Diazepam Autoinjector: Package Insert / Prescribing Info
Package insert / product label
Dosage form: injection
Drug classes: Benzodiazepine anticonvulsants, Benzodiazepines
J Code (medical billing code): J3360 (Up to 5 mg, injection)
Medically reviewed by Drugs.com. Last updated on Jan 2, 2024.
On This Page
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs for patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS).
- The use of benzodiazepines, including Diazepam injection, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing Diazepam injection and throughout treatment, assess each patient's risk for abuse, misuse, and addiction (see WARNINGS).
- The continued use of benzodiazepines may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Although Diazepam injection is indicated only for intermittent use (see INDICATIONS AND USAGE and DOSAGE AND ADMINISTRATION), if used more frequently than recommended, abrupt discontinuation or rapid dosage reduction of DIAZEPAM INJECTION may precipitate acute withdrawal reactions, which can be life-threatening. For patients using DIAZEPAM INJECTION more frequently than recommended, to reduce the risk of withdrawal reactions, use a gradual taper to discontinue DIAZEPAM INJECTION (see WARNINGS).
Diazepam Autoinjector Description
Diazepam injection is a sterile solution packaged within a device that delivers its entire 2 mL contents automatically upon activation. Each mL contains 5 mg diazepam compounded with 40% propylene glycol, 10% ethyl alcohol, 5% sodium benzoate and benzoic acid as buffers, and 1.5% benzyl alcohol as preservative.
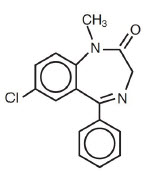
Diazepam is a benzodiazepine derivative. Chemically, diazepam is 7-chloro-1,3- dihydro- 1-methyl-5-phenyl- 2H-1, 4-benzodiazepine-2-one. It is a colorless crystalline compound, insoluble in water and has a molecular weight of 284.74. Its structural formula is as follows:
Diazepam Autoinjector - Clinical Pharmacology
In animals, diazepam appears to act on parts of the limbic system, the thalamus and hypothalamus, and induces calming effects. Diazepam, unlike chlorpromazine and reserpine, has no demonstrable peripheral autonomic blocking action, nor does it produce extrapyramidal side effects. However, animals treated with diazepam do have a transient ataxia at higher doses. Diazepam was found to have transient cardiovascular depressor effects in dogs. Long-term experiments in rats revealed no disturbances of endocrine function. Injections into animals have produced localized irritation of tissue surrounding injection sites and some thickening of veins after intravenous use.
Pharmacokinetics (autoinjector)
A study performed in 24 healthy male subjects comparing the I.M. Injection of 10 mg of diazepam in the mid-anterior/lateral thigh by the autoinjector versus 10 mg I.M. by a syringe (operated manually) indicates that the mean percent availability of the drug from the autoinjector is 100% of that obtained from the syringe.
In addition, the mean Cmax value from the autoinjector was 314 ng/mL (c.v. = 18.7%, range 185 to 439 ng/mL) and 48.6 ng/mL (c.v. = 19.8%, range 29.4 to 69.7 ng/mL) for diazepam and desmethyldiazepam, respectively, while the syringe gave corresponding values of 287 ng/mL (c.v. = 18.9%, range 174 to 378 ng/mL) and 47.2 ng/mL (c.v. =19.4 %, range 33.1 to 61.2 ng/mL) for diazepam and desmethyldiazepam, respectively. The corresponding mean Tmax values were 1.47 hours (c.v. = 69.9%, range 0.8 to 6 hours) and 61.0 hours (c.v. = 56.8%, range 24 to 144 hours) for diazepam and desmethyldiazepam for the autoinjector whereas the syringe gave values of 1.31 hours (c.v. =32%, range 0.7 to 2.0 hours) and 54.5 hours (c.v. = 47.3%, range 12 to 96 hours) for diazepam and desmethyldiazepam, respectively.
Indications and Usage for Diazepam Autoinjector
Diazepam is indicated for the management of anxiety disorders for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic.
- Note: Because the autoinjector provides a minimum dose of 10 mg diazepam, it should not be used to treat individuals with mild and moderate degrees of anxiety and anxiety related disorders that would ordinarily be managed with intramuscular doses of less than 10 mg.
In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis.
As an adjunct prior to endoscopic procedures if apprehension, anxiety or acute stress reactions are present, and to diminish the patient's recall of the procedures (See WARNINGS).
Diazepam is a useful adjunct for the relief of skeletal muscle spasm due to reflex spasm to local pathology (such as inflammation of the muscles or joints, or secondary to trauma), spasticity caused by upper motor neuron disorders (such as cerebral palsy and paraplegia); athetosis; stiff man syndrome; and tetanus.
Diazepam injection is a useful adjunct in status epilepticus and severe recurrent convulsive seizures.
Diazepam is useful premedication for relief of anxiety and tension in patients who are to undergo surgical procedures.
Contraindications
INTRAVENOUS ADMINISTRATION OF DIAZEPAM WITH THE AUTOINJECTOR IS CONTRAINDICATED.
Diazepam injection is contraindicated in patients with a known hypersensitivity to this drug; acute narrow angle glaucoma; and open angle glaucoma unless patients are receiving appropriate therapy.
Warnings
Diazepam Autoinjector is to be administered only by the intramuscular (I.M.) route.
Risks from Concomitant Use With Opioids
Concomitant use of benzodiazepines, including diazepam, and opioids may result in profound sedation, respiratory depression, coma, and death. Because of these risks, reserve concomitant prescribing of benzodiazepines and opioids for patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioids alone. If a decision is made to prescribe diazepam concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. Advise both patients and caregivers about the risks of respiratory depression and sedation when diazepam is used with opioids (see PRECAUTIONS).
Abuse, Misuse, and Addiction
The use of benzodiazepines, including Diazepam injection, exposes users to the risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines often (but not always) involve the use of doses greater than the maximum recommended dosage and commonly involve concomitant use of other medications, alcohol, and/or illicit substances, that is associated with an increased frequency of serious adverse outcomes, including respiratory depression, overdose, or death (see DRUG ABUSE AND DEPENDENCE: Abuse).
Before prescribing Diazepam injection and throughout treatment, assess each patient's risk for abuse, misuse, and addiction. Use of Diazepam injection, particularly in patients at elevated risk, necessitates counseling about the risks and proper use of Diazepam injection along with monitoring for signs and symptoms of abuse, misuse, and addiction. Do not exceed the recommended dosing frequency; avoid or minimize concomitant use of CNS depressants and other substances associated with abuse, misuse, and addiction (e.g., opioid analgesics, stimulants); and advise patients on the proper disposal of unused drug. If a substance use disorder is suspected, evaluate the patient and institute (or refer them for) early treatment, as appropriate.
Dependence and Withdrawal Reactions After Use of Diazepam injection More Frequently Than Recommended
For patients using Diazepam injection more frequently than recommended, to reduce the risk of withdrawal reactions, use a gradual taper to discontinue Diazepam injection (a patient-specific plan should be used to taper the dose).
Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages, and those who have had longer durations of use.
Acute Withdrawal Reactions
The continued use of benzodiazepines may lead to clinically significant physical dependence. Although Diazepam injection is indicated only for intermittent use (see INDICATIONS AND USAGE and DOSAGE AND ADMINISTRATION), if used more frequently than recommended, abrupt discontinuation or rapid dosage reduction of Diazepam injection, or administration of flumazenil (a benzodiazepine antagonist) may precipitate acute withdrawal reactions, which can be life-threatening (e.g., seizures) (see DRUG ABUSE AND DEPENDENCE: Dependence).
Protracted Withdrawal Syndrome
In some cases, benzodiazepine users have developed a protracted withdrawal syndrome with withdrawal symptoms lasting weeks to more than 12 months (see DRUG ABUSE AND DEPENDENCE: Dependence).
Respiratory and Central Nervous System Depression
Extreme care must be used in administering injectable diazepam to the elderly, to very ill patients and to those with limited pulmonary reserve because of the possibility that apnea and/or cardiac arrest may occur. Concomitant use of barbiturates, alcohol, or other central nervous system depressants increases depression with increased risk of apnea. Resuscitative equipment including that necessary to support respiration should be readily available.
When diazepam is used with a narcotic analgesic, the dosage of the narcotic should be reduced by at least one-third and administered in small increments. In some cases the use of a narcotic may not be necessary (see PRECAUTIONS).
Diazepam injection should not be administered to patients in shock, coma, or in acute alcoholic intoxication with depression of vital signs. As is true of most CNS-acting drugs, patients receiving diazepam should be cautioned against engaging in hazardous occupations requiring complete mental alertness, such as operating machinery or driving a motor vehicle.
Use in Patients with Petit Mal Status
Tonic status epilepticus has been precipitated in patients treated with I.V. diazepam for petit mal status or petit mal variant status.
Neonatal Sedation and Withdrawal Syndrome
Use of Diazepam injection late in pregnancy can result in sedation (respiratory depression, lethargy, hypotonia) and/or withdrawal symptoms (hyperreflexia, irritability, restlessness, tremors, inconsolable crying, and feeding difficulties) in the neonate (see PRECAUTIONS: Pregnancy). Monitor neonates exposed to Diazepam injection during pregnancy or labor for signs of sedation and monitor neonates exposed to Diazepam injection during pregnancy for signs of withdrawal; manage these neonates accordingly.
Precautions
Drug Interactions
Effect of Concomitant Use of Benzodiazepines and Opioids
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites, and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and follow patients closely for respiratory depression and sedation.
Concomitant Use with Other Psychotropic Agents or Anticonvulsant Drugs
If diazepam is to be combined with other psychotropic agents or anticonvulsant drugs, careful consideration should be given to the pharmacology of the agents to be employed particularly with known compounds which may potentiate the action of diazepam, such as phenothiazines, narcotics, barbiturates, MAO inhibitors and other antidepressants. In highly anxious patients with evidence of accompanying depression, particularly those who may have suicidal tendencies, protective measures may be necessary. The usual precautions in treating patients with impaired hepatic function should be observed. Metabolites of diazepam are excreted by the kidney; to avoid their excess accumulation, caution should be exercised in the administration to patients with compromised kidney function.
Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to AEDs, such as Diazepam injection, during pregnancy. Healthcare providers are encouraged to recommend that pregnant women taking Diazepam injection enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry by calling 1-888- 233- 2334 or online at http://www.aedpregnancyregistry.org/.
Risk Summary
Neonates born to mothers using benzodiazepines late in pregnancy have been reported to experience symptoms of sedation and/or neonatal withdrawal (see WARNINGS: Neonatal Sedation and Withdrawal Syndrome, and Clinical Considerations). Available data from published observational studies of pregnant women exposed to benzodiazepines do not report a clear association with benzodiazepines and major birth defects (see Human Data).
In animal studies, administration of diazepam during the organogenesis period of pregnancy resulted in increased incidences of fetal malformations at doses greater than those used clinically. Data for diazepam and other benzodiazepines suggest the possibility of increased neuronal cell death and long-term effects on neurobehavioral and immunological function based on findings in animals following prenatal or early postnatal exposure at clinically relevant doses (see Animal Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and of miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Benzodiazepines cross the placenta and may produce respiratory depression, hypotonia, and sedation in neonates. Monitor neonates exposed to Diazepam injection during pregnancy or labor for signs of sedation, respiratory depression, hypotonia, and feeding problems. Monitor neonates exposed to Diazepam injection during pregnancy for signs of withdrawal. Manage these neonates accordingly (see WARNINGS: Neonatal Sedation and Withdrawal Syndrome).
Data
Human Data
Published data from observational studies on the use of benzodiazepines during pregnancy do not report a clear association with benzodiazepines and major birth defects.
Although early studies reported an increased risk of congenital malformations with diazepam and chlordiazepoxide, there was no consistent pattern noted. In addition, the majority of more recent case-control and cohort studies of benzodiazepine use during pregnancy, which were adjusted for confounding exposures to alcohol, tobacco and other medications, have not confirmed these findings.
Animal Data
Diazepam has been shown to produce increased incidences of fetal malformations in mice and hamsters when given orally at single doses of 100 mg/kg or greater (approximately 8 times the maximum recommended human dose [1 mg/kg/day] or greater on a mg/m2 basis). Cleft palate and exencephaly are the most common and consistently reported malformations produced in these species by administration of high, maternally-toxic doses of diazepam during organogenesis.
In published animal studies, administration of benzodiazepines or other drugs that enhance GABAergic neurotransmission to neonatal rats has been reported to result in widespread apoptotic neurodegeneration in the developing brain at plasma concentration relevant for seizure control in humans. The window of vulnerability to these changes in rats (postnatal days 0-14) includes a period of brain development that takes place during the third trimester of pregnancy in humans.
Nursing Mothers
Risk Summary
Diazepam is present in breastmilk. There are reports of sedation, poor feeding and poor weight gain in infants exposed to benzodiazepines through breast milk. There are no data on the effects of diazepam on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Diazepam injection and any potential adverse effects on the breastfed infant from Diazepam injection or from the underlying maternal condition.
Administration Considerations
Long Term Control of Seizures
Although seizures may be brought under control promptly with a single 10 mg intramuscular dose, a significant proportion of patients may experience a return to seizure activity. Consequently, it may become necessary to re-administer the drug. The cumulative maximum dose of diazepam should not exceed 30 mg; the interval between doses should be no less than 10 minutes. Diazepam is not recommended for maintenance, and once seizures are brought under control, consideration should be given to the administration of agents useful in longer term control of seizures.
Since an increase in cough reflex and laryngospasm may occur with peroral endoscopic procedures, the use of a topical anesthetic agent and the availability of necessary countermeasures are recommended.
Adverse Reactions/Side Effects
Side effects most commonly reported were drowsiness, fatigue and ataxia; venous thrombosis and phlebitis at the site of injection. Other adverse reactions less frequently reported include: CNS: confusion, depression, dysarthria, headache, hypoactivity, slurred speech, syncope, tremor, vertigo. G.I.: constipation, nausea. G.U.: incontinence, changes in libido, urinary retention. Cardiovascular: bradycardia, cardiovascular collapse, hypotension. EENT: blurred vision, diplopia, nystagmus. Skin: urticaria, skin rash. Other: hiccups, changes in salivation, neutropenia, jaundice. Paradoxical reactions such as acute hyperexcited states, anxiety, hallucinations, increased muscle spasticity, insomnia, rage, sleep disturbances and stimulation have been reported; should these occur, use of the drug should be discontinued.
Minor changes in EEG patterns, usually low-voltage fast activity, have been observed in patients during and after diazepam therapy and are of no known significance.
In peroral endoscopic procedures, coughing, depressed respiration, dyspnea, hyperventilation, laryngospasm and pain in throat or chest have been reported.
Because of isolated reports of neutropenia and jaundice, periodic blood counts and liver function tests are advisable during long-term therapy.
Related/similar drugs
Drug Abuse and Dependence
Abuse
Diazepam injection is a benzodiazepine and a CNS depressant with a potential for abuse and addiction. Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence. Even taking benzodiazepines as prescribed may put patients at risk for abuse and misuse of their medication. Abuse and misuse of benzodiazepines may lead to addiction.
Abuse and misuse of benzodiazepines often (but not always) involve the use of doses greater than the maximum recommended dosage and commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes, including respiratory depression, overdose, or death. Benzodiazepines are often sought by individuals who abuse drugs and other substances, and by individuals with addictive disorders (see WARNINGS: Abuse, Misuse, and Addiction).
The following adverse reactions have occurred with benzodiazepine abuse and/or misuse: abdominal pain, amnesia, anorexia, anxiety, aggression, ataxia, blurred vision, confusion, depression, disinhibition, disorientation, dizziness, euphoria, impaired concentration and memory, indigestion, irritability, muscle pain, slurred speech, tremors, and vertigo.
The following severe adverse reactions have occurred with benzodiazepine abuse and/or misuse: delirium, paranoia, suicidal ideation and behavior, seizures, coma, breathing difficulty, and death. Death is more often associated with polysubstance use (especially benzodiazepines with other CNS depressants such as opioids and alcohol).
Dependence
Physical Dependence After Use of Diazepam Injection More Frequently Than Recommended
Diazepam injection may produce physical dependence if used more frequently than recommended. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. Although Diazepam injection is indicated only for intermittent use (see INDICATIONS AND USAGE and DOSAGE AND ADMINISTRATION), if used more frequently than recommended, abrupt discontinuation or rapid dosage reduction or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening. Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use (see WARNINGS: Dependence and Withdrawal Reactions).
For patients using Diazepam injection more frequently than recommended, to reduce the risk of withdrawal reactions, use a gradual taper to discontinue Diazepam injection (see WARNINGS: Dependence and Withdrawal Reactions).
Acute Withdrawal Signs and Symptoms
Acute withdrawal signs and symptoms associated with benzodiazepines have included abnormal involuntary movements, anxiety, blurred vision, depersonalization, depression, derealization, dizziness, fatigue, gastrointestinal adverse reactions (e.g., nausea, vomiting, diarrhea, weight loss, decreased appetite), headache, hyperacusis, hypertension, irritability, insomnia, memory impairment, muscle pain and stiffness, panic attacks, photophobia, restlessness, tachycardia, and tremor. More severe acute withdrawal signs and symptoms, including life-threatening reactions, have included catatonia, convulsions, delirium tremens, depression, hallucinations, mania, psychosis, seizures, and suicidality.
Protracted Withdrawal Syndrome
Protracted withdrawal syndrome associated with benzodiazepines is characterized by anxiety, cognitive impairment, depression, insomnia, formication, motor symptoms (e.g., weakness, tremor, muscle twitches), paresthesia, and tinnitus that persists beyond 4 to 6 weeks after initial benzodiazepine withdrawal. Protracted withdrawal symptoms may last weeks to more than 12 months. As a result, there may be difficulty in differentiating withdrawal symptoms from potential re-emergence or continuation of symptoms for which the benzodiazepine was being used.
Tolerance
Tolerance to Diazepam injection may develop after use more frequently than recommended. Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose). Tolerance to the therapeutic effect of benzodiazepines may develop; however, little tolerance develops to the amnestic reactions and other cognitive impairments caused by benzodiazepines.
Overdosage
Overdosage of benzodiazepines is characterized by central nervous system depression ranging from drowsiness to coma. In mild to moderate cases, symptoms can include drowsiness, confusion, dysarthria, lethargy, hypnotic state, diminished reflexes, ataxia, and hypotonia. Rarely, paradoxical or disinhibitory reactions (including agitation, irritability, impulsivity, violent behavior, confusion, restlessness, excitement, and talkativeness) may occur. In severe overdosage cases, patients may develop respiratory depression and coma. Overdosage of benzodiazepines in combination with other CNS depressants (including alcohol and opioids) may be fatal (see Warnings: Abuse, Misuse, and Addiction). Markedly abnormal (lowered or elevated) blood pressure, heart rate, or respiratory rate raise the concern that additional drugs and/or alcohol are involved in the overdosage.
In managing benzodiazepine overdosage, employ general supportive measures, including intravenous fluids and airway maintenance. Flumazenil, a specific benzodiazepine receptor antagonist indicated for the complete or partial reversal of the sedative effects of benzodiazepines in the management of benzodiazepine overdosage, can lead to withdrawal and adverse reactions, including seizures, particularly in the context of mixed overdosage with drugs that increase seizure risk (e.g., tricyclic and tetracyclic antidepressants) and in patients with long-term benzodiazepine use and physical dependency. The risk of withdrawal seizures with flumazenil use may be increased in patients with epilepsy. Flumazenil is contraindicated in patients who have received a benzodiazepine for control of a potentially life-threatening condition (e.g., status epilepticus). If the decision is made to use flumazenil, it should be used as an adjunct to, not as a substitute for, supportive management of benzodiazepine overdosage. See the flumazenil injection Prescribing Information.
Consider contacting the Poison Help line (1-800-221-2222) or a medical toxicologist for additional overdosage management recommendations.
Diazepam Autoinjector Dosage and Administration
Administration of the Diazepam Autoinjector
- 1.
- Pull off gray safety cap
- 2.
- Place Black end on mid outer thigh
- 3.
- Push hard until injector functions
- 4.
- Withdraw after 10 seconds
Dosage
Caution: Because the autoinjector automatically delivers a fixed dose of 10 mg of diazepam, it cannot be used in situations requiring lower total doses or those in which small incremental increases of diazepam are required.
The usual recommended dose in older children and adults ranges from 10 mg to 20 mg I.M. depending on the indication and its severity. The cumulative total dose and individual maximum dose for intramuscular administration will vary with the specific indication (See dosage for specific indications).
Intramuscular
When instructions are followed properly the Diazepam Autoinjector injects deeply into the muscle.
Acute Alcohol Withdrawal
As an aid in symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis.
Endoscopic procedures
Adjunctively, if apprehension, anxiety or acute stress reactions are present prior to endoscopic procedures. Dosage of narcotics should be reduced by at least a third and in some cases may be omitted. See Precautions for peroral procedures.
Muscle Spasm
Associated with local pathology, cerebral palsy, athetosis, stiff-man syndrome or tetanus.
Status Epilepticus and Severe Recurrent Convulsive Seizures
In the convulsing patient, the I.V. route is preferred. However, if conditions preclude intravenous administration, the I.M. route may be used.
Usual Adult Dosage: 10 mg, initially. This injection may be repeated if necessary at 10 to 15 minute intervals up to a maximum dose of 30 mg. If necessary, therapy with diazepam may be repeated in 2 to 4 hours; however, residual active metabolites may persist, and re-administration should be made with this consideration.
Extreme caution must be exercised with individuals with chronic lung disease or unstable cardiovascular status.
Preoperative Medication
To relieve anxiety and tension. (if atropine, scopolamine or other premedications are desired, they must be administered in separate syringes).
General
Once the acute symptomatology has been properly controlled with diazepam injection, the patient may be placed on oral therapy with diazepam if further treatment is required. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever the solution and container permit.
How is Diazepam Autoinjector supplied
Diazepam injection, 5 mg per mL, is available in 2 mL Disposable Autoinjectors.
Office of the Surgeon General
U.S. Army Medical Research and Development Command (MCMR-RCQ-HR) Fort Detrick
Frederick, Maryland 21702-5012
Manufactured by:
Meridian Medical Technologies,
LLC
Revision: 1/2023
0001966
PRINCIPAL DISPLAY PANEL - 10 mg Autoinjector Label
Meridian™
MEDICAL TECHNOLOGIES
St. Louis, MO 63146
AUTOINJECTOR
FOR BUDDY USE
Store at 25°C (77°F); Excursions permitted
to 15 - 30°C (59 - 86°F). [See USP Controlled
Room Temperature]. Do not refrigerate.
AUTOINJECTOR
FOR BUDDY USE
Rx Only
1. Pull off gray safety cap
2. Place black end on mid outer thigh and
3. Push hard until injector functions
4. Withdraw after 10 seconds
Meridian™
MEDICAL TECHNOLOGIES
0002201
NSN 6505-01-274-0951 C-IV
DIAZEPAM INJECTION, USP, 10 mg AUTOMATIC
| DIAZEPAM
diazepam injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Meridian Medical Technologies LLC (049504624) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Meridian Medical Technologies LLC | 049504624 | MANUFACTURE(11704-600) , LABEL(11704-600) , PACK(11704-600) , ANALYSIS(11704-600) , STERILIZE(11704-600) | |
More about diazepam
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (900)
- Drug images
- Latest FDA alerts (2)
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: benzodiazepine anticonvulsants
- Breastfeeding
Patient resources
Professional resources
- Diazepam monograph
- Diazepam (FDA)
- Diazepam Injection (FDA)
- Diazepam Oral Solution Concentrate (FDA)
- Diazepam Rectal Gel (FDA)
Other brands
Valium, Valtoco, Diastat, Libervant