Urea Hydrating Foam: Package Insert / Prescribing Info
Package insert / product label
Dosage form: aerosol, foam
Drug class: Topical emollients
Medically reviewed by Drugs.com. Last updated on May 26, 2024.
On This Page
DESCRIPTION
Urea 35% Hydrating Topical Foam is a keratolytic emollient in a water and lipid based foam containing lactic acid which is a gentle, but potent, tissue softener for skin and nails.
Each gram of Urea 35% Hydrating Topical Foam contains Urea 35% as the active ingredient, and the following inactive ingredients: dimethicone, ethylparaben, glycerin, lactic acid,
methylparaben, phenoxyethanol, polysorbate 20, povidone, propylene glycol, propylparaben, purified water, stearic acid, trolamine, and in propellants butane and propane.
CHEMICAL STRUCTURE
Urea has the following chemical structure:
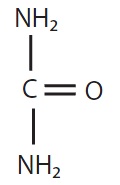
CLINICAL PHARMACOLOGY
Topically applied urea dissolves the intercellular matrix of the skin which results in enhanced shedding of scaly, dry skin and thus a softening of the hyperkeratotic areas of the skin.
Urea topically applied to the nail plate has a similar effect on the intercellular matrix of the nail plate.
INDICATIONS AND USAGE
For enzymatic debridement and promotion of normal healing of surface lesions, particularly where healing is retarded by local infection, necrotic tissue, fibrinous or purulent debris,
or eschar. Topically applied urea is useful for the treatment of hyperkeratotic conditions such as dermatitis, psoriasis, xerosis, ichthyosis, eczema, keratosis, keratoderma, and dry,
rough skin, as well as corns and calluses and damaged, ingrown and devitalized nails.
WARNINGS
Urea 35% Hydrating Topical Foam is for external use only. It is not for ophthalmic, oral, anal or intravaginal use. Contact with eyes, lips, and all mucous membranes should be
avoided. Urea 35% Hydrating Topical Foam should not be used by persons who have a known hypersensitivity to urea or any of the other listed ingredients.
PRECAUTIONS
Urea 35% Hydrating Topical Foam should be used only as directed by a physician and should not be used to treat any condition other than that for which it is prescribed. If redness
or irritation occurs, discontinue use and consult with a prescribing physician.
Pregnancy (Category B) – Animal reproduction studies have not been performed with topically applied urea and it is not known whether Urea 35% Hydrating Topical Foam can
cause fetal harm when administered to a pregnant woman. Nevertheless, Urea 35% Hydrating Topical Foam should be used by a pregnant woman only if necessary.
Nursing Mothers – It is not known whether topically applied urea is excreted in human milk. Due to the fact that many drugs are excreted in human milk, caution should be
exercised by physicians when administering Urea 35% Hydrating Topical Foam to nursing mothers.
DOSAGE AND ADMINISTRATION
Unless otherwise directed by a prescribing physician, Urea 35% Hydrating Topical Foam should be applied to affected area twice a day. Urea 35% Hydrating Topical Foam should
be rubbed into the skin until it is completely absorbed.
HOW SUPPLIED
Urea 35% Hydrating Topical Foam is supplied in a 150 gram or 5.3 ounce aerosolized canister bearing the NDC Number 42192-115-15.
NDC 42192-115-15
Urea 35% Hydrating
Topical Foam
Rx Only
Net Wt. 5.3 oz. (150 g)
NDC 42192-115-15
Rx Only
Net Wt. 5.3 oz. (150 g)
Dosage and Administration: Clean and dry affected
skin. Then apply Urea 35% Hydrating Topical Foam
topically to cover affected skin twice per day, or as
directed by a physician. Rub in until completely absorbed.
Shake vigorously before each application and invert
can to administer.
Store at room temperature 59° - 77°F (15° - 25°C).
See prescribing information for additional details.
Ingredients: urea 35%, dimethicone, ethylparaben,
glycerin, lactic acid, methylparaben, phenoxyethanol,
polysorbate 20, povidone, propylene glycol, propylparaben,
purified water, stearic acid, trolamine, and in
propellants butane and propane.
Warning: Contents under pressure. Do not puncture or
incinerate. Do not expose to temperatures over 120°F
(48°C) even when empty.
Keep out of reach of children.
Manufactured for:
Acella Pharmaceuticals, LLC
Alpharetta, GA 30009
1-800-541-4802

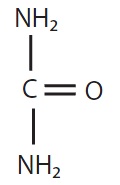
| UREA HYDRATING TOPICAL
urea aerosol, foam |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| Labeler - Acella Pharmaceuticals (825380939) |
| Registrant - Acella Pharmaceuticals (825380939) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Acella Pharmaceuticlas | 825380939 | manufacture(42192-115) | |
Related/similar drugs
More about urea topical
- Compare alternatives
- Pricing & coupons
- Reviews (28)
- Side effects
- Dosage information
- Drug class: topical emollients
Patient resources
Professional resources
Other brands
MeTopic, Keralac, Rynoderm, Umecta Mousse, ... +2 more
