Rynatan: Package Insert / Prescribing Info
Package insert / product label
Generic name: phenylephrine tannate and chlorpheniramine tannate
Dosage form: tablet
Drug class: Upper respiratory combinations
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
The Rynatan brand name has been discontinued in the U.S. If generic versions of this product have been approved by the FDA, there may be generic equivalents available.
On This Page
Rynatan Description
Rynatan® Tablets are an antihistamine/nasal decongestant combination. Each tablet contains:
Phenylephrine Tannate 25 mg
Chlorpheniramine Tannate 9 mg
Other ingredients: corn starch, dibasic calcium phosphate, magnesium stearate, methylcellulose, polygalacturonic acid, povidone, talc.
Rynatan - Clinical Pharmacology
Rynatan® Tablets combine the sympathomimetic decongestant effect of phenylephrine with the antihistaminic action of chlorpheniramine.
Indications and Usage for Rynatan
Rynatan® Tablets are indicated for symptomatic relief of the coryza and nasal congestion associated with the common cold, sinusitis, allergic rhinitis and other upper respiratory tract conditions. Appropriate therapy should be provided for the primary disease.
Contraindications
Rynatan® Tablets are contraindicated for newborns, nursing mothers and patients sensitive to any of the ingredients or related compounds.
Warnings
Use with caution in patients with hypertension, cardiovascular disease, hyperthyroidism, diabetes, narrow angle glaucoma or prostatic hypertrophy. Use with caution or avoid use in patients taking monoamine oxidase (MAO) inhibitors, or within 14 days of stopping such treatment. This product contains an antihistamine which may cause drowsiness and may have additive central nervous system (CNS) effects with alcohol or other CNS depressants (e.g., hypnotics, sedatives, tranquilizers).
Precautions
General: Antihistamines are more likely to cause dizziness, sedation and hypotension in elderly patients. Antihistamines may cause excitation, particularly in children, but their combination with sympathomimetics may cause either mild stimulation or mild sedation.
Information for patients: Caution patients against drinking alcoholic beverages or engaging in potentially hazardous activities requiring alertness, such as driving a car or operating machinery while using this product. Patients should be warned not to use this product if they are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If patients are uncertain whether a prescription drug contains an MAOI, they should be instructed to consult a health professional before taking such a product.
Drug interactions: MAO inhibitors may prolong and intensify the anticholinergic effects of antihistamines and the overall effects of sympathomimetic agents.
Carcinogenesis, mutagenesis, impairment of fertility: No long term animal studies have been performed with Rynatan® Tablets.
Pregnancy: Teratogenic effects: Pregnancy Category C. Animal reproduction studies have not been conducted with Rynatan® Tablets. It is also not known whether Rynatan® Tablets can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Rynatan® Tablets should be given to a pregnant woman only if clearly needed.
Nursing mothers: Rynatan® Tablets should not be administered to a nursing woman.
Adverse Reactions/Side Effects
To report SUSPECTED ADVERSE REACTIONS, contact Meda Pharmaceuticals Inc. at 1-800-526-3840 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Adverse effects associated with Rynatan® Tablets at recommended doses have been minimal. The most common have been drowsiness, sedation, dryness of mucous membranes, and gastrointestinal effects. Serious side effects with oral antihistamines or sympathomimetics have been rare.
Related/similar drugs
Overdosage
Signs & symptoms: May vary from CNS depression to stimulation (restlessness to convulsions). Antihistamine overdosage in young children may lead to convulsions and death. Atropine-like signs and symptoms may be prominent.
Treatment: Induce vomiting if it has not occurred spontaneously. Precautions must be taken against aspiration especially in infants, children and comatose patients. If gastric lavage is indicated, isotonic or half-isotonic saline solution is preferred. Stimulants should not be used. If hypotension is a problem, vasopressor agents may be considered.
Rynatan Dosage and Administration
Administer the recommended dose every 12 hours.
Rynatan® Tablets: Adults — 1 or 2 tablets.
How is Rynatan supplied
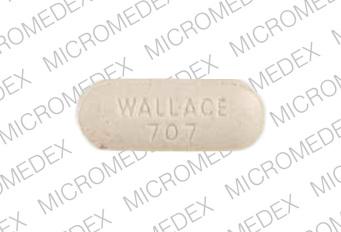
Rynatan® Tablets (phenylephrine tannate 25 mg, chlorpheniramine tannate 9 mg): buff-colored, capsule-shaped, scored on one side and imprinted RYNATAN 707 on the other side. The tablets are available in bottles of 100 (NDC 0037-0707-10).
Storage: Store at controlled room temperature 20°-25°C (68°-77°F). Protect from moisture.
Dispense in a tight container.
To report SUSPECTED ADVERSE REACTIONS, contact Meda Pharmaceuticals Inc. at 1-800-526-3840 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
U.S. Patent 6,037,358
Produced under license from
JFC Technologies
Bound Brook, NJ, U.S.A.
U.S. Patents 5,599,846; 5,663,415
MEDA Pharmaceuticals®
Meda Pharmaceuticals Inc.
Somerset, New Jersey 08873-4120
Printed in U.S.A. Rev. 1/09
Package Label - Principal Display Panel – 100 Tablet Bottle, Rynatan TitratableTablets
NDC 0037-0707-10
RYNATAN®
(phenylephrine tannate,
chlorpheniramine tannate)
TITRATABLE TABLETS
100 Tablets
Each tablet contains:
Phenylephrine Tannate, 25 mg
Chlorpheniramine Tannate, 9 mg
U.S. Patent 6,037,358
Rx only
MEDA
Pharmaceuticals™
LB-070710-06 Rev. 2/08
Usual Dose: See package insert.
Store at controlled room temperature
20°-25°C (68°-77°F).
Protect from moisture.
Dispense in a tight container.
Produced under license from
JFC Technologies
Bound Brook, NJ U.S.A.
U.S. Patents 5,599,846;5,663,415
MEDA
Pharmaceuticals™
Meda Pharmaceuticals Inc.
Somerset, New Jersey 08873-4120

| RYNATAN
phenylephrine tannate and chlorpheniramine tannate tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Meda Pharmaceuticals Inc. (051229602) |
More about Rynatan (chlorpheniramine / phenylephrine)
- Check interactions
- Compare alternatives
- Reviews (5)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: upper respiratory combinations