Brompheniramine Maleate Drops: Package Insert / Prescribing Info
Package insert / product label
Dosage form: oral liquid
Drug class: Antihistamines
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
Brompheniramine Maleate Drops Description
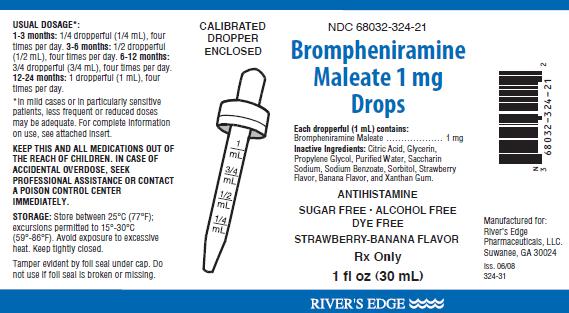
Brompheniramine Maleate 1 mg Drops is an antihistaminic drop for oral administration containing the following amounts of active ingredients in each 1 mL dropperful:Brompheniramine Maleate, USP …….....…………………. 1 mg
Inactive Ingredients: Banana Flavor, Citric Acid, Glycerin, Propylene Glycol, Purified Water, Saccharin Sodium, Sodium Benzoate, Sorbitol, Strawberry Flavor, and Xanthan Gum.
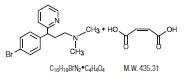
Brompheniramine maleate is an antihistamine having the chemical name 2-pyridinepropanamine, -[4-bromophenyl)-N,N-dimethyl-,(±)-,[Z]-2-butenedioate (1:1) with the following chemical structure:
Brompheniramine Maleate Drops - Clinical Pharmacology
Brompheniramine MaleateBrompheniramine Maleate is a histamine antagonist, specifically an H1-receptor-blocking agent belonging to the alkylamine class of antihistamines. Antihistamines appear to compete with histamine for receptor sites on effector cells. Brompheniramine also has anticholinergic (drying) and sedative effects. Among the antihistaminic effects, it antagonizes the allergic response (vasodilatation, increased vascular permeability, increased mucus secretion) of nasal tissue. Brompheniramine is well absorbed from the gastrointestinal tract, with peak plasma concentration after a single, oral dose of 4 mg reached in 5 hours; urinary excretion is the major route of elimination, mostly as products of biodegradation; the liver is assumed to be the main site of metabolic transformation.
Indications and Usage for Brompheniramine Maleate Drops
Brompheniramine Maleate 1 mg Drops is indicated for symptomatic relief of upper respiratory symptoms, including nasal congestion, associated with allergy or the common cold.Contraindications
Patients with hypersensitivity or idiosyncrasy to Brompheniramine Maleate. Do not use in newborn infants, premature infants, in nursing mothers, in patients with severe hypertension, severe coronary artery disease, ischemic heart disease, or in those receiving monoamine oxidase (MAO) inhibitors. Antihistamines are contraindicated in patients with narrow-angle glaucoma, urinary retention, peptic ulcer, and during an asthma attack. Antihistamines should not be used to treat lower respiratory tract conditions including asthma.Warnings
Antihistamines may diminish mental alertness, and may cause hyperexcitability especially in children. At doses higher than the recommended dose, nervousness, dizziness, or sleeplessness may occur. Especially in infants and small children, antihistamines in overdosage may cause hallucinations, convulsions, and death. Use with caution in patients with hypertension, ischemic heart disease, and persons older than 60 years old. Do not give this product to children who have a chronic pulmonary disease, breathing problems such as chronic bronchitis, glaucoma, or those who are taking sedatives or tranquilizers without first consulting with a doctor. Considerable caution should be exercised in patients with hypertension, diabetes mellitus, ischemic heart disease, hyperthyroidism, increased intraocular pressure and prostatic hypertrophy. Do not exceed the recommended dosage. If nervousness, dizziness, or sleeplessness occurs, discontinue use and consult a doctor.Precautions
General
Because Brompheniramine Maleate 1 mg Drops contain an antihistamine, it should be used with caution in patients with a history of bronchial asthma, narrow angle glaucoma, gastrointestinal obstruction, or urinary bladder neck obstruction. Due to its sympathomimetic component, Brompheniramine Maleate 1 mg Drops should be used with caution in patients with diabetes, hypertension, heart disease, or thyroid disease.
Information for Patients (or Parents)
Patients (or parents) should be warned about engaging in activities requiring mental alertness. In mild cases or in particularly sensitive patients, less frequent doses may be adequate.
Drug Interactions
Do not use this medication in a child who is taking a prescription monoamine oxidase (MAO) inhibitor (certain drugs used for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAO inhibitor. MAO inhibitors prolong and intensify the anticholinergic effects of antihistamines and may enhance the effect of pseudoephedrine HCl. Sympathomimetic agents may reduce the effects of anti-hypertensive drugs. Antihistamines have additive effects with alcohol and other CNS (central nervous system) depressants (hypnotics, sedatives, tranquilizers, anti-anxiety agents, etc.)
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies have not been performed to assess the carcinogenic and mutagenic potential of Brompheniramine Maleate 1 mg Drops or its effect on fertility.
Pregnancy, Teratogenic Effects- Pregnancy Category C
Animal reproductive studies have not been performed with Brompheniramine Maleate 1 mg Drops. It is also not known if it can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Brompheniramine Maleate 1 mg Drops should be given to a pregnant woman only if clearly needed.
Nursing Mothers
Because of the higher risk of intolerance of antihistamines in small infants generally, and in newborns and prematures in particular, Brompheniramine Maleate 1 mg Drops is contraindicated in nursing mothers.
Geriatric Use
The elderly (60 years of age or older) are more likely to experience adverse reactions. Caution should be taken when prescribing this drug to the elderly.
Adverse Reactions/Side Effects
The most frequent adverse reactions to Brompheniramine Maleate 1 mg Drops include: sedation, dryness of mouth, nose and throat; thickening of bronchial secretions; dizziness.
Other adverse reactions may include:
Dermatologic- urticaria, drug rash, photosensitivity, pruritus
Cardiovascular System- hypotension, hypertension, cardiac arrhythmias, palpitations
Central Nervous System (CNS)- disturbed coordination, tremor, irritability, insomnia, visual disturbances, weakness, nervousness, convulsion, headache, euphoria, and dysphoria.
G.U. System- urinary frequency, difficult urination.
G.I. System- epigastric discomfort, anorexia, nausea, vomiting, diarrhea, constipation.
Respiratory System- tightness of chest and wheezing, shortness of breath.
Hematologic System- hemolytic anemia, thrombocytopenia, agranulocytosis.
Related/similar drugs
Overdosage
Signs and SymptomsCentral nervous system effects from overdosage of brompheniramine may vary from depression to stimulation, especially in children. Anticholinergic effects may also occur.
Treatment
Include emesis if patient is alert and is seen prior to 6 hours following ingestion. Precautions against aspiration must be taken, especially in infants and small children. Gastric lavage may be carried out, but in some cases tracheotomy may be needed prior to lavage. For CNS hyperactivity or convulsive seizures, intravenous shortacting barbiturates may be indicated. Hypertensive responses and/or tachycardia should be treated appropriately. Oxygen, intravenous fluids and other supportive measures should be used as indicated.
Brompheniramine Maleate Drops Dosage and Administration
Take by mouth only.
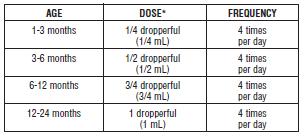
Do not exceed 4 doses during a 24-hour period.
*In mild cases or in particularly sensitive patients, less frequent or reduced doses may be adequate.
How is Brompheniramine Maleate Drops supplied
Brompheniramine Maleate 1 mg Drops is a strawberry-banana flavored, sugar free, alcohol free, dye free liquid packaged in 1 fl oz (30 mL) bottles for dropper dosage, NDC 68032-324-21. Calibrated, shatterproof dropper enclosed in each carton.Storage
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F). Dispense in a tight, light resistant container as defined is the USP/NF- with a child resistant cap. Dispense in the original container. Avoid exposure to excessive heat. Keep tightly closed. Tamper evident by foil seal under cap. Do not use if foil seal is broken or missing.
WARNING: KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN. IN CASE OF ACCIDENTAL OVERDOSE, SEEK PROFESSIONAL ASSISTANCE OR CONTACT A POISON CONTROL CENTER IMMEDIATELY.
Manufactured for:
River's Edge Pharmaceuticals, LLC.
Suwanee, GA 30024
Iss. 06/08 324-11
| BROMPHENIRAMINE MALEATE
brompheniramine maleate liquid |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - River's Edge Pharmaceuticals, LLC (133879135) |
More about brompheniramine
- Check interactions
- Compare alternatives
- Reviews (7)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: antihistamines
- Breastfeeding