Sipuleucel-T Dosage
Medically reviewed by Drugs.com. Last updated on Dec 27, 2024.
Usual Adult Dose for:
Additional dosage information:
Usual Adult Dose for Prostate Cancer
3 complete doses given at approximately 2-week intervals; infuse drug intravenously over 60 minutes.
Comments:
- Peripheral blood mononuclear cells from patients are obtained via the leukapheresis procedure, which should be performed approximately 3 days prior to each infusion.
- Patients should be pre-medicated orally with acetaminophen and an antihistamine (such as diphenhydramine) approximately 30 minutes prior to administration to minimize potential acute infusion reactions.
- The patient should be observed for acute infusion reactions for at least 30 minutes following each infusion.
- The maximum dosing interval has not been established.
- This drug is intended solely for autologous use.
- This drug is for IV use only; a cell filter should not be used.
Use: Treatment of asymptomatic or minimally symptomatic metastatic castrate-resistant (hormone-refractory) prostate cancer.
Renal Dose Adjustments
Data not available
Liver Dose Adjustments
Data not available
Dose Adjustments
If Acute Infusion Reactions Occur:
- Interrupt or slow the infusion depending on the reaction severity, and administer appropriate medical treatment as needed.
- An interrupted infusion should not be resumed if the infusion bag will be at room temperature for more than 3 hours.
Precautions
Safety and efficacy have not been established in patients younger than 18 years.
Consult WARNINGS section for additional precautions.
Dialysis
Data not available
Other Comments
Administration Advice:
- The infusion should not be initiated until confirmation of product release is received from the manufacturer. The manufacturer will send the Final Product Disposition Notification form containing the patient identifiers, expiration date and time, and the disposition status (approved for infusion or rejected) to the infusion site.
- The infusion must begin prior to the expiration date and time on the Final Product Disposition Notification form and product label; expired product should not be administered.
- Before infusion, it should be confirmed that the patient's identity matches the patient identifiers on the infusion bag and the Final Product Disposition Notification form.
- Infusion bags that leak, are damaged, or contain clumps after gentle manual mixing should not be administered.
- Patients unable to receive a scheduled infusion need to undergo an additional leukapheresis procedure prior to continuing a treatment course.
- A patient needs adequate peripheral venous access or a central venous catheter to accommodate the leukapheresis procedure and infusions of this drug.
Storage and Handling Requirements:
- The infusion bag should be gently mixed and re-suspended manually to disperse small clumps of cellular material.
- The infusion bag should remain in the insulated polyurethane container (which should not be removed from the shipping box), and the lid of the insulated container should not be opened until administration time.
- Once the infusion bag is removed from the insulated container, it should remain at room temperature for no more than 3 hours.
General:
- This drug consists of autologous peripheral blood mononuclear cells, including antigen presenting cells (APCs) that have been activated during a defined culture period with recombinant human protein PAP-GM-CSF, which consists of an antigen expressed in prostate cancer tissue (prostatic acid phosphatase/PAP) linked to an immune cell activator (granulocyte-macrophage colony-stimulating factor/GM-CSF).
- This drug contains APCs, T cells, B cells, natural killer cells and other cells; the number of cells present and the cellular composition of each dose will vary as it is dependent on the composition of cells obtained from the leukapheresis procedure.
- The potency of this drug is partly determined by measuring the increased expression of the CD54 molecule (ICAM-1) on the surface of APCs after culture with PAP-GM-CSF.
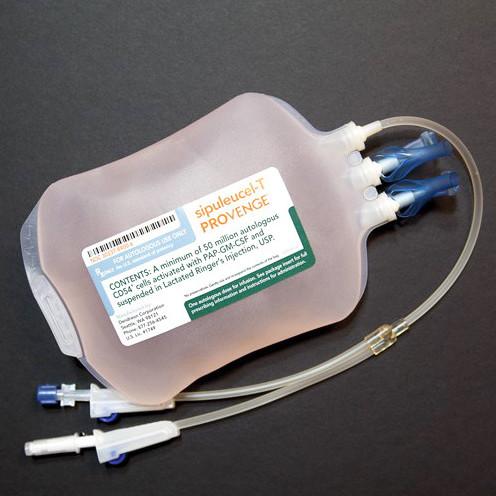
- Each dose contains a minimum of 50 million autologous CD54+ cells activated with PAP-GM-CSF, suspended in 250 mL of Lactated Ringer's Injection.
- There are no known instances of overdosage from either a single infusion or a full course of therapy.
Patient Advice:
- If you have a central venous catheter, inform your healthcare provider if you have fevers or any swelling/redness around the catheter site because these symptoms could be signs of an infected catheter.
- Let your healthcare provider know if you are taking immunosuppressive agents
More about sipuleucel-T
- Check interactions
- Compare alternatives
- Reviews (7)
- Side effects
- During pregnancy
- Drug class: therapeutic vaccines
- En español
Patient resources
Other brands
Professional resources
Other brands
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.