Ilumya Injection Dosage
Generic name: TILDRAKIZUMAB 100mg in 1mL
Dosage form: injection, solution
Drug class: Interleukin inhibitors
Medically reviewed by Drugs.com. Last updated on Feb 26, 2025.
Recommended Evaluation and Immunization Prior to Treatment Initiation
- Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with ILUMYA.
- Consider completion of all age appropriate immunizations according to current immunization guidelines.
Dosage
ILUMYA is administered by subcutaneous injection. The recommended dosage is 100 mg at Weeks 0, 4, and every 12 weeks thereafter. Each syringe contains 1 mL of 100 mg/mL tildrakizumab-asmn.
Important Administration Instructions
ILUMYA should only be administered by a healthcare provider. Administer ILUMYA subcutaneously. Each prefilled syringe is for single-dose only. Inject the full amount (1 mL), which provides 100 mg of tildrakizumab per syringe. If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regularly scheduled interval.
Preparation and Administration of ILUMYA
Before injection, remove ILUMYA carton from the refrigerator, and let the prefilled syringe (in the ILUMYA carton with the lid closed) sit at room temperature for 30 minutes.
Follow the instructions on the ILUMYA carton to remove the prefilled syringe correctly, and remove only when ready to inject. Do not pull off the needle cover until you are ready to inject.
Inspect ILUMYA visually for particulate matter and discoloration prior to administration. ILUMYA is a clear to slightly opalescent, colorless to slightly yellow solution. Do not use if the liquid contains visible particles or the syringe is damaged. Air bubbles may be present; there is no need to remove them.
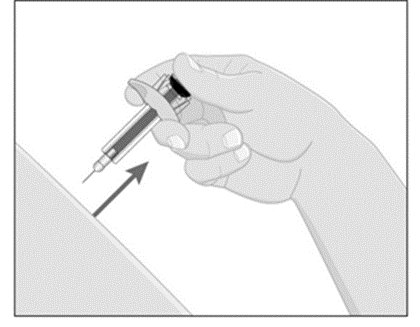
Choose an injection site with clear skin and easy access (such as abdomen, thighs, or upper arm). Do not administer 2 inches around the navel or where the skin is tender, bruised, erythematous, indurated, or affected by psoriasis. Also do not inject into scars, stretch marks, or blood vessels.
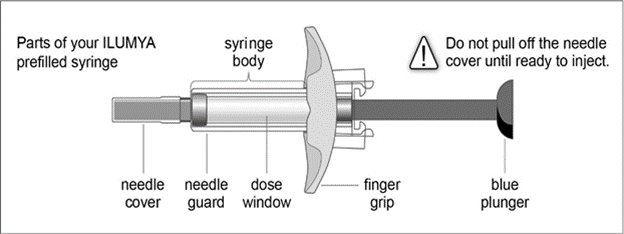
- While holding the body of the syringe, pull the needle cover straight off (do not twist) and discard.
- Inject ILUMYA subcutaneously as recommended.
- Press down the blue plunger until it can go no further. This activates the safety mechanism that will ensure full retraction of the needle after the injection is given.
- Remove the needle from the skin entirely before letting go of the blue plunger. After the blue plunger is released, the safety lock will draw the needle inside the needle guard.
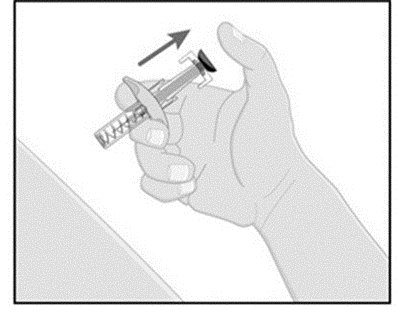
- Discard any unused portion. Dispose of used syringe.
Frequently asked questions
- What are IL-23 Inhibitors and how do they work?
- Skyrizi vs Ilumya. What’s the difference?
- Does Ilumya treat psoriatic arthritis?
- How does Ilumya compare to other treatments?
- How does Ilumya work to treat psoriasis?
- How long does it take Ilumya to work?
- How does Ilumya affect my immune system?
- What are the side effects of Ilumya?
- How is Ilumya administered?
More about Ilumya (tildrakizumab)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (8)
- Drug images
- Side effects
- Patient tips
- During pregnancy
- Support group
- FDA approval history
- Drug class: interleukin inhibitors
- Breastfeeding
- En español
Patient resources
Professional resources
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.