URE-39 Cream: Package Insert / Prescribing Info
Package insert / product label
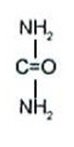
Generic name: urea
Dosage form: cream
Medically reviewed by Drugs.com. Last updated on Mar 31, 2025.
On This Page
URE-39 Cream Description
URE-39 Cream is a potent keratolytic emollient which is a gentle, yet potent, tissue softener for skin and/or nails.
Each gram of URE-39 Cream contains:
ACTIVE: 39% Urea in a cream base of:
INACTIVES: Deionized Water, Carthamus Tinctorius (Safflower) Seed Oil, Emulsifying Wax, Glycerine, Propylene Glycol, Glyceryl Stearate, PEG 100 Stearate, Cetyl Alcohol, Dimethicone, C12/15 Alkyl Benzoate, Anthemis Nobilis (Chamomile) Flower Extract, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Aloe Barbadensis Leaf, Phenoxyethanol, Methylparaben, Ethylparaben, Butylparaben, Propylparaben, and Isobutylparaben.
URE-39 Cream - Clinical Pharmacology
Urea gently dissolves the intercellular matrix which results in loosening the horny layer of skin and shedding scaly skin at regular intervals, thereby softening hyperkeratotic areas. Urea also hydrates and gently dissolves the intercellular matrix of the nail plate, which can result in the softening and eventual debridement of the nail plate.
Indications and Usage for URE-39 Cream
For debridement and promotion of normal healing of hyperkeratotic surface lesions, particularly where healing is retarded by local infection, necrotic tissue, fibrinous or prurient debris or eschar. Urea is useful for the treatment of hyperkeratotic conditions such as dry, rough skin, dermatitis, psoriasis, xerosis, ichthyosis, eczema, keratosis, keratoderma, corns and calluses, as well as damaged, ingrown and devitalized nails.
Contraindications
URE-39 Cream is contraindicated in patients with known hypersensitivity to any of the listed ingredients.
Warnings
For external use only. Avoid contact with eyes, lips or mucous membranes. Do not use on areas of broken skin.
Precautions
Stop use and ask a doctor if redness or irritation develops. After applying this medication, wash hands and unaffected areas thoroughly. If swallowed, get medical help or contact Poison Control Center right away. KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN.
PREGNANCY
Pregnancy Category B
Animal reproduction studies have revealed no evidence of harm to the fetus, however, there are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, URE-39 Cream should be given to a pregnant woman only if clearly needed.
Adverse Reactions/Side Effects
Transient stinging, burning, itching or irritation may occur and normally disappear on discontinuing the medication.
Related/similar drugs
URE-39 Cream Dosage and Administration
Apply URE-39 Cream to affected skin two to three times per day as needed or as directed by a physician. Rub in until completely absorbed. Apply to diseased or damaged nail tissue two to three times per day or as directed by a physician. Best applied to affected areas immediately after showering and just before bedtime.
| URE-39
urea cream |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - SOLUTECH PHARMACEUTICALS LLC (080040396) |
More about URE-39 (urea topical)
Professional resources
Other brands
MeTopic, Keralac, Rynoderm, Umecta Mousse, URE-K