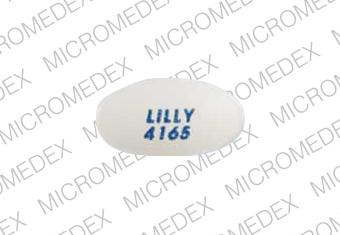
LILLY 4165 Pill: white, oval, 12mm
Generic Name: raloxifene
The pill with imprint LILLY 4165 (White, Oval, 12mm) has been identified as Evista 60 mg and is used for Breast Cancer, Prevention, Prevention of Osteoporosis, and Osteoporosis. It belongs to the drug classes hormones/antineoplastics, selective estrogen receptor modulators and is not a controlled substance.
Images for LILLY 4165




Evista
- Generic Name
- raloxifene
- Imprint
- LILLY 4165
- Strength
- 60 mg
- Color
- White
- Size
- 12.00 mm
- Shape
- Oval
- Availability
- Prescription only
- Drug Class
- Hormones/antineoplastics, Selective estrogen receptor modulators
- Pregnancy Category
- X - Not for use in pregnancy
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Lilly, Eli and Company
- Inactive Ingredients
-
carnauba wax,
crospovidone,
hypromelloses,
lactose monohydrate,
lactose anhydrous,
magnesium stearate,
polyethylene glycol,
povidone,
titanium dioxide,
polysorbate 80,
propylene glycol
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 00002-4165 (Discontinued) | Eli Lilly and Company |
| 00002-4184 | Eli Lilly and Company |
| 49999-0458 (Discontinued) | Lake Erie Medical and Surgical Supply (repackager) |
| 54569-4628 (Discontinued) | A-S Medication Solutions, LLC (repackager) |
| 54868-4170 (Discontinued) | Physicians Total Care Inc. (repackager) |
| 55289-0266 (Discontinued) | PDRX Pharmaceuticals Inc. (repackager) |
See also:
More about Evista (raloxifene)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (12)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Generic availability
- Drug class: hormones/antineoplastics
- En español
Patient resources
Professional resources
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
