Belsomra: Package Insert / Prescribing Info
Package insert / product label
Generic name: suvorexant
Dosage form: tablet, film coated
Drug class: Miscellaneous anxiolytics, sedatives and hypnotics
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Drug Abuse and Dependence
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
- Medication Guide
Highlights of Prescribing Information
BELSOMRA® (suvorexant) tablets, for oral use, C-IV
Initial U.S. Approval: 2014
Indications and Usage for Belsomra
BELSOMRA is an orexin receptor antagonist indicated for the treatment of insomnia, characterized by difficulties with sleep onset and/or sleep maintenance (1).
Belsomra Dosage and Administration
- Use the lowest dose effective for the patient (2.1).
- Recommended dose is 10 mg, no more than once per night taken within 30 minutes of going to bed, with at least 7 hours remaining before the planned time of awakening. If the 10 mg dose is well-tolerated but not effective, the dose can be increased, not to exceed 20 mg once daily (2.1, 2.2).
- Time to effect may be delayed if taken with or soon after a meal (2.1).
Dosage Forms and Strengths
Tablets, 5 mg, 10 mg, 15 mg, 20 mg (3).
Contraindications
BELSOMRA is contraindicated in patients with narcolepsy (4).
Warnings and Precautions
- CNS Depressant Effects and Daytime Impairment: Risk of impaired alertness and motor coordination, including impaired driving; risk increases with dose; caution patients taking 20 mg against next-day driving and other activities requiring complete mental alertness (5.1).
- Worsening of Depression/Suicidal Ideation: Worsening of depression or suicidal thinking may occur. Risk increases with dose. Immediately evaluate any new behavioral changes (5.2).
- Complex Sleep Behaviors: Behaviors including sleep-walking, sleep-driving, and engaging in other activities while not fully awake may occur. Discontinue immediately if a complex sleep behavior occurs (5.3).
- Sleep Paralysis, Hypnagogic/Hypnopompic Hallucinations, and Cataplexy-like Symptoms: May occur with the use of BELSOMRA. Risk increases with dose (5.4).
- Compromised Respiratory Function: Effect on respiratory function should be considered (5.5, 8.6).
- Need to Evaluate for Co-morbid Diagnoses: Reevaluate if insomnia persists after 7 to 10 days of treatment (5.6).
Adverse Reactions/Side Effects
The most common adverse reaction (reported in 5% or more of patients treated with BELSOMRA and at least twice the placebo rate) with BELSOMRA was somnolence (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
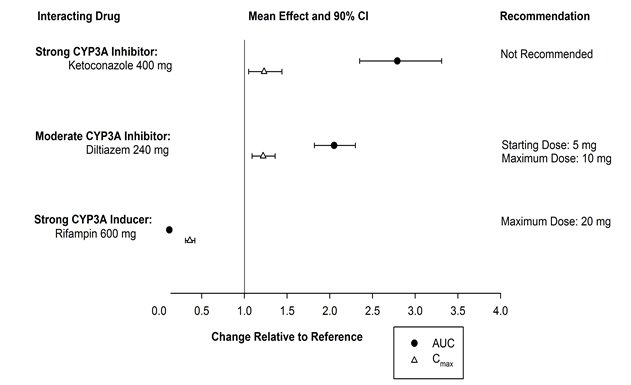
- CYP3A inhibitors: Recommended dose is 5 mg when used with moderate CYP3A inhibitors. Dose can be increased to 10 mg once per night if the 5 mg dose is not effective. Not recommended for use in patients taking strong CYP3A inhibitors (2.4, 7.2).
- Strong CYP3A inducers: Efficacy may be reduced (7.2).
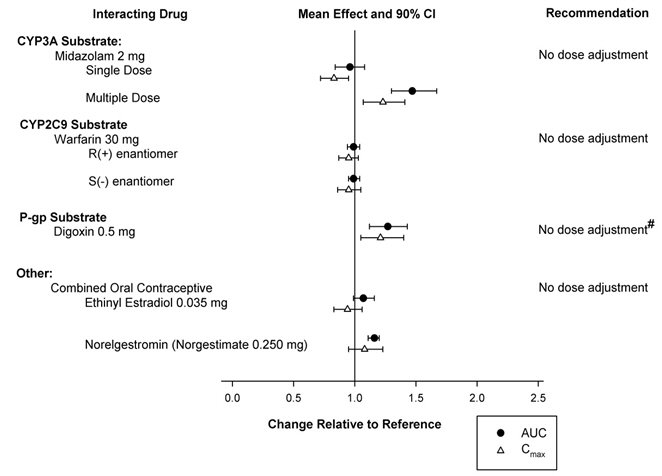
- Digoxin: Monitor digoxin concentrations (7.3).
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 3/2025
Full Prescribing Information
1. Indications and Usage for Belsomra
BELSOMRA® (suvorexant) is indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance.
2. Belsomra Dosage and Administration
2.1 Dosing Information
Use the lowest dose effective for the patient. For all BELSOMRA doses, take no more than once per night within 30 minutes of going to bed (with at least 7 hours remaining prior to planned awakening). Time to effect of BELSOMRA may be delayed if taken with or soon after a meal [see Clinical Pharmacology (12.3)].
The recommended dose for BELSOMRA is 10 mg, taken no more than once per night. If the 10 mg dose is well-tolerated but not effective, the dose can be increased. The maximum recommended dose of BELSOMRA is 20 mg taken no more than once per night.
2.2 Special Populations
Exposure to BELSOMRA is increased in obese compared to non-obese patients, and in women compared to men. Particularly in obese women, the increased risk of exposure-related adverse effects should be considered before increasing the dose [see Clinical Pharmacology (12.3)].
2.3 Use with CNS Depressants
When BELSOMRA is combined with other CNS depressant drugs, dosage reduction of BELSOMRA and/or the other drug(s) may be necessary because of potentially additive effects [see Warnings and Precautions (5.1)].
2.4 Dosage Adjustments with CYP3A Inhibitors
When used with moderate CYP3A inhibitors, the recommended dosage of BELSOMRA is 5 mg taken no more than once per night (the dose generally should not exceed 10 mg). BELSOMRA is not recommended for use with strong CYP3A inhibitors [see Drug Interactions (7.2)].
3. Dosage Forms and Strengths
- 5 mg tablets are yellow, round, film-coated tablets with "5" on one side and plain on the other side.
- 10 mg tablets are green, round, film-coated tablets with "33" on one side and plain on the other side.
- 15 mg tablets are white, oval, film-coated tablets with the corporate logo on one side and "325" on the other side.
- 20 mg tablets are white, round, film-coated tablets with the corporate logo and "335" on one side and plain on the other side.
5. Warnings and Precautions
5.1 CNS Depressant Effects and Daytime Impairment
BELSOMRA is a central nervous system (CNS) depressant that can impair daytime wakefulness even when used as prescribed. Prescribers should monitor for somnolence and CNS depressant effects, but impairment can occur in the absence of symptoms, and may not be reliably detected by ordinary clinical exam (i.e., less than formal testing of daytime wakefulness and/or psychomotor performance). CNS depressant effects may persist in some patients for up to several days after discontinuing BELSOMRA.
BELSOMRA can impair driving skills and may increase the risk of falling asleep while driving. Discontinue or decrease the dose in patients who drive if daytime somnolence develops. In a study of healthy adults, driving ability was impaired in some individuals taking 20 mg BELSOMRA [see Clinical Studies (14.2)]. Although pharmacodynamic tolerance or adaptation to some adverse depressant effects of BELSOMRA may develop with daily use, patients using the 20 mg dose of BELSOMRA should be cautioned against next-day driving and other activities requiring full mental alertness. Patients taking lower doses of BELSOMRA should also be cautioned about the potential for driving impairment because there is individual variation in sensitivity to BELSOMRA.
Co-administration with other CNS depressants (e.g., benzodiazepines, opioids, tricyclic antidepressants, alcohol) increases the risk of CNS depression. Patients should be advised not to consume alcohol in combination with BELSOMRA because of additive effects [see Drug Interactions (7.1)]. Dosage adjustments of BELSOMRA and of concomitant CNS depressants may be necessary when administered together because of potentially additive effects. The use of BELSOMRA with other drugs to treat insomnia is not recommended [see Dosage and Administration (2.3)].
The risk of next-day impairment, including impaired driving, is increased if BELSOMRA is taken with less than a full night of sleep remaining, if a higher than the recommended dose is taken, if co-administered with other CNS depressants, or if co-administered with other drugs that increase blood levels of BELSOMRA. Patients should be cautioned against driving and other activities requiring complete mental alertness if BELSOMRA is taken in these circumstances.
Because BELSOMRA can cause drowsiness, patients, particularly the elderly, are at higher risk of falls.
5.2 Worsening of Depression/Suicidal Ideation
In clinical studies, a dose-dependent increase in suicidal ideation was observed in patients taking BELSOMRA as assessed by questionnaire. Immediately evaluate patients with suicidal ideation or any new behavioral sign or symptom.
In primarily depressed patients treated with sedative-hypnotics, worsening of depression, and suicidal thoughts and actions (including completed suicides) have been reported. Suicidal tendencies may be present in such patients and protective measures may be required. Intentional overdose is more common in this group of patients; therefore, the lowest number of tablets that is feasible should be prescribed for the patient at any one time.
The emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
5.3 Complex Sleep Behaviors
Complex sleep behaviors, including sleep-walking, sleep-driving, and engaging in other activities while not fully awake (e.g., preparing and eating food, making phone calls, having sex), have been reported to occur with the use of hypnotics such as BELSOMRA. These events can occur in hypnotic-naïve as well as in hypnotic-experienced persons. Patients usually do not remember these events. Complex sleep behaviors may occur following the first or any subsequent use of BELSOMRA, with or without the concomitant use of alcohol and other CNS depressants [see Drug Interactions (7.1)]. Discontinue BELSOMRA immediately if a patient experiences a complex sleep behavior.
5.4 Sleep Paralysis, Hypnagogic/Hypnopompic Hallucinations, Cataplexy-Like Symptoms
Sleep paralysis, an inability to move or speak for up to several minutes during sleep-wake transitions, and hypnagogic/hypnopompic hallucinations, including vivid and disturbing perceptions by the patient, can occur with the use of BELSOMRA. Prescribers should explain the nature of these events to patients when prescribing BELSOMRA.
Symptoms similar to mild cataplexy can occur, with risk increasing with the dose of BELSOMRA. Such symptoms can include periods of leg weakness lasting from seconds to a few minutes, can occur both at night and during the day, and may not be associated with an identified triggering event (e.g., laughter or surprise).
5.5 Patients with Compromised Respiratory Function
Effect of BELSOMRA on respiratory function should be considered if prescribed to patients with compromised respiratory function. BELSOMRA has not been studied in patients with severe obstructive sleep apnea (OSA) or severe chronic obstructive pulmonary disease (COPD) [see Use in Specific Populations (8.6)].
5.6 Need to Evaluate for Co-morbid Diagnoses
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, treatment of insomnia should be initiated only after careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. Worsening of insomnia or the emergence of new cognitive or behavioral abnormalities may be the result of an unrecognized underlying psychiatric or physical disorder and can emerge during the course of treatment with hypnotic drugs such as BELSOMRA.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are discussed in greater detail in other sections:
- CNS Depressant Effects and Daytime Impairment [see Warnings and Precautions (5.1)]
- Worsening of Depression/Suicidal Ideation [see Warnings and Precautions (5.2)]
- Complex Sleep Behaviors [see Warnings and Precautions (5.3)]
- Sleep Paralysis, Hypnagogic/Hypnopompic Hallucinations, Cataplexy-Like Symptoms [see Warnings and Precautions (5.4)]
- Patients with Compromised Respiratory Function [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In 3-month controlled efficacy trials (Study 1 and Study 2), 1263 patients were exposed to BELSOMRA including 493 patients who received BELSOMRA 15 mg or 20 mg (see Table 1).
In a long-term study, additional patients (n=521) were treated with BELSOMRA at higher than recommended doses, including a total of 160 patients who received BELSOMRA for at least one year.
| Patients Treated | BELSOMRA 15 mg | BELSOMRA 20 mg |
|---|---|---|
| For ≥ 1 Day (n) | 202 | 291 |
| Men (n) | 69 | 105 |
| Women (n) | 133 | 186 |
| Mean Age (years) | 70 | 45 |
| For ≥ 3 Months (n) | 118 | 172 |
The pooled safety data described below (see Table 2) reflect the adverse reaction profile during the first 3 months of treatment.
Adverse Reactions Resulting in Discontinuation of Treatment
The incidence of discontinuation due to adverse reactions for patients treated with 15 mg or 20 mg of BELSOMRA was 3% compared to 5% for placebo. No individual adverse reaction led to discontinuation at an incidence ≥1%.
Most Common Adverse Reactions
In clinical trials of patients with insomnia treated with BELSOMRA 15 mg or 20 mg, the most common adverse reaction (reported in 5% or more of patients treated with BELSOMRA and at least twice the placebo rate) was somnolence (BELSOMRA 7%; placebo 3%).
Table 2 shows the percentage of patients with adverse reactions during the first three months of treatment, based on the pooled data from 3-month controlled efficacy trials (Study 1 and Study 2).
At doses of 15 or 20 mg, the incidence of somnolence was higher in females (8%) than in males (3%). Of the adverse reactions reported in Table 2, the following occurred in women at an incidence of at least twice that in men: headache, abnormal dreams, dry mouth, cough, and upper respiratory tract infection.
The adverse reaction profile in elderly patients was generally consistent with non-elderly patients. The adverse reactions reported during long-term treatment up to 1 year were generally consistent with those observed during the first 3 months of treatment.
| Placebo | BELSOMRA (20 mg in non-elderly or 15 mg in elderly patients) |
|
|---|---|---|
| n=767 | n=493 | |
| Gastrointestinal Disorders | ||
| Diarrhea | 1 | 2 |
| Dry mouth | 1 | 2 |
| Infections and Infestations | ||
| Upper respiratory tract infection | 1 | 2 |
| Nervous System Disorders | ||
| Headache | 6 | 7 |
| Somnolence | 3 | 7 |
| Dizziness | 2 | 3 |
| Psychiatric Disorders | ||
| Abnormal dreams | 1 | 2 |
| Respiratory, Thoracic and Mediastinal Disorders | ||
| Cough | 1 | 2 |
Dose Relationship for Adverse Reactions
There is evidence of a dose relationship for many of the adverse reactions associated with BELSOMRA use, particularly for certain CNS adverse reactions.
In a placebo-controlled crossover study (Study 3), non-elderly adult patients were treated for up to one month with BELSOMRA at doses of 10 mg, 20 mg, 40 mg (2 times the maximum recommended dose) or 80 mg (4 times the maximum recommended dose). In patients treated with BELSOMRA 10 mg (n=62), the types of adverse reactions observed were similar to those observed in patients treated with BELSOMRA 20 mg. BELSOMRA was associated with a dose-related increase in somnolence: 2% at the 10 mg dose, 5% at the 20 mg dose, 12% at the 40 mg dose, and 11% at the 80 mg dose, compared to <1% for placebo. BELSOMRA was also associated with a dose-related increase in serum cholesterol: 1 mg/dL at the 10 mg dose, 2 mg/dL at the 20 mg dose, 3 mg/dL at the 40 mg dose, and 6 mg/dL at the 80 mg dose after 4 weeks of treatment, compared to a 4 mg/dL decrease for placebo.
Insomnia Study in Patients with Mild to Moderate Alzheimer's Disease
In a 4-week insomnia study of BELSOMRA in 285 patients (BELSOMRA n=142; placebo n=143) with mild to moderate Alzheimer's Disease, the adverse reactions occurring ≥2% and greater than placebo were somnolence (4% compared to 1% for placebo), dry mouth (2% compared to 1% for placebo), and falls (2% compared to 0% for placebo) [see Clinical Studies (14.1)].
6.2 Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of BELSOMRA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac disorders: palpitations, tachycardia
Gastrointestinal disorders: nausea, vomiting
Nervous system disorders: psychomotor hyperactivity
Psychiatric disorders: anxiety
Skin and subcutaneous tissue disorders: pruritus
Related/similar drugs
7. Drug Interactions
7.1 CNS-Active Agents
When BELSOMRA was co-administered with alcohol, additive psychomotor impairment was demonstrated. There was no alteration in the pharmacokinetics of BELSOMRA [see Warnings and Precautions (5.1, 5.3) and Clinical Pharmacology (12.3)].
7.2 Effects of Other Drugs on BELSOMRA
Metabolism by CYP3A is the major elimination pathway for suvorexant.
CYP3A Inhibitors
Concomitant use of BELSOMRA with strong inhibitors of CYP3A (e.g., ketoconazole, itraconazole, posaconazole, clarithromycin, nefazodone, ritonavir, saquinavir, nelfinavir, indinavir, boceprevir, telaprevir, telithromycin and conivaptan) is not recommended [see Clinical Pharmacology (12.3)].
The recommended dose of BELSOMRA is 5 mg in subjects receiving moderate CYP3A inhibitors (e.g., amprenavir, aprepitant, atazanavir, ciprofloxacin, diltiazem, erythromycin, fluconazole, fosamprenavir, grapefruit juice, imatinib, verapamil). The dose generally should not exceed 10 mg in patients receiving moderate CYP3A inhibitors [see Clinical Pharmacology (12.3)].
CYP3A Inducers
Suvorexant exposure can be substantially decreased when co-administered with strong CYP3A inducers (e.g., rifampin, carbamazepine and phenytoin). The efficacy of BELSOMRA may be reduced [see Clinical Pharmacology (12.3)].
7.3 Effects of BELSOMRA on Other Drugs
Digoxin
Concomitant administration of BELSOMRA with digoxin slightly increased digoxin levels due to inhibition of intestinal P-gp. Digoxin concentrations should be monitored when co-administering BELSOMRA with digoxin [see Clinical Pharmacology (12.3)].
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Available data from postmarketing reports with BELSOMRA use in pregnant women are insufficient to establish a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes.
In animal reproduction studies, oral administration of suvorexant to pregnant rats and rabbits during the period of organogenesis decreased maternal body weight and/or weight gain at doses ≥ 30 and 28 times the maximum recommended human dose (MRHD) of 20 mg based on AUC in the rat and rabbit, respectively. Suvorexant caused decreased fetal weight at doses ≥ 86 times the MRHD based on AUC in the rat and did not cause significant fetal toxicity at doses up to 28 times the MRHD based on AUC in the rabbit. The no observed adverse effect levels (NOAELs) for fetal toxicity are 25 and 28 times the MRHD based on AUC in the rat and rabbit, respectively. Oral administration of suvorexant to pregnant rats during pregnancy and lactation caused decreased maternal and pup body weight or weight gain at approximately 48 times the MRHD based on AUC. The NOAEL for development toxicity in the rat is 25 times the MRHD based on AUC (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Suvorexant was administered orally to pregnant rats during the period of organogenesis in two separate studies at doses of 30, 150, and 1000 mg/kg/day or 30, 80, and 325 mg/kg/day, which are approximately 3 to 93 times the MRHD based on AUC. Suvorexant decreased maternal weights at doses ≥ 150 mg/kg/day and fetal weights at doses ≥ 325 mg/kg/day. The NOAEL for both maternal and fetal toxicity is 80 mg/kg/day, which is approximately 25 times the MRHD based on AUC.
Suvorexant was administered orally to pregnant rabbits during the period of organogenesis in two separate studies at doses of 40, 100, and 300 mg/kg/day or 50, 150, and 325 mg/kg/day, which are approximately 3 to 70 times the MRHD based on AUC. Suvorexant decreased maternal body weight or weight gain at doses ≥ 150 mg/kg/day. Suvorexant caused excessive maternal toxicity that led to premature deaths at 325 mg/kg/day, which precluded fetal evaluation. Suvorexant did not cause significant fetal toxicity at doses up to 300 mg/kg/day. The NOAELs for maternal and fetal toxicities are 100 mg/kg/day and 300 mg/kg/day, respectively, which are approximately 10 and 28 times the MRHD based on AUC, respectively.
Suvorexant was administered orally to pregnant rats during pregnancy and lactation at doses of 30, 80, and 200 mg/kg/day, which are approximately 8 to 48 times the MRHD based on AUC. Suvorexant caused maternal toxicity of decreased body weight and weight gain and food consumption at 200 mg/kg/day. At this maternally toxic dose, suvorexant caused decreased weight gain in offspring pups. The NOAEL for maternal and developmental toxicity is 80 mg/kg/day, which is approximately 25 times the MRHD based on AUC.
8.2 Lactation
Risk Summary
Suvorexant and its metabolite, hydroxy suvorexant, are present in low concentrations in breast milk with a relative infant dose of less than 1% following maternal oral administration (see Data). There are no data on the effects of suvorexant on the breastfed infant, or the effects on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for BELSOMRA and any potential adverse effects on the breastfed infant from BELSOMRA or from the underlying maternal condition.
A single dose (20 mg) milk and plasma lactation study was conducted in 12 healthy lactating women. The mean amount of suvorexant recovered in breast milk was 0.011 mg following a 20 mg maternal dose. The calculated mean daily infant dose was 0.002 mg/kg/day based on the actual infant body weight (i.e., 6.2 kg). The relative infant dose is less than 1% of the maternal dose. Approximately 82% of the amount of suvorexant excreted in breast milk was excreted by 24 hours after a single maternal dose administration.
Suvorexant and its metabolite, hydroxy suvorexant, are present in breast milk and the transfer of hydroxy suvorexant into breast milk is lower than suvorexant (metabolite/parent: 0.13).
8.5 Geriatric Use
Of the total number of patients treated with BELSOMRA (n=1784) in controlled clinical safety and efficacy studies, 829 patients were 65 years and over, and 159 patients were 75 years and over. No clinically meaningful differences in safety or effectiveness were observed between these patients and younger patients at the recommended doses [see Clinical Pharmacology (12.3) and Clinical Studies (14)].
Because BELSOMRA can increase drowsiness, patients, particularly the elderly, are at a higher risk of falls [see Warnings and Precautions (5.1)].
8.6 Patients with Compromised Respiratory Function
Effects of BELSOMRA on respiratory function should be considered if prescribed to patients with compromised respiratory function.
Obstructive Sleep Apnea
The respiratory depressant effect of BELSOMRA was evaluated after one night and after four consecutive nights of treatment in a randomized, placebo-controlled, 2-period crossover study in patients (n=26) with mild to moderate obstructive sleep apnea. Following once-daily doses of 40 mg, the mean Apnea/Hypopnea Index treatment difference (suvorexant – placebo) on Day 4 was 2.7 (90% CI: 0.22 to 5.09), but there was wide inter- and intra-individual variability such that clinically meaningful respiratory effects of BELSOMRA in obstructive sleep apnea cannot be excluded. BELSOMRA has not been studied in patients with severe obstructive sleep apnea [see Warnings and Precautions (5.5)].
Chronic Obstructive Pulmonary Disease
The respiratory depressant effect of BELSOMRA was evaluated after one night and after four consecutive nights of treatment in a randomized, placebo-controlled, 2-period crossover study in patients (n=25) with mild to moderate chronic obstructive pulmonary disease (COPD). BELSOMRA (40 mg in non-elderly, 30 mg in elderly) had no respiratory depressant effects in patients with mild to moderate COPD, as measured by oxygen saturation. There was wide inter- and intra-individual variability such that clinically meaningful respiratory effects of BELSOMRA in COPD cannot be excluded. BELSOMRA has not been studied in patients with severe COPD [see Warnings and Precautions (5.5)].
8.7 Patients with Hepatic Impairment
No dose adjustment is required in patients with mild and moderate hepatic impairment. BELSOMRA has not been studied in patients with severe hepatic impairment and is not recommended for these patients [see Clinical Pharmacology (12.3)].
8.8 Patients with Renal Impairment
No dose adjustment is required in patients with renal impairment [see Clinical Pharmacology (12.3)].
9. Drug Abuse and Dependence
9.2 Abuse
Abuse of BELSOMRA poses an increased risk of somnolence, daytime sleepiness, impaired reaction time and impaired driving skills [see Warnings and Precautions (5.1)]. Patients at risk for abuse may include those with prolonged use of BELSOMRA, those with a history of drug abuse, and those who use BELSOMRA in combination with alcohol or other abused drugs.
Drug abuse is the intentional non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than to other activities and obligations), and possible tolerance or physical dependence.
In an abuse liability study conducted in recreational polydrug users (n=36), suvorexant (40, 80 and 150 mg) produced similar effects as zolpidem (15, 30 mg) on subjective ratings of "drug liking" and other measures of subjective drug effects. Because individuals with a history of abuse or addiction to alcohol or other drugs may be at increased risk for abuse and addiction to BELSOMRA, follow such patients carefully.
9.3 Dependence
Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. In completed clinical trials with BELSOMRA, there was no evidence for physical dependence with the prolonged use of BELSOMRA. There were no reported withdrawal symptoms after discontinuation of BELSOMRA.
10. Overdosage
There is limited premarketing clinical experience with an overdosage of BELSOMRA. In clinical pharmacology studies, healthy subjects who were administered morning doses of up to 240 mg of suvorexant showed dose-dependent increases in the frequency and duration of somnolence.
General symptomatic and supportive measures should be used, along with immediate gastric lavage where appropriate. Intravenous fluids should be administered as needed. As in all cases of drug overdose, vital signs should be monitored and general supportive measures employed. The value of dialysis in the treatment of overdosage has not been determined. As suvorexant is highly protein-bound, hemodialysis is not expected to contribute to elimination of suvorexant.
As with the management of all overdosage, the possibility of multiple drug ingestion should be considered. Consider contacting a poison control center for up-to-date information on the management of hypnotic drug product overdosage.
11. Belsomra Description
BELSOMRA tablets contain suvorexant, an orexin receptor antagonist.
Suvorexant is described chemically as:
[(7R)-4-(5-chloro-2-benzoxazolyl) hexahydro-7-methyl-1H-1,4-diazepin-1-yl][5-methyl-2-(2H-1,2,3-triazol-2-yl)phenyl]methanone
Its empirical formula is C23H23ClN6O2 and the molecular weight is 450.92. Its structural formula is:
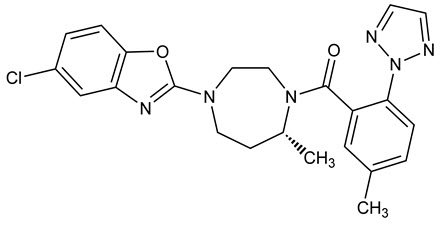
Suvorexant is a white to off-white powder that is insoluble in water.
Each film coated tablet contains 5 mg, 10 mg, 15 mg, or 20 mg of suvorexant and the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and polyvinylpyrrolidone/vinyl acetate copolymer (copovidone).
In addition, the film coating contains the following inactive ingredients: hypromellose, lactose monohydrate, titanium dioxide, and triacetin. The film coating for the 5 mg tablets also contains iron oxide black and iron oxide yellow, and the film coating for the 10 mg tablets also contains FD&C Blue #1/Brilliant Blue FCF Aluminum Lake and iron oxide yellow.
12. Belsomra - Clinical Pharmacology
12.1 Mechanism of Action
The mechanism of action of suvorexant in the treatment of insomnia is presumed to be through antagonism of orexin receptors. The orexin neuropeptide signaling system plays a role in wakefulness. Blocking the binding of wake-promoting neuropeptides orexin A and orexin B to receptors OX1R and OX2R is thought to suppress wake drive.
12.2 Pharmacodynamics
Suvorexant binds to orexin receptors, OX1R and OX2R (Ki= 0.55 and 0.35 nM, respectively).
Antagonism of orexin receptors may also underlie potential adverse effects such as signs of narcolepsy/cataplexy. Genetic mutations in the orexin system in animals result in hereditary narcolepsy; loss of orexin neurons has been reported in humans with narcolepsy.
Evaluation of QTc Interval
The effects of suvorexant on the QTc interval were evaluated in a randomized, placebo-, and active-controlled (moxifloxacin 400 mg) crossover study in healthy subjects (n=53). The upper bound of the one-sided 95% confidence interval for the largest placebo-adjusted, baseline-corrected QTc interval was below 10 ms based on analysis of suvorexant doses up to 240 mg, 12 times the maximum recommended dose. BELSOMRA thus does not prolong the QTc interval to any clinically relevant extent.
12.3 Pharmacokinetics
Suvorexant exposure increases in a less than strictly dose-proportional manner over the range of 10-80 mg because of decreased absorption at higher doses. Suvorexant pharmacokinetics are similar in healthy subjects and patients with insomnia.
Absorption
Suvorexant peak concentrations occur at a median Tmax of 2 hours (range 30 minutes to 6 hours) under fasted conditions. The mean absolute bioavailability of 10 mg is 82%.
Effect of Food
Ingestion of BELSOMRA with a high-fat meal resulted in no meaningful change in AUC or Cmax but a delay in Tmax of approximately 1.5 hours [see Dosage and Administration (2.1)].
Distribution
The mean volume of distribution of suvorexant is approximately 49 liters. Suvorexant is extensively bound (>99%) to human plasma proteins and does not preferentially distribute into red blood cells. Suvorexant binds to both human serum albumin and α1-acid glycoprotein.
Elimination
Metabolism
Suvorexant is mainly eliminated by metabolism, primarily by CYP3A with a minor contribution from CYP2C19. The major circulating entities are suvorexant and a hydroxy-suvorexant metabolite. This metabolite is not expected to be pharmacologically active.
Excretion
The primary route of elimination is through the feces, with approximately 66% of radiolabeled dose recovered in the feces compared to 23% in the urine. The systemic pharmacokinetics of suvorexant are linear with an accumulation of approximately 1- to 2-fold with once-daily dosing. Steady-state is achieved by 3 days. The mean t1/2 is approximately 12 hours (95% CI: 12 to 13).
Specific Populations
Gender, age, body mass index (BMI), and race were included as factors assessed in the population pharmacokinetic model to evaluate suvorexant pharmacokinetics in healthy subjects and to predict exposures in the patient population. Age and race are not predicted to have any clinically meaningful changes on suvorexant pharmacokinetics; therefore, no dose adjustment is warranted based upon these factors.
Suvorexant exposure is higher in females than in males. In females, the AUC and Cmax are increased by 17% and 9%, respectively, following administration of BELSOMRA 40 mg. The average concentration of suvorexant 9 hours after dosing is 5% higher for females across the dose range studied (10-40 mg). Dose adjustment of BELSOMRA is generally not needed based on gender only.
Apparent oral clearance of suvorexant is inversely related to body mass index. In obese patients, the AUC and Cmax are increased by 31% and 17%, respectively. The average concentration of suvorexant approximately 9 hours after a 20 mg dose is 15% higher in obese patients (BMI > 30 kg/m2) relative to those with a normal BMI (BMI ≤ 25 kg/m2).
In obese females, the AUC and Cmax are increased by 46% and 25%, respectively, compared to non-obese females. The higher exposure to suvorexant in obese females should be considered before increasing dose [see Dosage and Administration (2.2)].
The effects of renal and hepatic impairment on the pharmacokinetics of suvorexant were evaluated in specific pharmacokinetic studies.
Suvorexant exposure after a single dose was similar in patients with moderate hepatic insufficiency (Child-Pugh category 7 to 9) and healthy matched control subjects; however, the suvorexant apparent terminal half-life was increased from approximately 15 hours (range 10 - 22 hours) in healthy subjects to approximately 19 hours (range 11 - 49 hours) in patients with moderate hepatic insufficiency [see Use in Specific Populations (8.7)].
Suvorexant exposure (expressed as total and unbound concentrations) was similar between patients with severe renal impairment (urinary creatinine clearance ≤30 mL/min/1.73m2) and healthy matched control subjects. No dose adjustment is required in patients with renal impairment [see Use in Specific Populations (8.8)].
Drug Interaction Studies
CNS-Active Drugs
An additive effect on psychomotor performance was observed when a single dose of 40 mg of suvorexant was co-administered with a single dose of 0.7 g/kg alcohol. Suvorexant did not affect alcohol concentrations and alcohol did not affect suvorexant concentrations [see Warnings and Precautions (5.1, 5.3) and Drug Interactions (7.1)].
An interaction study with a single dose of 40 mg suvorexant and paroxetine 20 mg at steady-state levels in healthy subjects did not demonstrate a clinically significant pharmacokinetic or pharmacodynamic interaction.
Effects of Other Drugs on BELSOMRA
The effects of other drugs on the pharmacokinetics of suvorexant are presented in Figure 1 as change relative to suvorexant administered alone (test/reference). Strong (e.g., ketoconazole or itraconazole) and moderate (e.g., diltiazem) CYP3A inhibitors significantly increased suvorexant exposure. Strong CYP3A inducers (e.g., rifampin) substantially decreased suvorexant exposure [see Drug Interactions (7.2)].
Figure 1: Effects of Co-Administered Drugs on the Pharmacokinetics of Suvorexant
Effects of BELSOMRA on Other Drugs
In vitro metabolism studies demonstrate that suvorexant has the potential to inhibit CYP3A and intestinal P-gp; however, suvorexant is unlikely to cause clinically significant inhibition of human CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19 or CYP2D6. In addition, no clinically meaningful inhibition of OATP1B1, BCRP and OCT2 transporters is anticipated. Chronic administration of suvorexant is unlikely to induce the metabolism of drugs metabolized by major CYP isoforms. Specific in vivo effects on the pharmacokinetics of midazolam, warfarin, digoxin and oral contraceptives are presented in Figure 2 as a change relative to the interacting drug administered alone (test/reference) [see Drug Interactions (7.3)].
| # Monitor digoxin concentrations as clinically indicated [see Drug Interactions (7.3)]. |
|
| Figure 2: Effects of Suvorexant* on the Pharmacokinetics of Co-Administered Drugs |
|
|
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Rats were orally administered suvorexant at doses of 80, 160, and 325 mg/kg/day [males] or 40, 80, and 325 mg/kg/day [females] for 2 years. Suvorexant increased the incidences of thyroid follicular cell adenoma and combined adenoma/carcinoma in females at 325 mg/kg/day, thyroid follicular cell adenoma in males at ≥ 160 mg/kg/day, and hepatocellular adenoma in males at 325 mg/kg/day. These findings were consistent with increased TSH and hepatic enzyme induction, respectively, which are mechanisms believed to be rodent-specific. Suvorexant did not increase the incidence of tumors in rats at 80 mg/kg/day, which is approximately 7 times the MRHD based on AUC.
Suvorexant did not increase the incidence of tumors in Tg.rasH2 mice treated for 26 weeks at oral doses of 25, 50, 200, and 650 mg/kg/day.
Mutagenesis
Suvorexant was not mutagenic in the in vitro bacterial reverse mutation (Ames) assay or clastogenic in the in vitro mammalian chromosomal aberration assay or in the in vivo mouse and rat bone marrow micronucleus assays.
Impairment of Fertility
In two separate studies, suvorexant was orally administered to male and female rats at doses of 80, 160, and 325 mg/kg/day or 100, 300, and 1200 mg/kg/day [males] and 30, 80, and 325 mg/kg/day or 25, 75, and 1200 mg/kg/day [females] prior to and throughout mating and continuing in females to gestation day 7. Increases in pre-implantation loss and resorption and decreases in live fetuses were observed at the highest doses of 325 or 1200 mg/kg/day, when treated males and females were mated with untreated animals. The NOAELs for fertility are 160 and 80 mg/kg/day in males and females, respectively, which are approximately 20 times the MRHD based on AUC.
13.2 Animal Toxicology and/or Pharmacology
Suvorexant administered to dogs at oral doses of 5 and 30 mg/kg/day for 4 to 7 days resulted in behaviors characteristic of cataplexy (e.g., transient limb buckling, prone posture) when presented with food enrichment, a stimulus demonstrated to induce cataplexy in dogs with hereditary narcolepsy.
In the 2-year carcinogenicity study in rats, an increased incidence of retinal atrophy was observed at all doses. Plasma AUCs at the lowest dose tested were approximately 7 times that in humans at the MRHD.
In subsequent studies of suvorexant in albino and pigmented rats, retinal atrophy was delayed in onset and, after approximately one year of dosing, was of lower incidence and severity in pigmented rats.
14. Clinical Studies
14.1 Controlled Clinical Studies
BELSOMRA was evaluated in three clinical trials in patients with insomnia characterized by difficulties with sleep onset and sleep maintenance.
Two similarly designed, 3-month, randomized, double-blind, placebo-controlled, parallel-group studies were conducted (Study 1 and Study 2). In both studies, non-elderly (age 18-64) and elderly (age ≥ 65) patients were randomized separately. For the studies together, non-elderly adults (mean age 46 years; 465 females, 275 males) were treated with BELSOMRA 20 mg (n=291) or placebo (n=449). Elderly patients (mean age 71 years, 346 females, 174 males) were treated with BELSOMRA 15 mg (n=202) or placebo (n=318).
In Study 1 and Study 2, BELSOMRA 15 mg or 20 mg was superior to placebo for sleep latency as assessed both objectively by polysomnography (Table 3) and subjectively by patient-estimated sleep latency (Table 4). BELSOMRA 15 mg or 20 mg was also superior to placebo for sleep maintenance, as assessed both objectively by polysomnography (Table 5) and subjectively by patient-estimated total sleep time (Table 6). The effects of BELSOMRA at night 1 (objective) and week 1 (subjective) were generally consistent with later time points. The efficacy of BELSOMRA was similar between women and men and, based on limited data, between Caucasians and non-Caucasians. Twenty seven percent of patients treated with BELSOMRA 15 mg or 20 mg in Study 1 and Study 2 were non-Caucasians. The majority (69%) of the non-Caucasian patients was Asian.
| Mean Baseline and Change from Baseline†
After 1 and 3 Months (minutes) | Difference† Between BELSOMRA and Placebo (minutes) |
||
|---|---|---|---|
| † Change from baseline and treatment differences based upon estimated means. | |||
| ‡ 15 mg in elderly and 20 mg in non-elderly patients | |||
| * p<0.05; **p<0.01; ***p<0.001 | |||
| Study 1 | |||
| Placebo (n=290) | BELSOMRA 15-20 mg‡
(n=193) | ||
| Baseline | 66 | 69 | |
| Change from Baseline | |||
| Month 1 | - 23 | - 34 | - 10*** |
| Month 3 | - 27 | - 35 | - 8** |
| Study 2 | |||
| Placebo (n=286) | BELSOMRA 15-20 mg‡
(n=145) | ||
| Baseline | 69 | 65 | |
| Change from Baseline | |||
| Month 1 | - 25 | - 33 | - 8* |
| Month 3 | - 29 | - 29 | 0 |
| Mean Baseline and Change from Baseline†
After 1 and 3 Months (minutes) | Difference† Between BELSOMRA and Placebo (minutes) |
||
|---|---|---|---|
| † Change from baseline and treatment differences based upon estimated means. | |||
| ‡ 15 mg in elderly and 20 mg in non-elderly patients | |||
| * p<0.05; **p<0.01; ***p<0.001 | |||
| Study 1 | |||
| Placebo (n=382) | BELSOMRA 15-20 mg‡
(n=251) | ||
| Baseline | 67 | 64 | |
| Change from Baseline | |||
| Month 1 | - 12 | - 17 | - 5 |
| Month 3 | - 17 | - 23 | - 5* |
| Study 2 | |||
| Placebo (n=369) | BELSOMRA 15-20 mg‡
(n=231) | ||
| Baseline | 83 | 86 | |
| Change from Baseline | |||
| Month 1 | - 14 | - 21 | - 7* |
| Month 3 | - 21 | - 28 | - 8* |
| Mean Baseline and Change from Baseline†
After 1 and 3 Months (minutes) | Difference† Between BELSOMRA and Placebo (minutes) |
||
|---|---|---|---|
| † Change from baseline and treatment differences based upon estimated means. | |||
| ‡ 15 mg in elderly and 20 mg in non-elderly patients | |||
| * p<0.05; **p<0.01; ***p<0.001 | |||
| Study 1 | |||
| Placebo (n=290) | BELSOMRA 15-20 mg‡
(n=193) | ||
| Baseline | 115 | 120 | |
| Change from Baseline | |||
| Month 1 | - 19 | - 45 | - 26*** |
| Month 3 | - 25 | - 42 | - 17*** |
| Study 2 | |||
| Placebo (n=286) | BELSOMRA 15-20 mg‡
(n=145) | ||
| Baseline | 118 | 119 | |
| Change from Baseline | |||
| Month 1 | - 23 | - 47 | - 24*** |
| Month 3 | - 25 | - 56 | - 31*** |
| Mean Baseline and Change from Baseline†
After 1 and 3 Months (minutes) | Difference† Between BELSOMRA and Placebo (minutes) |
||
|---|---|---|---|
| † Change from baseline and treatment differences based upon estimated means. | |||
| ‡ 15 mg in elderly and 20 mg in non-elderly patients | |||
| * p<0.05; **p<0.01; ***p<0.001 | |||
| Study 1 | |||
| Placebo (n=382) | BELSOMRA 15-20 mg‡
(n=251) | ||
| Baseline | 315 | 322 | |
| Change from Baseline | |||
| Month 1 | 23 | 39 | 16*** |
| Month 3 | 41 | 51 | 11* |
| Study 2 | |||
| Placebo (n=369) | BELSOMRA 15-20 mg‡
(n=231) | ||
| Baseline | 307 | 299 | |
| Change from Baseline | |||
| Month 1 | 22 | 43 | 21*** |
| Month 3 | 38 | 60 | 22*** |
In the 1-month crossover study (Study 3), non-elderly adults (age 18-64 years, mean age 44 years) were treated with placebo (n=249) and BELSOMRA at a dose of 10 mg (n=62), 20 mg (n=61), or up to 80 mg. BELSOMRA 10 mg and 20 mg were superior to placebo for sleep latency and sleep maintenance, as assessed objectively by polysomnography.
BELSOMRA was also evaluated at doses of 30 mg and 40 mg in the 3-month placebo-controlled trials (Study 1 and Study 2). The higher doses were found to have similar efficacy to lower doses, but significantly more adverse reactions were reported at the higher doses.
Insomnia Study in Patients with Mild to Moderate Alzheimer's Disease
A randomized, double-blind, placebo-controlled, parallel-group, multi-site 4-week trial of BELSOMRA was conducted in patients with mild to moderate Alzheimer's disease (n=285) for the treatment of insomnia. Male and female subjects aged 50-90 years (inclusive) were treated with BELSOMRA (n=142) or placebo (n=143). Patients treated with BELSOMRA received 10 mg for approximately 14 days, of whom 77% were increased to 20 mg for approximately 14 additional days.
In this study, patients treated with BELSOMRA exhibited a statistically significant improvement for both Total Sleep Time (TST) and Wake After Sleep Onset (WASO) measures, compared to those treated with placebo, as assessed by polysomnography at Week 4.
14.2 Special Safety Studies
Effects on Driving
Two randomized, double-blind, placebo- and active-controlled, four-period crossover studies evaluated the effects of nighttime administration of BELSOMRA on next-morning driving performance 9 hours after dosing in 24 healthy elderly subjects (≥65 years old, mean age 69 years; 14 men, 10 women) who received 15 mg and 30 mg BELSOMRA, and 28 non-elderly subjects (mean age 46 years; 13 men, 15 women) who received 20 mg and 40 mg BELSOMRA. Testing was conducted after one night and after 8 consecutive nights of treatment with BELSOMRA at these doses.
The primary outcome measure was change in Standard Deviation of Lane Position (SDLP), a measure of driving performance, assessed using a symmetry analysis. The analysis showed clinically meaningful impaired driving performance in some subjects. After one night of dosing, this effect was observed in non-elderly subjects after either a 20 mg or 40 mg dose of BELSOMRA. A statistically significant effect was not observed in elderly subjects after a 15 mg or 30 mg dose of BELSOMRA. Across these two studies, five subjects (4 non-elderly women on BELSOMRA; 1 elderly woman on placebo) prematurely stopped their driving tests due to somnolence. Patients using the 20 mg dose of BELSOMRA should be cautioned against next-day driving and other activities requiring full mental alertness. Patients taking lower doses of BELSOMRA should also be cautioned about the potential for driving impairment because there is individual variation in sensitivity to BELSOMRA [see Warnings and Precautions (5.1)].
Effects on Next-Day Memory and Balance in Elderly and Non-Elderly
Four placebo-controlled trials evaluated the effects of nighttime administration of BELSOMRA on next-day memory and balance using word learning tests and body sway tests, respectively. Three trials showed no significant effects on memory or balance compared to placebo. In a fourth trial in healthy non-elderly subjects, there was a significant decrease in word recall after the words were presented to subjects in the morning following a single dose of 40 mg BELSOMRA, and there was a significant increase on body sway area in the morning following a single dose of 20 mg or 40 mg BELSOMRA.
Middle of the Night Safety in Elderly Subjects
A double-blind, randomized, placebo-controlled trial evaluated the effect of a single dose of BELSOMRA on balance, memory and psychomotor performance in healthy elderly subjects (n=12) after being awakened during the night. Nighttime dosing of BELSOMRA 30 mg resulted in impairment of balance (measured by body sway area) at 90 minutes as compared to placebo. Memory was not impaired, as assessed by an immediate and delayed word recall test at 4 hours post-dose.
Rebound Effects
In 3-month controlled safety and efficacy trials (Study 1, Study 2), rebound insomnia was assessed following discontinuation of BELSOMRA relative to placebo and baseline in non-elderly adult patients receiving BELSOMRA 40 mg or 20 mg and in elderly patients receiving BELSOMRA 30 mg or 15 mg. No clear effects were observed on measures of sleep onset or maintenance.
Withdrawal Effects
In 3-month controlled safety and efficacy trials (Study 1, Study 2), withdrawal effects were assessed following discontinuation in non-elderly adult patients who received BELSOMRA 40 mg or 20 mg and elderly patients who received BELSOMRA 30 mg or 15 mg. The analysis showed no clear evidence of withdrawal in the overall study population based on assessment of patient responses to the Tyrer Withdrawal Symptom Questionnaire or assessment of withdrawal-related adverse events following the discontinuation of BELSOMRA.
Respiratory Safety
Use in Healthy Subjects with Normal Respiratory Function
A randomized, placebo-controlled, double-blind, crossover trial in healthy non-elderly subjects (n=12) evaluated the respiratory depressant effect of BELSOMRA (40 mg and 150 mg) after one night of treatment. At the doses studied, BELSOMRA had no respiratory depressant effect as measured by oxygen saturation [see Warnings and Precautions (5.5) and Use in Specific Populations (8.6)].
16. How is Belsomra supplied
16.1 How Supplied
BELSOMRA tablets, 5 mg, are yellow, round, film-coated tablets, with "5" on one side and plain on the other side. They are supplied as follows: NDC 0006-0005-30 unit-of-use blisters of 30
BELSOMRA tablets, 10 mg, are green, round, film-coated tablets, with "33" on one side and plain on the other side. They are supplied as follows: NDC 0006-0033-30 unit-of-use blisters of 30
BELSOMRA tablets, 15 mg, are white, oval, film-coated tablets with the corporate logo on one side and "325" on the other side. They are supplied as follows: NDC 0006-0325-30 unit-of-use blisters of 30
BELSOMRA tablets, 20 mg, are white, round, film-coated tablets with the corporate logo and "335" on one side and plain on the other side. They are supplied as follows: NDC 0006-0335-30 unit-of-use blisters of 30
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
CNS Depressant Effects and Next-Day Impairment
Tell patients that BELSOMRA has the potential to cause next-day impairment, and that this risk is increased with higher doses or if dosing instructions are not carefully followed. Patients using the 20 mg dose should be cautioned against next-day driving and other activities requiring full mental alertness as this dose is associated with a higher risk of impaired driving. Patients taking lower doses should also be cautioned about the potential for driving impairment because there is individual variation in sensitivity to BELSOMRA.
Patients should not drive or engage in other activities requiring full alertness within 8 hours of dosing of BELSOMRA. Advise patients that increased drowsiness may increase the risk of falls in some patients [see Warnings and Precautions (5.1)].
Sleep-Driving and Other Complex Behaviors
Instruct patients to inform their families that BELSOMRA has been associated with getting out of bed while not being fully awake, and tell patients and their families to call their healthcare providers if this occurs.
Hypnotics, like BELSOMRA, have been associated with "sleep-driving" and other complex behaviors while not being fully awake (preparing and eating food, making phone calls, or having sex). Tell patients and their families to call their healthcare providers if they develop any of these symptoms.
Alcohol and Other Drugs
Ask patients about alcohol consumption, prescription medicines they are taking, and drugs they may be taking without a prescription. Advise patients not to use BELSOMRA if they drank alcohol that evening or before bed.
Tolerance, Abuse, and Dependence
Tell patients not to increase the dose of BELSOMRA on their own, and to inform you if they believe the drug "does not work."
Administration Instructions
Advise patients to take BELSOMRA only when preparing for or getting into bed and only if they can stay in bed for a full night before being active again. Advise patients to report all of their prescription and nonprescription medicines, vitamins and herbal supplements to the prescriber.
Distributed By: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent
uspi-mk4305-t-2503r011
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 3/2025 |
| MEDICATION GUIDE BELSOMRA® (bell-SOM-rah) suvorexant Tablets C-IV |
|
| What is the most important information I should know about BELSOMRA? BELSOMRA may cause serious side effects including:
See "What are the possible side effects of BELSOMRA?" for more information about side effects. |
|
What is BELSOMRA?
BELSOMRA is a federally controlled substance (C-IV) because it can be abused or cause dependence. Keep BELSOMRA in a safe place to prevent misuse and abuse. Selling or giving away BELSOMRA may harm others and is against the law. Tell your doctor if you have ever abused or have been dependent on alcohol, prescription medicines or street drugs. |
|
| Who should not take BELSOMRA? | |
| Do not take BELSOMRA if you fall asleep often at unexpected times (narcolepsy). | |
| What should I tell my doctor before taking BELSOMRA? | |
Before taking BELSOMRA, tell your doctor about all of your medical conditions, including if you:
|
|
| Tell your doctor about all the medicines you take, including prescription or over-the-counter medicines, vitamins, or herbal supplements. Medicines can interact with each other, sometimes causing serious side effects. Do not take BELSOMRA with other medicines that can make you sleepy unless your doctor tells you to. | |
| Know the medicines you take. Keep a list of your medicines with you to show your doctor and pharmacist each time you get a new medicine. | |
How should I take BELSOMRA?
|
|
What should I avoid while taking BELSOMRA?
|
|
| What are the possible side effects of BELSOMRA? See "What is the most important information I should know about BELSOMRA?" BELSOMRA may cause serious side effects including: |
|
|
|
| The most common side effects of BELSOMRA include drowsiness the next day after you take BELSOMRA. The following additional side effects have been reported with BELSOMRA: abnormal dreams. |
|
|
These are not all the possible side effects of BELSOMRA. For more information, ask your doctor or pharmacist. |
|
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |
How should I store BELSOMRA?
|
|
| General information about the safe and effective use of BELSOMRA. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use BELSOMRA for a condition for which it was not prescribed. Do not give BELSOMRA to other people, even if they have the same symptoms that you have. It may harm them. | |
| This Medication Guide summarizes the most important information about BELSOMRA. You can ask your pharmacist or doctor for information about BELSOMRA that is written for health professionals. | |
| For more information, go to www.BELSOMRA.com or call 1-800-622-4477. | |
| What are the ingredients in BELSOMRA? | |
| Active ingredient: Suvorexant | |
| Inactive ingredients: Croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and polyvinylpyrrolidone/vinyl acetate copolymer (copovidone). The film coating contains: hypromellose, lactose monohydrate, titanium dioxide, and triacetin. The film coating for the 5 mg tablets also contains iron oxide black and iron oxide yellow, and the film coating for the 10 mg tablets also contains FD&C Blue #1/Brilliant Blue FCF Aluminum Lake and iron oxide yellow. | |
| Distributed by: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA |
|
| For patent information: www.msd.com/research/patent | |
| Copyright © 2014-2025 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved. |
|
| usmg-mk4305-t-2503r007 | |
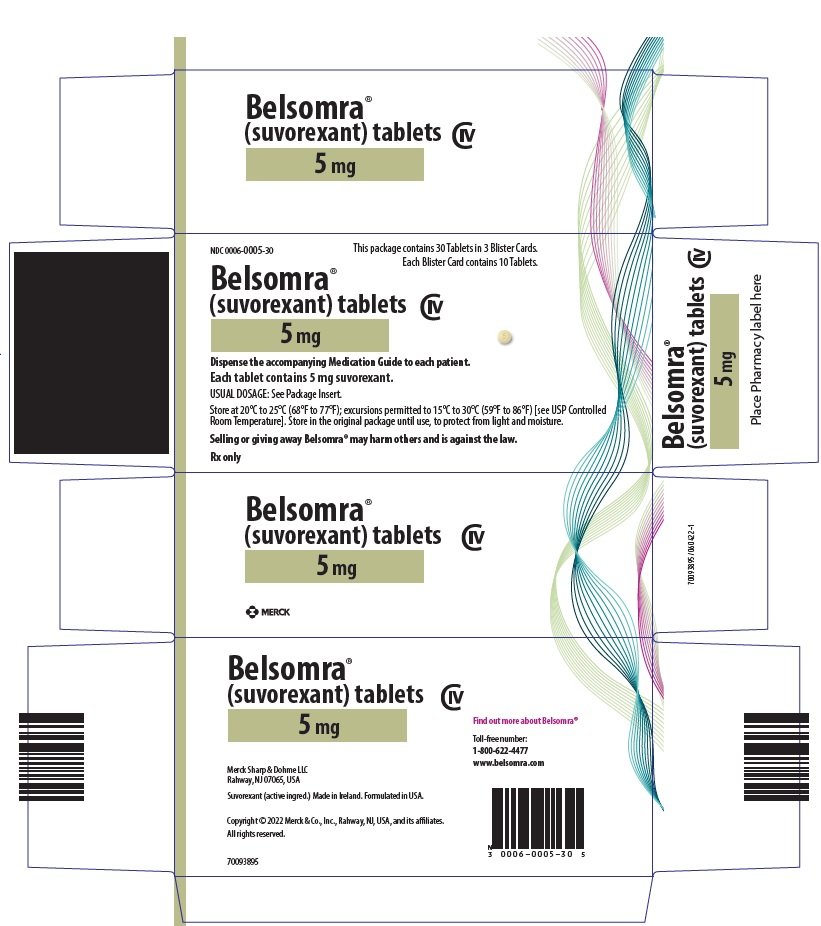
PRINCIPAL DISPLAY PANEL - 5 mg Tablet Blister Card Case Carton
NDC 0006-0005-30
This package contains 30 Tablets in 3 Blister Cards.
Each Blister Card contains 10 Tablets.
Belsomra®
(suvorexant) tablets
CIV
5 mg
Dispense the accompanying Medication Guide to each patient.
Each tablet contains 5 mg suvorexant.
USUAL DOSAGE: See Package Insert.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled
Room Temperature]. Store in the original package until use, to protect from light and moisture.
Selling or giving away Belsomra® may harm others and is against the law.
Rx only
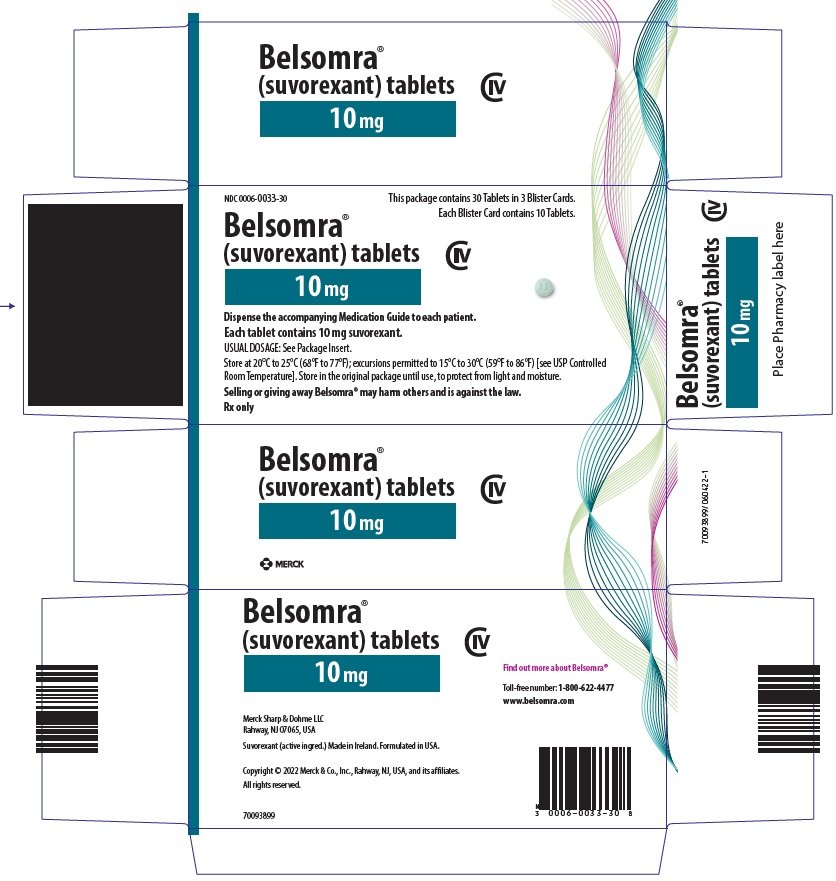
PRINCIPAL DISPLAY PANEL - 10 mg Tablet Blister Card Case Carton
NDC 0006-0033-30
This package contains 30 Tablets in 3 Blister Cards.
Each Blister Card contains 10 Tablets.
Belsomra®
(suvorexant) tablets
CIV
10 mg
Dispense the accompanying Medication Guide to each patient.
Each tablet contains 10 mg suvorexant.
USUAL DOSAGE: See Package Insert.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled
Room Temperature]. Store in the original package until use, to protect from light and moisture.
Selling or giving away Belsomra® may harm others and is against the law.
Rx only
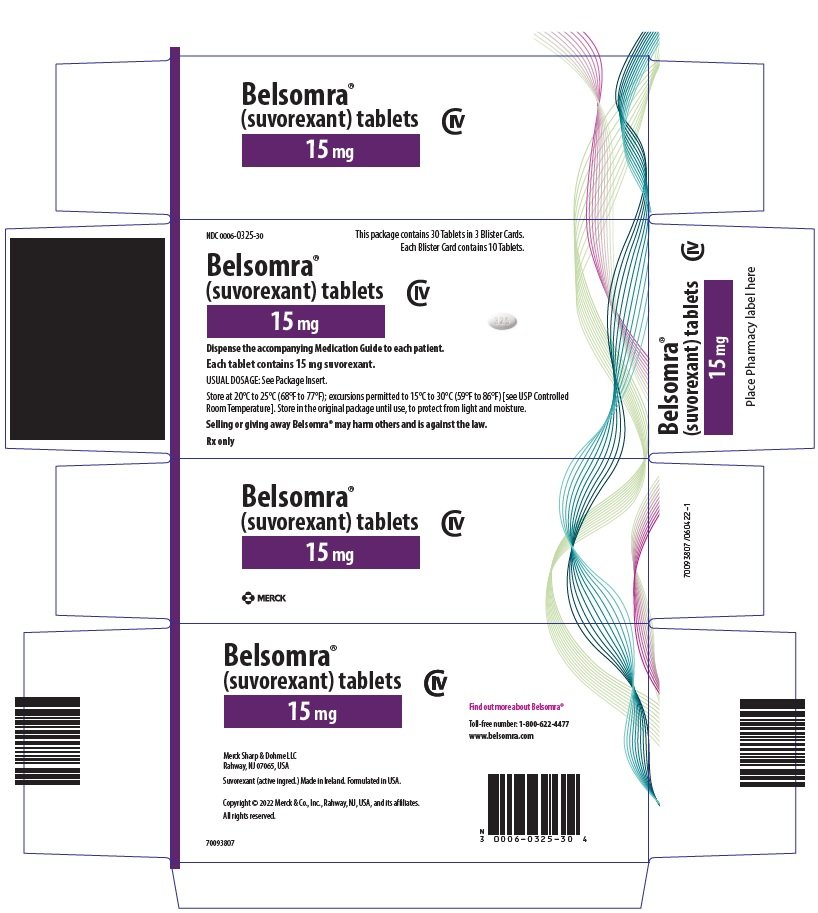
PRINCIPAL DISPLAY PANEL - 15 mg Tablet Blister Card Case Carton
NDC 0006-0325-30
This package contains 30 Tablets in 3 Blister Cards.
Each Blister Card contains 10 Tablets.
Belsomra®
(suvorexant) tablets
CIV
15 mg
Dispense the accompanying Medication Guide to each patient.
Each tablet contains 15 mg suvorexant.
USUAL DOSAGE: See Package Insert.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled
Room Temperature]. Store in the original package until use, to protect from light and moisture.
Selling or giving away Belsomra® may harm others and is against the law.
Rx only
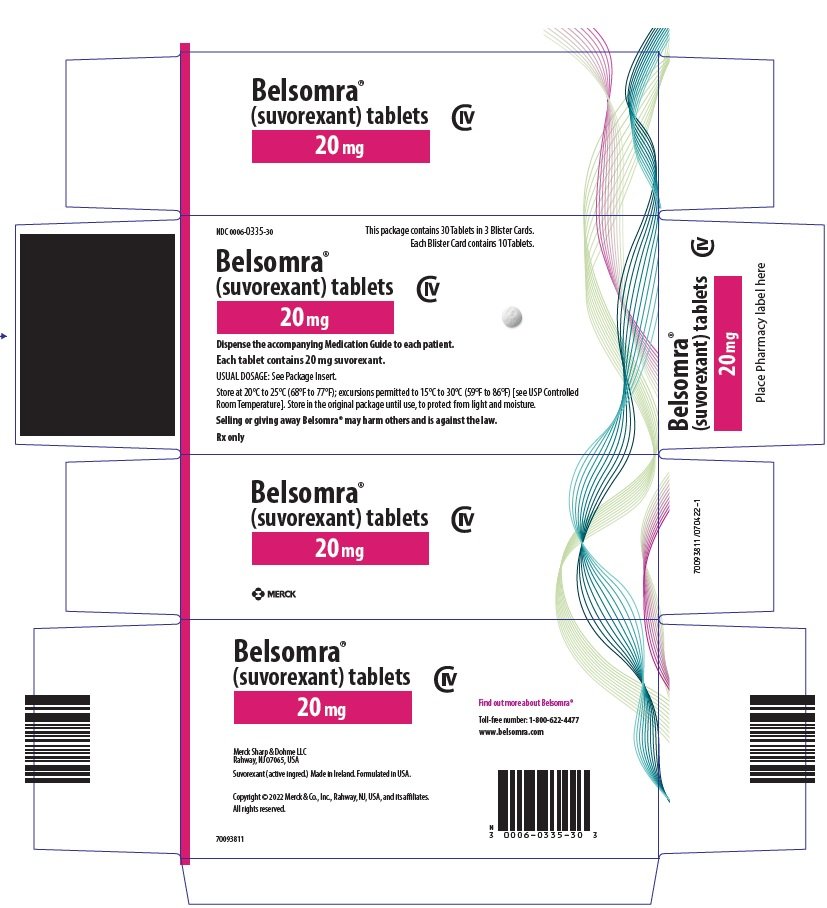
PRINCIPAL DISPLAY PANEL - 20 mg Tablet Blister Card Case Carton
NDC 0006-0335-30
This package contains 30 Tablets in 3 Blister Cards.
Each Blister Card contains 10 Tablets.
Belsomra®
(suvorexant) tablets
CIV
20 mg
Dispense the accompanying Medication Guide to each patient.
Each tablet contains 20 mg suvorexant.
USUAL DOSAGE: See Package Insert.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled
Room Temperature]. Store in the original package until use, to protect from light and moisture.
Selling or giving away Belsomra® may harm others and is against the law.
Rx only
| BELSOMRA
suvorexant tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| BELSOMRA
suvorexant tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| BELSOMRA
suvorexant tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| BELSOMRA
suvorexant tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Merck Sharp & Dohme LLC (118446553) |
Frequently asked questions
More about Belsomra (suvorexant)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (546)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: miscellaneous anxiolytics, sedatives and hypnotics
- Breastfeeding
- En español
Patient resources
Professional resources
Related treatment guides
Copyright © 2014-2025 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.