Topcare Aspirin: Package Insert / Prescribing Info
Package insert / product label
Generic name: aspirin
Dosage form: tablet, chewable
Drug classes: Platelet aggregation inhibitors, Salicylates
Medically reviewed by Drugs.com. Last updated on Aug 14, 2025.
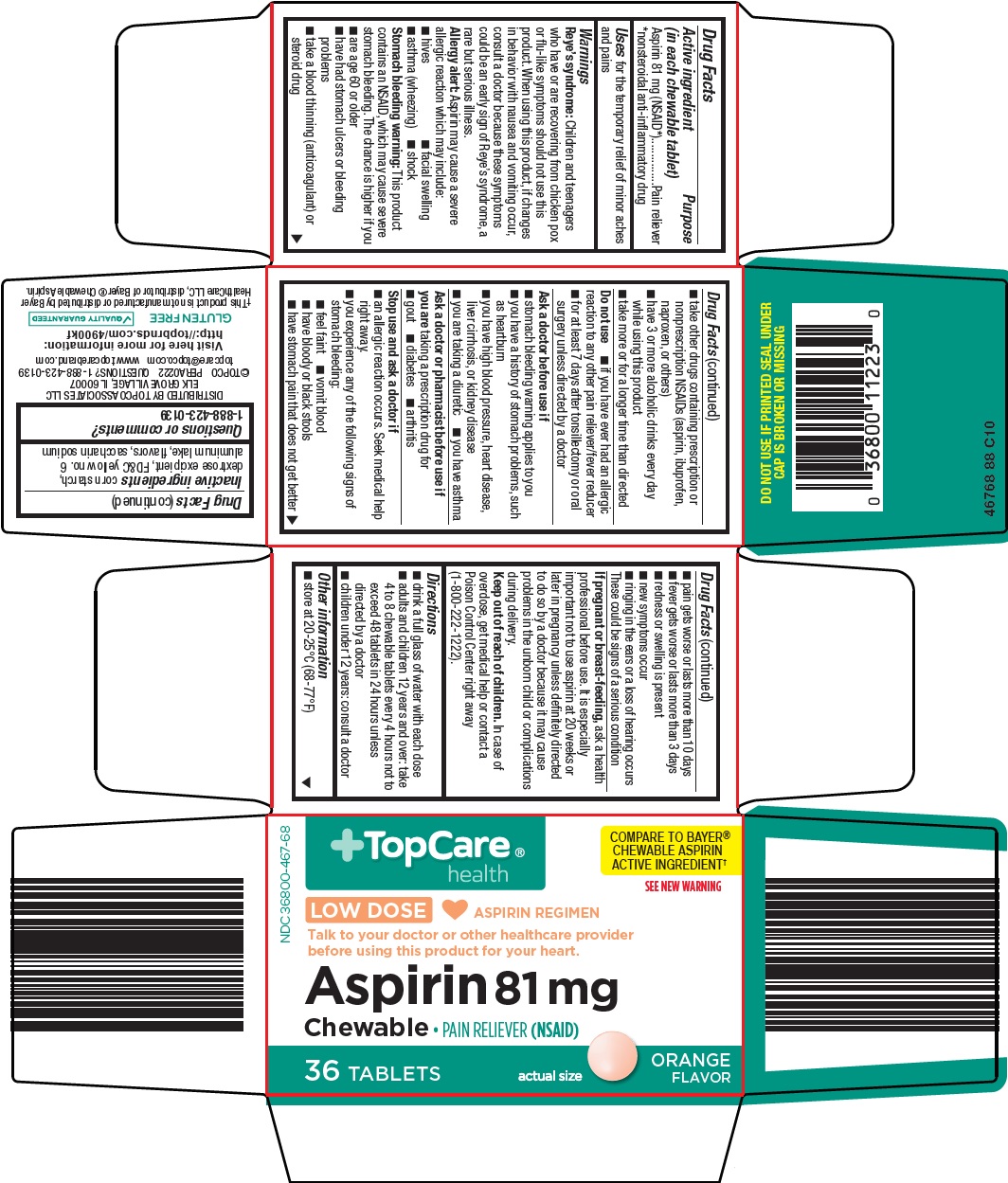
Active ingredient (in each chewable tablet)
Aspirin 81 mg (NSAID*)
*nonsteroidal anti-inflammatory drug
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- •
- hives
- •
- facial swelling
- •
- asthma (wheezing)
- •
- shock
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- •
- are age 60 or older
- •
- have had stomach ulcers or bleeding problems
- •
- take a blood thinning (anticoagulant) or steroid drug
- •
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- •
- have 3 or more alcoholic drinks every day while using this product
- •
- take more or for a longer time than directed
Do not use
- •
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- •
- for at least 7 days after tonsillectomy or oral surgery unless directed by a doctor
Ask a doctor before use if
- •
- stomach bleeding warning applies to you
- •
- you have a history of stomach problems, such as heartburn
- •
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- •
- you are taking a diuretic
- •
- you have asthma
Ask a doctor or pharmacist before use if you are
taking a prescription drug for
- •
- diabetes
- •
- gout
- •
- arthritis
Stop use and ask a doctor if
- •
- an allergic reaction occurs. Seek medical help right away.
- •
- you experience any of the following signs of stomach bleeding:
- •
- feel faint
- •
- vomit blood
- •
- have bloody or black stools
- •
- have stomach pain that does not get better
- •
- pain gets worse or lasts for more than 10 days
- •
- fever gets worse or lasts for more than 3 days
- •
- redness or swelling is present
- •
- new symptoms occur
- •
- ringing in the ears or loss of hearing occurs
These could be signs of a serious condition
Topcare Aspirin Dosage and Administration
- •
- drink a full glass of water with each dose
- •
- adults and children 12 years and over: take 4 to 8 tablets every 4 hours not to exceed 48 tablets in 24 hours unless directed by a doctor
- •
- children under 12 years: consult a doctor
Inactive ingredients
corn starch, dextrose excipient, FD&C yellow no. 6 aluminum lake, flavors, saccharin sodium
Principal Display Panel
TopCare® health
COMPARE TO BAYER® CHEWABLE ASPIRIN ACTIVE INGREDIENT
SEE NEW WARNING
LOW DOSE
ASPIRIN REGIMEN
Talk to your doctor or other healthcare provider before using this product for your heart.
Aspirin 81 mg
Chewable • PAIN RELIEVER (NSAID)
36 TABLETS
actual size
ORANGE FLAVOR
| TOPCARE ASPIRIN
aspirin tablet, chewable |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Topco Associates LLC (006935977) |
Frequently asked questions
- What's the difference between aspirin and ibuprofen?
- Can You Take Tramadol with Acetaminophen, Ibuprofen, or Aspirin?
- What temperature is considered a fever?
- What cold medicine can you take with diabetes?
- Aspirin Overdose: Symptoms, Diagnosis, Emergency Treatment
- Which painkiller should you use?
- Does aspirin lower blood pressure?
- Can I give Aspirin to my dog or cat?
- Can you take ibuprofen with Excedrin Migraine?
More about aspirin
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (52)
- Drug images
- Latest FDA alerts (3)
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: platelet aggregation inhibitors
- Breastfeeding