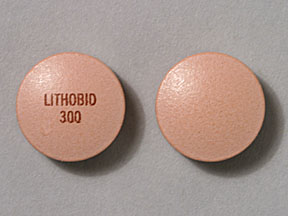
LITHOBID 300 Pill: peach, round, 10mm
The pill with imprint LITHOBID 300 (Peach, Round, 10mm) has been identified as Lithium Carbonate Extended-Release 300 mg and is used for Bipolar Disorder, Schizoaffective Disorder, and Mania. It belongs to the drug class antimanic agents and is not a controlled substance.
Images for LITHOBID 300
Lithium Carbonate Extended-Release
- Imprint
- LITHOBID 300
- Strength
- 300 mg
- Color
- Peach
- Size
- 10.00 mm
- Shape
- Round
- Availability
- Prescription only
- Drug Class
- Antimanic agents
- Pregnancy Category
- D - Positive evidence of risk
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- ANI Pharmaceuticals, Inc.
- Inactive Ingredients
-
calcium stearate,
carnauba wax,
hypromellose 2910 (50 mPa.s),
FD&C Blue No. 2,
FD&C Red No. 40,
FD&C Yellow No. 6,
povidone k30,
propylene glycol,
sodium chloride,
sodium lauryl sulfate,
sodium starch glycolate type A potato,
sorbitol,
titanium dioxide
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 62559-0340 | |
| 68968-4492 (Discontinued) | Noven Pharmaceuticals, Inc. |
Related images for "LITHOBID 300"
More about lithium
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (499)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: antimanic agents
- Breastfeeding
- En español
Patient resources
Other brands
Eskalith, Lithobid, Eskalith-CR
Professional resources
- Lithium monograph
- Lithium (FDA)
- Lithium Capsules (FDA)
- Lithium Carbonate (FDA)
- Lithium Carbonate ER Tablets (FDA)
Other brands
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.