Kyleena Dosage
Generic name: LEVONORGESTREL 19.5mg
Dosage form: intrauterine device
Drug classes: Contraceptives, Progestogens
Medically reviewed by Drugs.com. Last updated on Feb 19, 2025.
Kyleena contains 19.5 mg of levonorgestrel (LNG) released in vivo at a rate of approximately 17.5 mcg/day after 24 days. This rate decreases progressively to 9.8 mcg/day after 1 year and to 7.4 mcg/day after 5 years. The average in vivo release rate of LNG is approximately 12.6 mcg/day over the first year and 9.0 mcg/day over a period of 5 years.
Kyleena must be removed by the end of the fifth year and can be replaced at the time of removal with a new Kyleena if continued contraceptive protection is desired.
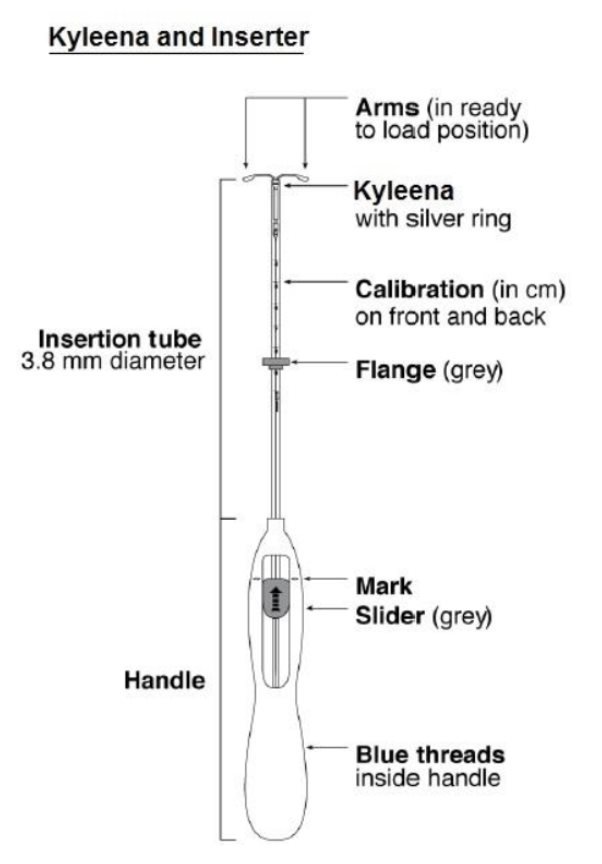
Kyleena can be physically distinguished from other intrauterine systems (IUSs) by the combination of the visibility of the silver ring on ultrasound and the blue color of the removal threads.
Kyleena is supplied in a sterile package within an inserter that enables single-handed loading (see Figure 1). Do not open the package until required for insertion. Do not use if the seal of the sterile package is broken or appears compromised. Use strict aseptic techniques throughout the insertion procedure.
Insertion Instructions
- •
- Obtain a complete medical and social history to determine conditions that might influence the selection of a levonorgestrel-releasing intrauterine system (LNG IUS) for contraception. If indicated, perform a physical examination and appropriate tests for any forms of genital or other sexually transmitted infections.
- •
- Follow the insertion instructions exactly as described to ensure proper placement and avoid premature release of Kyleena from the inserter. Once released, Kyleena cannot be re-loaded.
- •
- Check expiration date of Kyleena prior to initiating insertion.
- •
- Kyleena should be inserted by a trained healthcare provider. Healthcare providers should become thoroughly familiar with the insertion instructions before attempting insertion of Kyleena.
- •
- Insertion may be associated with some pain and/or bleeding or vasovagal reactions (for example, syncope, bradycardia) or with seizure, especially in patients with a predisposition to these conditions. Consider administering analgesics prior to insertion.
Timing of Insertion
|
Starting Kyleena in women not currently using hormonal or intrauterine contraception |
|
|
Switching to Kyleena from an oral, transdermal or vaginal hormonal contraceptive |
|
|
Switching to Kyleena from an injectable progestin contraceptive |
|
|
Switching to Kyleena from a contraceptive implant or another IUS |
|
|
Inserting Kyleena after abortion or miscarriage |
|
|
First-trimester |
|
|
Second-trimester |
|
|
Inserting Kyleena after Childbirth |
|
Tools for Insertion
Preparation for insertion
- •
- Exclude pregnancy and confirm that there are no other contraindications to the use of Kyleena.
- •
- Ensure that the patient understands the contents of the Patient Information Booklet and obtain the signed patient informed consent located on the last page of the Patient Information Booklet.
- •
- With the patient comfortably in lithotomy position, do a bimanual exam to establish the size, shape and position of the uterus.
- •
- Gently insert a speculum to visualize the cervix.
- •
- Thoroughly cleanse the cervix and vagina with a suitable antiseptic solution.
- •
- Prepare to sound the uterine cavity. Grasp the upper lip of the cervix with a tenaculum forceps and gently apply traction to stabilize and align the cervical canal with the uterine cavity. Perform a paracervical block if needed. If the uterus is retroverted, it may be more appropriate to grasp the lower lip of the cervix. The tenaculum should remain in position and gentle traction on the cervix should be maintained throughout the insertion procedure.
- •
- Gently insert a uterine sound to check the patency of the cervix, measure the depth of the uterine cavity in centimeters, confirm cavity direction, and detect the presence of any uterine anomaly. If you encounter difficulty or cervical stenosis, use dilatation, and not force, to overcome resistance. If cervical dilatation is required, consider using a paracervical block.
Insertion Procedure
Proceed with insertion only after completing the above steps and ascertaining that the patient is appropriate for Kyleena. Ensure use of aseptic technique throughout the entire procedure.
Step 1–Opening of the package
- •
- Open the package (Figure 1). The contents of the package are sterile.
Figure 1. Opening the Kyleena Package
- •
- Using sterile gloves, lift the handle of the sterile inserter and remove from the sterile package.
Step 2–Load Kyleena into the insertion tube
- •
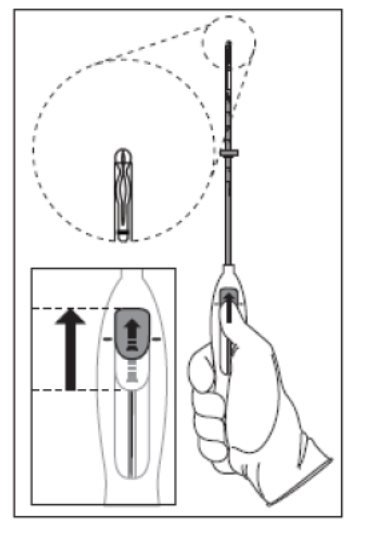
- Push the slider forward as far as possible in the direction of the arrow, thereby moving the insertion tube over the Kyleena T-body to load Kyleena into the insertion tube (Figure 2). The tips of the arms will meet to form a rounded end that extends slightly beyond the insertion tube.
Figure 2. Move slider all the way to the forward position to load Kyleena
- •
- Maintain forward pressure with your thumb or forefinger on the slider. DO NOT move the slider downward at this time as this may prematurely release the threads of Kyleena. Once the slider is moved below the mark, Kyleena cannot be re-loaded.
Step 3–Setting the Flange
- •
- Holding the slider in this forward position, set the upper edge of the flange to correspond to the uterine depth (in centimeters) measured during sounding (Figure 3).
Figure 3. Setting the flange
Step 4–Kyleena is now ready to be inserted
- •
- Continue holding the slider in this forward position. Advance the inserter through the cervix until the flange is approximately 1.5–2 cm from the cervix and then pause (Figure 4).
Figure 4. Advancing insertion tube until flange is 1.5 to 2 cm from the cervix
Do not force the inserter. If necessary, dilate the cervical canal.
Step 5–Open the arms
- •
- While holding the inserter steady, move the slider down to the mark to release the arms of Kyleena (Figure 5). Wait 10 seconds for the horizontal arms to open completely.
Figure 5. Move the slider back to the mark to release and open the arms
Step 6–Advance to fundal position
Advance the inserter gently towards the fundus of the uterus until the flange touches the cervix. If you encounter fundal resistance do not continue to advance. Kyleena is now in the fundal position (Figure 6). Fundal positioning of Kyleena is important to prevent expulsion.
Figure 6. Move Kyleena into the fundal position
Step 7–Release Kyleena and withdraw the inserter
- •
- Holding the entire inserter firmly in place, release Kyleena by moving the slider all the way down (Figure 7).
Figure 7. Move the slider all the way down to release Kyleena from the insertion tube
- •
- Continue to hold the slider all the way down while you slowly and gently withdraw the inserter from the uterus.
- •
- Using a sharp, curved scissor, cut the threads perpendicular, leaving about 3 cm visible outside of the cervix [cutting threads at an angle may leave sharp ends (Figure 8)]. Do not apply tension or pull on the threads when cutting to prevent displacing Kyleena.
Figure 8. Cutting the threads
Kyleena insertion is now complete. Prescribe analgesics, if indicated. Keep a copy of the Consent Form with lot number for your records.
Important information to consider during or after insertion
- •
- If you suspect that Kyleena is not in the correct position, check placement (for example, using transvaginal ultrasound). Remove Kyleena if it is not positioned completely within the uterus. Do not reinsert a removed Kyleena.
- •
- If there is clinical concern, exceptional pain or bleeding during or after insertion, take appropriate steps (such as physical examination and ultrasound) immediately to exclude perforation.
Patient Follow-up
- •
- Reexamine and evaluate patients 4 to 6 weeks after insertion and once a year thereafter, or more frequently if clinically indicated.
Removal of Kyleena
Timing of Removal
- •
- Kyleena should not remain in the uterus after 5 years.
- •
- If pregnancy is not desired, the removal should be carried out during the first 7 days of the menstrual cycle, provided the woman is experiencing regular menses. If removal will occur at other times during the cycle or the woman does not experience regular menses, she is at risk of pregnancy: start a new contraceptive method a week prior to removal for these women.
Removal Procedure
- •
- Remove Kyleena by applying gentle traction on the threads with forceps (Figure 9).
Figure 9. Removal of Kyleena
- •
- If the threads are not visible, determine location of Kyleena by ultrasound.
- •
- If Kyleena is found to be in the uterine cavity on ultrasound exam, it may be removed using a narrow forceps, such as an alligator forceps. This may require dilation of the cervical canal. After removal of Kyleena, examine the system to ensure that it is intact.
- •
- Removal may be associated with some pain and/or bleeding or vasovagal reactions (for example, syncope, bradycardia) or seizure, especially in patients with a predisposition to these conditions.
Continuation of Contraception after Removal
- •
- If pregnancy is not desired and if a woman wishes to continue using Kyleena, a new system can be inserted immediately after removal any time during the cycle.
- •
- If a patient with regular cycles wants to start a different contraceptive method, time removal and initiation of the new method to ensure continuous contraception. Either remove Kyleena during the first 7 days of the menstrual cycle and start the new method immediately thereafter or start the new method at least 7 days prior to removing Kyleena if removal is to occur at other times during the cycle.
- •
- If a patient with irregular cycles or amenorrhea wants to start a different contraceptive method, start the new method at least 7 days before removal.
Frequently asked questions
- What are the side effects of IUDs?
- Can antibiotics affect IUDs like Kyleena, Mirena, or Skyla?
- Can Kyleena cause weight gain?
- Can Kyleena cause acne?
- Can Kyleena make your period stop?
- How long does it take for Kyleena to work?
- How long does Kyleena last?
- Mirena, Kyleena, Skyla & Liletta - What's the difference?
- After having a Kyleena insertion, how long do I have to wait to have intercourse?
More about Kyleena (levonorgestrel)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (1,641)
- Drug images
- Latest FDA alerts (1)
- Side effects
- During pregnancy
- Support group
- FDA approval history
- Drug class: contraceptives
- En español
Patient resources
Other brands
Mirena, Plan B One-Step, Skyla, Liletta, ... +14 more
Professional resources
Other brands
Mirena, Plan B One-Step, Skyla, Liletta, ... +2 more
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.