrillator (ICD) (3.2%) or a cardiac resynchronization therapy (CRT) device (1.1%). Median follow-up was 22.9 months. At 1 month, 63%, 26%, and 8% of Corlanor-treated patients were taking 7.5, 5, and 2.5 mg BID, whereas 3% had withdrawn from the drug, primarily for bradycardia.
SHIFT demonstrated that Corlanor reduced the risk of the combined endpoint of hospitalization for worsening heart failure or cardiovascular death based on a time-to-event analysis (hazard ratio: 0.82, 95% confidence interval [CI]: 0.75, 0.90, p < 0.0001) (Table 3). The treatment effect reflected only a reduction in the risk of hospitalization for worsening heart failure; there was no favorable effect on the mortality component of the primary endpoint. In the overall treatment population, Corlanor had no statistically significant benefit on cardiovascular death.
| Corlanor
(N = 3,241) | Placebo
(N = 3,264) | ||||||||
| Endpoint | n | % | % PY | n | % | % PY | Hazard
Ratio | [95% CI] | p-value |
| Primary composite endpoint: Time to first hospitalization for worsening heart failure or cardiovascular deatha | 793 | 24.5 | 14.5 | 937 | 28.7 | 17.7 | 0.82 | [0.75, 0.90] | < 0.0001 |
| Hospitalization for worsening heart failure | 505 | 15.6 | 9.2 | 660 | 20.2 | 12.5 | |||
| Cardiovascular death as first event | 288 | 8.9 | 4.8 | 277 | 8.5 | 4.7 | |||
| Patients with events at any time | |||||||||
| Hospitalization for worsening heart failureb | 514 | 15.9 | 9.4 | 672 | 20.6 | 12.7 | 0.74 | [0.66, 0.83] | |
| Cardiovascular deathb | 449 | 13.9 | 7.5 | 491 | 15.0 | 8.3 | 0.91 | [0.80, 1.03] | |
| a Patients who died on the same calendar day as their first hospitalization for worsening heart failure are counted under cardiovascular death. b Analyses of the components of the primary composite endpoint were not prospectively planned to be adjusted for multiplicity. N: number of patients at risk; n: number of patients having experienced the endpoint; %: incidence rate = (n/N) × 100; % PY: annual incidence rate = (n/number of patient-years) × 100; CI: confidence interval The hazard ratio between treatment groups (ivabradine/placebo) was estimated based on an adjusted Cox proportional hazards model with beta-blocker intake at randomization (yes/no) as a covariate; p-value: Wald test |
|||||||||
The Kaplan-Meier curve (Figure 3) shows time to first occurrence of the primary composite endpoint of hospitalization for worsening heart failure or cardiovascular death in the overall study.
Figure 3. SHIFT: Time to First Event of Primary Composite Endpoint
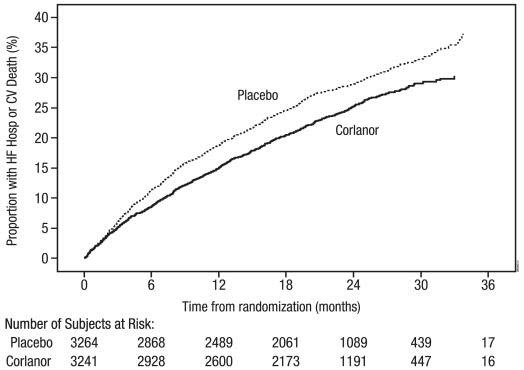
A wide range of demographic characteristics, baseline disease characteristics, and baseline concomitant medications were examined for their influence on outcomes. Many of these results are shown in Figure 4. Such analyses must be interpreted cautiously, as differences can reflect the play of chance among a large number of analyses.
Most of the results show effects consistent with the overall study result. Corlanor’s benefit on the primary endpoint in SHIFT appeared to decrease as the dose of beta-blockers increased, with little if any benefit demonstrated in patients taking guideline-defined target doses of beta-blockers.
Figure 4. Effect of Treatment on Primary Composite Endpoint in Subgroups
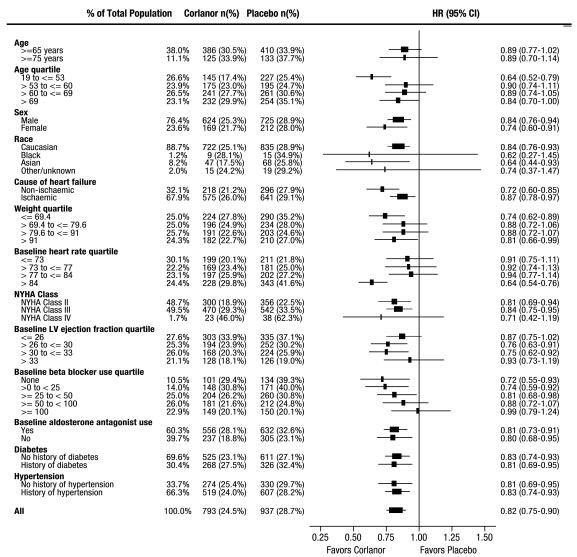
Note: The figure above presents effects in various subgroups, all of which are baseline characteristics. The 95% confidence limits that are shown do not take into account the number of comparisons made and may not reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
BEAUTIFUL and SIGNIFY: No benefit in stable coronary artery disease with or without stable heart failure
The Morbidity-mortality Evaluation of the If Inhibitor Ivabradine in Patients with Coronary Disease and Left Ventricular Dysfunction Trial (BEAUTIFUL) was a randomized, double-blind, placebo-controlled trial in 10,917 adult patients with coronary artery disease, impaired left ventricular systolic function (ejection fraction < 40%) and resting heart rate ≥ 60 bpm. Patients had stable symptoms of heart failure and/or angina for at least 3 months and were receiving conventional cardiovascular medications at stable doses for at least 1 month. Beta-blocker therapy was not required, nor was there a protocol mandate to achieve any specific dosing targets for patients who were taking beta-blockers. Patients were randomized 1:1 to Corlanor or placebo at an initial dose of 5 mg twice daily with the dose increased to 7.5 mg twice daily depending on resting heart rate and tolerability. The primary endpoint was the composite of time to first cardiovascular death, hospitalization for acute myocardial infarction, or hospitalization for new-onset or worsening heart failure. Most patients were NYHA class II (61.4%) or class III (23.2%) - none were class IV. Through a median follow-up of 19 months, Corlanor did not significantly affect the primary composite endpoint (HR 1.00, 95% CI = 0.91, 1.10).
The Study Assessing the Morbi-mortality Benefits of the If Inhibitor Ivabradine in Patients with Coronary Artery Disease Trial (SIGNIFY) was a randomized, double-blind trial administering Corlanor or placebo to 19,102 adult patients with stable coronary artery disease but without clinically evident heart failure (NYHA class I). Beta-blocker therapy was not required. Corlanor was initiated at a dose of 7.5 mg twice daily and the dose could be increased to as high as 10 mg twice daily or down-titrated to 5.0 mg twice daily to achieve a target heart rate of 55 to 60 bpm. The primary endpoint was a composite of the first occurrence of either cardiovascular death or myocardial infarction. Through a median follow-up of 24.1 months, Corlanor did not significantly affect the primary composite endpoint (HR 1.08, 95% CI = 0.96, 1.20).
14.2 Heart Failure in Pediatric Patients
Because Corlanor was effective in improving outcomes in patients with dilated cardiomyopathy (DCM) in SHIFT, the effect on heart rate was considered a reasonable basis to infer clinical benefits in pediatric patients with DCM. Thus, Corlanor was evaluated for its effect on heart rate in a multi-center, randomized, double-blind, placebo-controlled trial in children with symptomatic DCM. The study collected data from 116 patients aged 6 months to less than 18 years with DCM in sinus rhythm, NYHA/Ross class II to IV heart failure, and left ventricular ejection fraction ≤ 45%. Patients had to be clinically stable for at least 4 weeks and on optimized medical therapy with a resting heart rate (HR) complying with the following criteria:
- HR ≥ 105 bpm in the age-subset 6–12 months.
- HR ≥ 95 bpm in the age-subset 1–3 years.
- HR ≥ 75 bpm in the age-subset 3–5 years.
- HR ≥ 70 bpm in the age-subset 5–18 years.
Patients were randomized in a 2:1 ratio to receive Corlanor or placebo. Doses of study medication were titrated over a 2- to 8-week period to achieve a 20% heart rate reduction without inducing bradycardia. The target heart rate reduction was obtained at the end of the titration period in a significantly higher proportion of patients with Corlanor vs. placebo (72% vs. 16% respectively; Odds Ratio = 15; 95% CI = [5; 47]). A statistically significant reduction in HR was observed with Corlanor compared to placebo at the end of the titration period (-23 ± 11 bpm vs. -2 ± 12 bpm respectively).
16. How is Corlanor supplied
Tablets:
Corlanor (ivabradine) 5 mg tablets are formulated as salmon-colored, oval-shaped, film-coated tablets functionally scored on both edges, marked with “5” on one face and bisected on the other face. They are supplied as follows:
- Bottles of 60 tablets with child-resistant closure (NDC 55513-800-60)
Corlanor (ivabradine) 7.5 mg tablets are formulated as salmon-colored, triangular-shaped, film-coated tablets debossed with “7.5” on one face and plain on the other face. They are supplied as follows:
- Bottles of 60 tablets with child-resistant closure (NDC 55513-810-60)
Oral Solution:
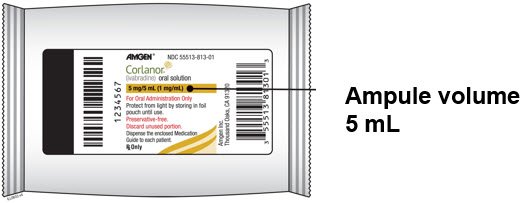
Corlanor (ivabradine) oral solution is a colorless liquid supplied in an opaque, low density polyethylene (LDPE) plastic ampule. Each 5 mL ampule is individually packaged in a child-resistant foil pouch and supplied in cartons containing 28 foil pouches. Corlanor oral solution is supplied as 5 mg/5 mL (1 mg/mL) (NDC 55513-813-01).
Storage
Store Corlanor tablets and oral solution at 25°C (77°F); excursions permitted to 15°C - 30°C (59°F - 86°F) [see USP Controlled Room Temperature]. Protect Corlanor ampule from light by storing in the foil pouch until use.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling [see Medication Guide and Instructions for Use].
-
Fetal Toxicity
Advise pregnant women of the potential risks to a fetus.
Advise females of reproductive potential to use effective contraception and to notify their healthcare provider with a known or suspected pregnancy [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1), (8.3)].
-
Low Heart Rate
Advise patients to report significant decreases in heart rate or symptoms such as dizziness, fatigue, or hypotension [see Warnings and Precautions (5.3)].
-
Atrial Fibrillation
Advise patients to report symptoms of atrial fibrillation, such as heart palpitations or racing, chest pressure, or worsened shortness of breath [see Warnings and Precautions (5.2)].
-
Phosphenes
Advise patients about the possible occurrence of luminous phenomena (phosphenes). Advise patients to use caution if they are driving or using machines in situations where sudden changes in light intensity may occur, especially when driving at night. Advise patients that phosphenes may subside spontaneously during continued treatment with Corlanor [see Adverse Reactions (6.1)].
-
Drug Interactions
Advise patients to avoid ingestion of grapefruit juice and St. John’s wort [see Drug Interactions (7.1)].
-
Intake with Food
Advise patients to take Corlanor twice daily with food [see Dosage and Administration (2)].
-
Oral Solution
Advise parents/caregivers on preparation and administration instructions including the use of a calibrated oral syringe and a medicine cup (provided by the pharmacy) to avoid dosing errors [see Dosage and Administration (2.2)].
Advise parents/caregivers that the oral solution should not be administered by the child.
Advise parents/caregivers to not double up doses (e.g., if patient spits out the drug or caregiver forgets to give the drug at the prescribed time).
Advise parents/caregivers to throw away the unused product remaining in the cup immediately after drawing up the prescribed dose in the syringe.
Corlanor® (ivabradine)
Manufactured by:
Amgen Inc.
One Amgen Center Drive
Thousand Oaks, California 91320-1799
Patent: http://pat.amgen.com/Corlanor/
© 2015, 2017, 2019, 2021 Amgen Inc. All rights reserved.
[part number] v5
Medication Guide
| MEDICATION GUIDE
Corlanor® (core lan ore) (ivabradine) tablets and oral solution |
| What is the most important information I should know about Corlanor?
Corlanor may cause serious side effects in adults and children, including:
|
| What is Corlanor?
Corlanor is a prescription medicine used:
|
| Who should not take Corlanor?
Do not take Corlanor if you have:
|
| What should I tell my doctor before taking Corlanor?
Before you take Corlanor, tell your doctor about all of your medical conditions, including if you:
|
How should you take Corlanor?
|
What should you avoid while taking Corlanor?
|
| What are the possible side effects of Corlanor?
Corlanor may cause serious side effects. See “What is the most important information I should know about Corlanor?” The most common side effects of Corlanor are:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store Corlanor?
|
| General information about the safe and effective use of Corlanor.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Corlanor for a condition for which it was not prescribed. Do not give Corlanor to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about Corlanor that is written for health professionals. |
| What are the ingredients in Corlanor?
Active ingredient: ivabradine Inactive ingredients: Tablet: colloidal silicon dioxide, glycerol, hypromellose, lactose monohydrate, magnesium stearate, maize starch, maltodextrin, polyethylene glycol 6000, red iron oxide, titanium dioxide, yellow iron oxide Oral Solution: Maltitol and water Manufactured by: Amgen Inc. One Amgen Center Drive Thousand Oaks, California 91320 1799 Patent: http://pat.amgen.com/Corlanor/ © 2015, 2017, 2019, 2021 Amgen Inc. All rights reserved. [part number] v5 For more information, go to www.Corlanor.com or call 1-800-772-6436. |
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: August 2021
| Instructions for Use
Corlanor® (core lan ore) (ivabradine) oral solution Ampule |
| Important |
| Before you use Corlanor oral solution, read this important information.
Using
Storing
Measuring
|
|
For more information or help, go to www.Corlanor.com or call 1-800-722-6436. |
| Supplies needed to take or give a dose of Corlanor | |
| From the pharmacist | |
 | 1 reusable medicine cup Measure the dose with the oral syringe. Do not measure the dose with the medicine cup. Do not throw away the medicine cup. |
 | 1 reusable oral syringe Oral syringes come in different sizes (Example: 0.5 mL, 1 mL, 3 mL, 5 mL, 10 mL). Your pharmacist will give you the correct syringe depending on the dose. If you do not receive an oral syringe and medicine cup from your pharmacist, or if you have any questions about your oral syringe, call your healthcare provider or pharmacist. Do not throw away the oral syringe unless you can no longer clearly see the markings. If you are not sure what to do, ask your healthcare provider or pharmacist. |
| The carton | |
 | The carton contains 28 ampules in individual foil pouches. |
 | Each foil pouch contains 1 ampule of Corlanor oral solution. |
| Step 1: Prepare | |
A. Check your prescription.
| |
|
|
|
B. Gather the supplies you will need.
|
|
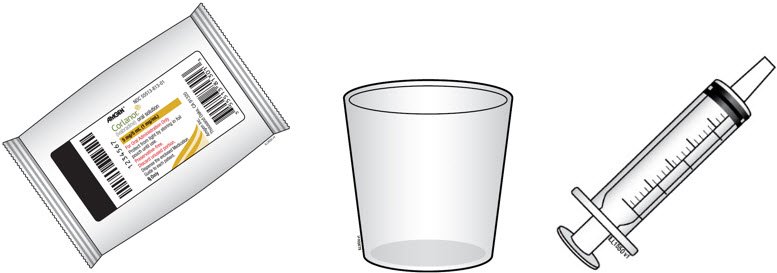 |
|
| C. Cut open the foil pouch and remove the ampule. Be careful not to cut the ampule inside. Repeat with another foil pouch if you need more than 1 ampule for the prescribed dose. |
|
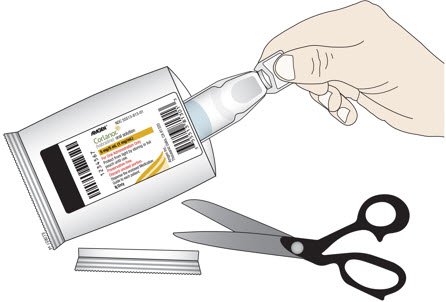 |
|
| Step 2: Empty |
D. To open the ampule, twist the plastic top in either direction. 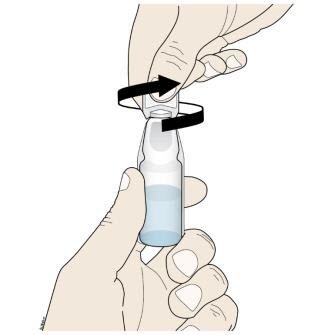
 Important:
|
| Step 3: Check |
| E. Check the dose in milliliters (mL) prescribed by your healthcare provider. Then find that number on the oral syringe. Your syringe and dose may look different from the example shown here. |
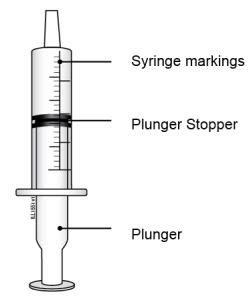 |
| Step 4: Measure |
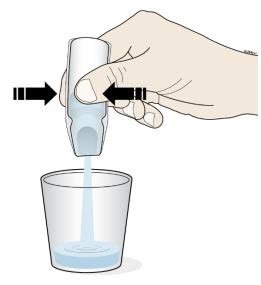
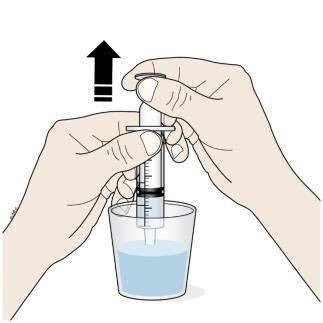
F. Insert the tip of the oral syringe into the oral solution and slowly pull up on the plunger until the plunger stopper lines up with the syringe marking for the prescribed dose.
|
 |
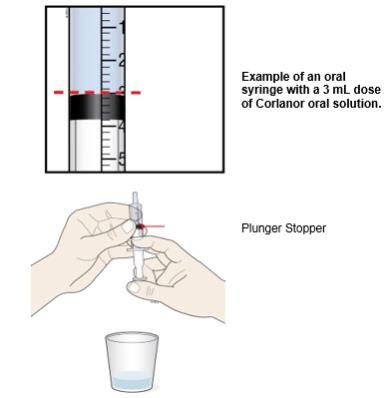
| G. Turn the oral syringe right side up. Hold the syringe over the medicine cup to catch any Corlanor that might drip. H. Adjust the amount of the Corlanor oral solution in the syringe if needed to match your prescribed dose:
|
 |
| Step 5: Give | |
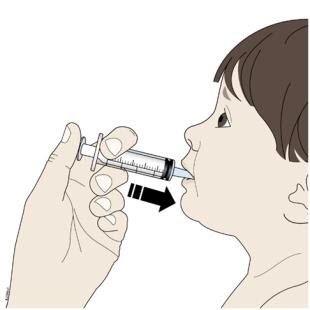
| I. Place the tip of the oral syringe into your or your child’s mouth and towards the cheek. Then slowly push down on the plunger until the oral syringe is empty. |
|
 |
|
| Important: If you miss a dose of Corlanor, do not give another dose. Give the next dose at the usual time. |
|
| Step 6: Finish | |
J. Rinse the reusable oral syringe and reusable medicine cup for the next dose.
| 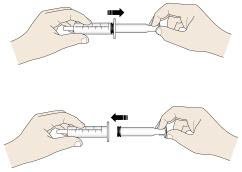 |
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. |
|
Manufactured by:
Amgen Inc.
Thousand Oaks, California 91320-1799
Patent: http://pat/amgen.com/Corlanor/
© 2019, 2021 Amgen Inc. All rights reserved.
[partnumber] v2
Issued: 08/2021
PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label
AMGEN®
NDC 55513-800-60
5
mg
Corlanor®
(ivabradine) tablets
Each tablet contains 5 mg ivabradine equivalent
to 5.39 mg ivabradine as hydrochloride.
Oral Use.
Store at 25°C (77°F); excursions permitted to
15°C to 30°C (between 59°F to 86°F)
[See USP for controlled room temperature].
Keep out of the sight and reach of children.
Dispense the enclosed Medication
Guide to each patient.
Rx Only
60
Film-coated
tablets

PRINCIPAL DISPLAY PANEL - 7.5 mg Tablet Bottle Label
AMGEN®
NDC 55513-810-60
7.5
mg
Corlanor®
(ivabradine) tablets
Each tablet contains 7.5 mg ivabradine equivalent
to 8.085 mg ivabradine as hydrochloride.
Oral Use.
Store at 25°C (77°F); excursions permitted to
15°C to 30°C (between 59°F to 86°F)
[See USP for controlled room temperature].
Keep out of the sight and reach of children.
Dispense the enclosed Medication
Guide to each patient.
Rx Only
60
Film-coated
tablets

PRINCIPAL DISPLAY PANEL - 5 mg/5 mL Ampule Carton
NDC 55513-813-28
Caregiver/Patient: Read the included "Instructions for Use" insert to be sure you prepare and administer the correct dose.
AMGEN®
ATTENTION
PHARMACIST:
Please dispense with
both a medicine cup
and an oral syringe.
Corlanor®
(ivabradine) oral solution
5 mg/5 mL (1 mg/mL)
Sterile Oral Solution in an Ampule
Store at 25°C (77°F); excursions permitted to 15°C to 30°C
(between 59°F to 86°F) [See USP for controlled room temperature].
Protect from light by storing the ampule in foil pouch until use.
Keep out of the sight and reach of children.
Dispense the enclosed Medication Guide to each patient.
Rx Only
Contains 28
ampules
| CORLANOR
ivabradine tablet, film coated |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| CORLANOR
ivabradine tablet, film coated |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| CORLANOR
ivabradine solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Amgen Inc (039976196) |
More about Corlanor (ivabradine)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (6)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- FDA approval history
- Drug class: miscellaneous cardiovascular agents
- Breastfeeding
- En español