Ultravate Lotion: Package Insert / Prescribing Info
Package insert / product label
Generic name: halobetasol propionate
Dosage form: lotion
Drug class: Topical steroids
Medically reviewed by Drugs.com. Last updated on May 2, 2025.
On This Page
Highlights of Prescribing Information
See full prescribing information for ULTRAVATE Lotion.
ULTRAVATE (halobetasol propionate) lotion, for topical use
Initial U.S. Approval: 1990
Recent Major Changes
| Indication and Usage ( 1) | 08/2020 |
| Warnings and Precautions (5.1) | 08/2020 |
Indications and Usage for Ultravate Lotion
ULTRAVATE lotion is a corticosteroid indicated for the topical treatment of plaque psoriasis in patients 12 years of age and older. ( 1)
Ultravate Lotion Dosage and Administration
- Apply a thin layer to the affected areas twice daily. (2)
- Limit use to 50 g/week. (2)
- Discontinue treatment when control is achieved. (2)
- If no improvement is seen within 2 weeks, reassess diagnosis. (2)
- Treatment beyond 2 consecutive weeks is not recommended. (2)
- Do not use with occlusive dressings unless directed by a physician. (2)
- Avoid use on the face, scalp, groin, or axillae. (2)
- Not for ophthalmic, oral, or intravaginal use. (2)
Dosage Forms and Strengths
Lotion: 0.05% (0.5 mg/g). (3) (3)
Contraindications
None. (4)
Warnings and Precautions
- Reversible hypothalamic-pituitary-adrenal (HPA) axis suppression may occur, with the potential for glucocorticosteroid insufficiency during or after treatment. Systemic absorption may require evaluation for HPA axis suppression. (5.1)
- Systemic effects of topical corticosteroids may also include Cushing's syndrome, hyperglycemia, and glucosuria. Use of potent corticosteroids on large areas, for prolonged durations, under occlusive dressings, or on an altered skin barrier may increase systemic exposure. (5.1)
- Children may be more susceptible to systemic toxicity when treated with topical corticosteroids. (5.1, 8.4)
- Local adverse reactions with topical steroids may include atrophy, striae, irritation, acneiform eruptions, hypopigmentation, and allergic contact dermatitis. Adverse reactions may be more likely to occur with occlusive use or more potent corticosteroids. (5.2, 5.5)
- Topical corticosteroids may increase the risk of cataract and glaucoma formation. If visual symptoms occur, consider referral to an ophthalmologist tor evaluation. (5.3)
- Initiate appropriate therapy if concomitant skin infections develop. (5.4)
Adverse Reactions/Side Effects
The most commonly reported adverse reactions (≥1%) are telangiectasia, application site atrophy, and headache. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact LAcer Pharma, LLC at 1-888-403-8874 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Revised: 4/2025
Full Prescribing Information
1. Indications and Usage for Ultravate Lotion
ULTRAVATE lotion is indicated for the topical treatment of plaque psoriasis in patients 12 years of age and older.
2. Ultravate Lotion Dosage and Administration
Apply a thin layer of ULTRAVATE lotion to the affected skin twice daily for up to two weeks. Rub in gently.
Discontinue therapy when control is achieved. If no improvement is seen within two weeks, reassessment of diagnosis may
be necessary.
Treatment beyond two weeks is not recommended and the total dosage should not exceed 50 grams (50 ml) per week because of the potential for the drug to suppress the hypothalamic-pituitary-adrenal (HPA) axis {see Warnings and Precautions 5.1].Do not use with occlusive dressings unless directed by a physician.
ULTRAVATE lotion is for external use only.
Avoid use on the face, scalp, groin, or axillae.
ULTRAVATE lotion is not for ophthalmic, oral, or intravaginal use.
3. Dosage Forms and Strengths
ULTRAVATE (halobetasol propionate) lotion, 0.05% is a white to off-white lotion. Each gram of ULTRAVATE lotion contains 0.5 mg of halobetasol propionate.
5. Warnings and Precautions
5.1 Effects on Endocrine System
ULTRAVATE lotion has been shown to suppress the hypothalamic-pituitary-adrenal (HPA) axis.Systemic effects of topical corticosteroids may include reversible HPA axis suppression, with the potential for glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment of the topical corticosteroid.The potential for hypothalamic-pituitary adrenal (HPA) suppression with ULTRAVATE lotion was evaluated in the following studies:
- In a study of 20 adult subjects with moderate to severe plaque psoriasis involving 20% of their body surtace area. ULTRAVATE lotion produced HPA axis suppression when used twice daily for two weeks in 5 out of 20 (25%) adult subjects with plaque psoriasis. The effects of HPA axis suppression were reversible on discontinuation of the treatment [see Clinical Pharmacology (12.2)].
- In another clinical study, 16 adolescent subjects (12 to less than 17 years old) with moderate to severe plaque psoriasis involving 10% or more of their body surtace area applied a maximum of approximately 50 grams of ULTRAVATE lotion to affected areas twice daily for two weeks. Of the 14 subjects evaluated for HPA axis suppression, adrenal suppression occurred in 1 subject (7%) which recovered upon retest [see Clinical Pharmacology (12.2)].
Because of the potential for systemic absorption, use of topical corticosteroids, including ULTRAVATE lotion, may require that patients be evaluated periodically for evidence of HPA axis suppression. Factors that predispose a patient using a topical corticosteroid to HPA axis suppression include the use of more potent corticosteroids, use over large surtace areas, prolonged use, occlusive use, use on an altered skin barrier, concomitant use of multiple corticosteroid-containing products, liver failure, and young age. An ACTH stimulation test may be helpful in evaluating patients for HPA axis suppression.If HPA axis suppression is documented, attempt to gradually withdraw the drug, reduce the frequency of application, or substitute a less potent steroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt and complete upon discontinuation of topical corticosteroids.Systemic effects of topical corticosteroids may also include Cushing's syndrome, hyperglycemia, and glucosuria. Use of more than one corticosteroid-containing product at the same time may increase the total systemic exposure to topical corticosteroids. Pediatric patients may be more susceptible than adults to systemic toxicity from the use of topical corticosteroids due to their larger surtace-to-body mass ratios [see Use in Specific Populations (8.4)].
5.2 Local Adverse Reactions
Local adverse reactions from topical corticosteroids may include atrophy, striae, telangiectasias, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria. These may be more likely to occur with occlusive use, prolonged use, or use of higher potency corticosteroids, including ULTRAVATE lotion. Some local adverse reactions may be irreversible.
5.3 Ophthalmic Adverse Reactions
Use of topical corticosteroids may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported in postmarketing experience with the use of topical corticosteroid products.
Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
5.4 Concomitant Skin Infections
Use an appropriate antimicrobial agent if a skin infection is present or develops. If a favorable response does not occur promptly, discontinue use of ULTRAVATE lotion until the infection has been adequately treated.
5.5 Allergic Contact Dermatitis
Allergic contact dermatitis with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation. Consider confirmation of a clinical diagnosis of allergic contact dermatitis by appropriate patch testing. Discontinue ULTRAVATE lotion if allergic contact dermatitis is established.
6. Adverse Reactions/Side Effects
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
During randomized, controlled, blinded clinical trials 277 adults with plaque psoriasis were treated with ULTRAVATE lotion twice daily for up to two weeks (up to approximately 50 grams/week).
Table 1 presents adverse reactions that occured in at least 1% of subjects treated with ULTRAVATE lotion twice daily for up to two weeks, and more frequently than in vehicle-treated subjects.
| ULTRAVATE Lotion
(N=277) | Vehicle Lotion
(N=259) |
|
|---|---|---|
| Adverse Reaction | % | % |
| Telangiectasia | 1% | 0% |
| Application site atrophy | 1% | <1% |
| Headache | 1% | <1% |
Less common dverse reactions (incidence less than 1% but greater than 0.1%) that occured in subjects treated with ULTRAVATE lotion included application site discoloration, herpes zoster, influenza, nasopharyngitis, otitis media acute, throat infection, wound, and increased blood pressure.
Related/similar drugs
8. Use In Specific Populations
8.1 Pregnancy
There are no available data on Ultravate lotion use in pregnant women to inform a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Published data report an increased risk of low birthweight with the use of greater than 300 grams of potent or very potent topical corticosteroid during pregnancy. In animal reproduction studies, halobetasol propionate administered systemically during organogenesis to pregnant rats at 13 and 33 times the human topical dose and to pregnant rabbits at 3 times the human topical dose resulted in teratogenic and embryotoxic effects [see Data]. The clinical relevance of the animal findings is not clear.
The background risk of major birth defects and miscarriage for the indicated population are unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Human Data
Multiple observational studies found no significant associations between maternal use of topical corticosteroids of any potency and congenital malformations, preterm delivery, or fetal mortality. However, when the dispensed amount of potent or very potent topical corticosteroid exceeded 300 g during the entire pregnancy, use was associated with an increase in low birth weight infants.
Animal Data
Halobetasol propionate has been shown to be teratogenic in rats and rabbits when given systemically during organogenesis at doses of 0.04 to 0.1 mg/kg/day in rats and 0.01 mg/kg/day in rabbits. These doses are approximately 13, 33, and 3 times, respectively, the human topical dose of halobetasol propionate, 0.05%. Halobetasol propionate was embryotoxic in rabbits but not in rats.Cleft palate was observed in both rats and rabbits. Omphalocele was seen in rats, but not in rabbits.
8.2 Lactation
Risk Summary
There are no data on the presence of halobetasol propionate or its metabolites in human milk, the effects on the breastted infant, or the effects on milk production after topical application to women who are breastteeding.
Systemically administered corticosteroids appear in human milk and could suppress growth, intertere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. The developmental and health benefits of breastteeding should be considered along with the mother's clinical need for UL TRAVATE lotion and any potentialadverse effects on the breastted infant from ULTRAVATE lotion or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of ULTRAVATE lotion for the treatment of moderate to severe plaque psoriasis have been established in patients 12 years of age and older. It is supported by evidence from adequate and well-controlled trials in adults and from one uncontrolled safety trial in 16 adolescents (12 to less than 17 years of age). Adolescent patients with moderate to severe plaque psoriasis covering a minimum of 10% of the total body surface area were treated twice daily for 2 weeks with ULTRAVATE lotion. Hypothalamic-pituitary adrenal (HPA) axis function (ACTH stimulation test) was evaluated in a subset of 14 patients. After 2 weeks of treatment, 1 of 14 patients (7%) experienced laboratory evidence of adrenal suppression (i.e., cortisol serum level of sl 8 μg/dL) that recovered upon retest. No other adverse reactions were reported in the study.
Because of higher skin surtace area to body mass ratios, pediatric patients are at a greater risk than adults of HPA axis suppression and Cushing's syndrome when they are treated with topical corticosteroids. They are therefore also at greater risk of adrenal insufficiency during or after withdrawal of treatment. Adverse reactions including striae have been reported with use of topical corticosteroids in infants and children.
HPA axis suppression, Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include low plasma cortisol levels and an absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
8.5 Geriatric Use
Clinical studies with ULTRAVATE lotion included 89 subjects aged 65 years and over. No overall differences in safety or effectiveness were observed between these subjects and those younger than 65 years. Clinical studies of ULTRAVATE lotion did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
10. Overdosage
Topically applied ULTRAVATE lotion can be absorbed in sufficient amounts to produce systemic effects [see Warnings and Precautions (5.1)].
11. Ultravate Lotion Description
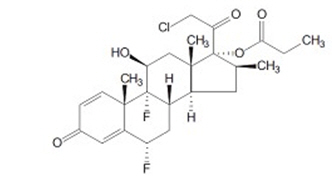
ULTRAVATE (halobetasol propionate) lotion, 0.05% for topical use contains a corticosteroid, halobetasol propionate. The chemical name of halobetasol propionate is 21 chloro-6α, 9-difluoro-11β, 17-dihydroxy-16β-methylpregna-1, 4-diene-3,20-dione 17 propionate. Halobetasol propionate is a white to off-white crystalline powder with a molecular weight of 484.96 and a molecular formula of C 25H 31ClF 2O 5. It is practically insoluble in water and freely soluble in dichloromethane and in acetone. It has the following structural formula:
Each gram of ULTRAVATE lotion contains 0.5 mg of halobetasol propionate in a white to off-white lotion base consisting of diisopropyl adipate, octyldodecanol, ceteth-20, poloxamer 407, cetyl alcohol, stearyl alcohol, propylparaben, butylparaben, propylene glycol, glycerin, carbomer homopolymer, sodium hydroxide, and water.
12. Ultravate Lotion - Clinical Pharmacology
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in plaque psoriasis is unknown.
12.2 Pharmacodynamics
Vasoconstriction:
A vasoconstrictor assay in healthy subjects with ULTRAVATE lotion indicated that the formulation is in the super-high range of potency as compared to other topical corticosteroids; however, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitaty-Adrenal (HPA) Axis Suppression:
The potential for hypothalamic-pituitary adrenal (HPA) suppression was evaluated in the following two studies. In both studies, the criteria for HPA-axis suppression was a serum cortisol level of less than or equal to 18 micrograms per deciliter 30 minutes after stimulation with cosyntropin (adrenocorticotropic hormone, ACTH). In the first study, ULTRAVATE lotion was applied to20 adult subjects with moderate to severe plaque psoriasis. A mean dose of 3.5 grams ULTRAVATE lotion was applied twice daily for two weeks and produced HPA axis suppression in 5 of 20 (25%) subjects. The effects of HPA axis suppression were reversible on retesting at least four weeks after discontinuation of the treatment. In the second study, ULTRAVATE lotion was applied to 16 adolescent subjects 12 years to less than 17 years of age with moderate to severe plaque psoriasis affecting a mean body surtace area of 11.5% (range from 10% to 14%). The mean dose was 3.6 grams applied twice daily for two weeks. A subset of 14 of the 16 completed subjects had evaluable ACTH stimulation tests, and HPA axis suppression was observed in 1 of these 14 subjects (7%). In the second study also, the effects of HPA axis suppression were reversible on retesting at least four weeks after discontinuation of the treatment.
12.3 Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption.
In the HPA clinical study /see Clinical Pharmacology (12.2)1, pharmacokinetics was evaluated in a subgroup of 12 adult subjects. On Day 8, blood was taken just prior to and at 1, 2, 4, 6, 8, and 12 hours following the last application. Plasma concentration of halobetasol propionate (HBP) was measurable in all subjects. Based on the geometric mean plasma concentrations at 12 hours post-application across time, steady-state was achieved by Day 8. The mean (±standard deviation) C maxconcentrations for ULTRAVATE lotion on Day 8 was 201.1 ± 157.5 pg/ml, with the corresponding median T maxvalue of 3 hours (range O - 6 hours); mean area under the halobetasol propionate concentration versus time curve over the dosing interval (AUCt) was 1632 ± 1147 pg•h/ml.
Specific Populations
Pediatric Patients
In the pediatric HPA study [see Clinical Pharmacology (12.2)], trough plasma concentrations of HBP were measured on Day 8 and Day 15 in a subset of 14 subjects. The HBP levels in the plasma were below the quantification limit (20 pg/ml) for all subjects at all time points with the exception of one subject at Day 15 (trough concentration of HBP of 28.2 pg/ml).
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of halobetasol propionate.
In a 90-day repeat-dose toxicity study in rats, topical administration of Halobetasol Propionate Topical Foam at dose concentrations from 0.005% to 0.05% or from 0.011 to 0.11 mg/kg/day of halobetasol propionate resulted in a toxicity profile consistent with long-term exposure to corticosteroids including adrenal atrophy, histopathological changes in several organ systems indicative of severe immune suppression, and opportunistic fungal and bacterial infections. A no observable adverse effect level could not be determined in this study. Although the clinical relevance of the findings in animals to humans is not clear, sustained glucocorticoid-related immune suppression may increase the risk of infection and possibly the risk of carcinogenesis.
Halobetasol propionate was not found to be genotoxic in the Ames/Salmonella assay, in the Chinese hamster CHO/HGPRT assay, in the mouse micronucleus test, in the sister chromatid exchange test in somatic cells of the Chinese hamster, or in the chromosome aberration test in somatic cells of Chinese hamsters. Positive mutagenicity effects were observed in two genotoxicity assays: Chinese hamster nuclear anomaly test and mouse lymphoma gene mutation assay in vitro.
Studies in the rat following oral administration at dose levels up to 0.05 mg/kg/day indicated no impairment of fertility or general reproductive performance.
14. Clinical Studies
ULTRAVATE lotion was evaluated for the treatment of moderate to severe plaque psoriasis in two multicenter, randomized, doble-blind, vehicle-controlled trials.
These trials were conducted in 443 subjects 18 years of age and older with plaque psoriasis involving between 2% and 12% body surface area. Baseline disease severity was determined using a static, five-level global evaluation scale, on which a subject scored either moderate or severe. Overall, 57% of subjects were male and 86% were Caucasian.
Subjects applied ULTRAVATE lotion or vehicle to all affected areas twice daily for up to 14 consecutive days.
The primary measure of efficacy was Overall Treatment Success, defined as the proportion of subjects who were cleared or almost cleared with at least a two grade improvement from baseline at Week 2 (end of treatment). Table 2 presents these results.
| Study 1 | Study 2 | |||
|---|---|---|---|---|
| ULTRAVATE Lotion
N=110 | Vehicle Lotion
N=76 | ULTRAVATE Lotion
N=110 | Vehicle Lotion
N=112 |
|
|
||||
| Overall Treatment Success * | 49 (44.5%) | 7 (6.3%) | 49 (44.5%) | 8 (7.1%) |
The secondary measures of efficacy were Treatment Success for individual signs of psoriasis (scaling, erythema, and plaque
elevation) at the end of treatment (see Table 3).
|
||||
| Study 1 | Study 2 | |||
| Treatment Success * | ULTRAVATE Lotion N=10 | Vehicle Lotion N=111 | ULTRAVATE Lotion N=110 | Vehicle Lotio N=112 |
| Scaling | 61 (55.5%) | 12 (10.8%) | 65 (59.1%) | 11 (9.8%) |
| Erythema | 40 (36.4%) | 8 (7.2%) | 48 (43.6%) | 12 (10.7%) |
| Plaque Elevation | 50 (45.5%) | 9 (8.1%) | 48 (43.6%) | 9 (8.0%) |
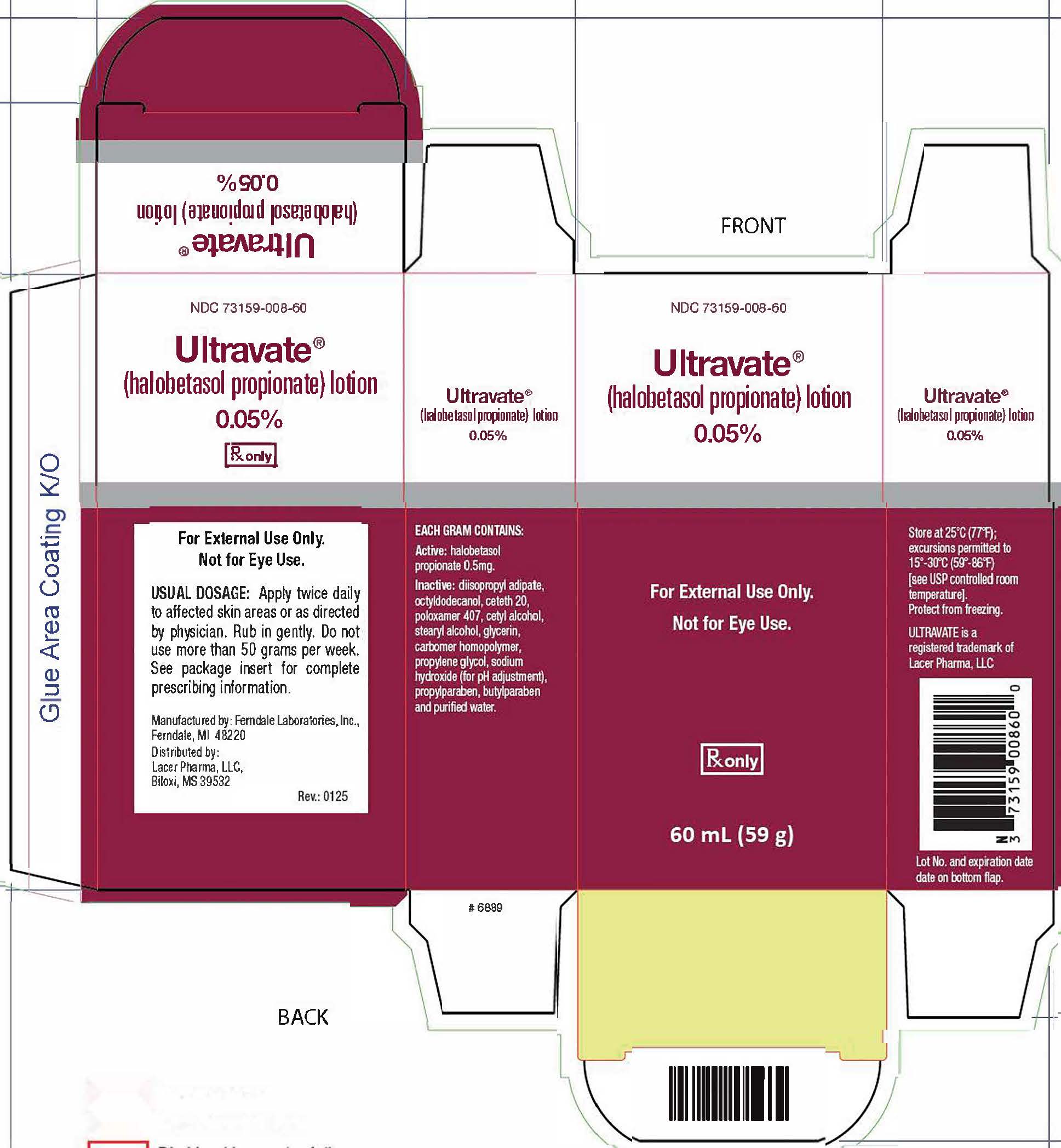
16. How is Ultravate Lotion supplied
ULTRAVATE lotion, 0.05 % is white to off-white lotion. It is supplied in an oval tapered white high-density polyethylene bottle with a white polypropylene disc cap. Each bottle contains 60 ml (59 g) of ULTRAVATE lotion.
NOC 73159-008-60 60 ml (59 g) bottle
Store at 25'C (77'F); excursions permitted to 15°G and 30°G (59° F to 86°F) [see USP Controlled Room Temperature]. Do not freeze.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Advise patients using ULTRAVATE lotion of the following information and instructions:
Important Administration Instructions
Instruct patients to discontinue ULTRAVATE lotion when psoriasis is controlled. ULTRAVATE lotion should not be used for longer than 2 weeks. Advise patients to contact the physician if no improvement is seen within 2 weeks. Inform patients that total dosage should not exceed 50 grams per week [see Dosage and Administration (2)].
Instruct patients to avoid bandaging, wrapping or otherwise occluding the treatment area(s), unless directed by physician. Advise patients to avoid use on the face, scalp, groin, or axillae [see Dosage and Administration (2)].
Effects on Endocrine System
UL TRAVATE lotion may cause HPA axis suppression. Advise patients that use of UL TRAVATE lotion, may require periodic evaluation for HPA axis suppression. Advise patients to avoid use of multiple corticosteroid-containing products [see Warnings and Precautions (5.1)].
Local Adverse Reactions
Inform patients that topical corticosteroids may cause local adverse reactions, some of which may be irreversible. These reactions may be more likely to occur with occlusive use, prolonged use or use of higher potency comcosteroids, including ULTRAVATE lotion [see Warnings and Precautions (5.2)].
Breastteeding women should not apply ULTRAVATE lotion directly to the nipple and areola to avoid directly exposing the infant [see Lactation (8.2)].
| ULTRAVATE
halobetasol propionate lotion |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Lacer Pharma, LLC (117072266) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ferndale Laboratories, Inc. | 005320536 | analysis(73159-008) , manufacture(73159-008) , pack(73159-008) , label(73159-008) | |
Frequently asked questions
- Halobetasol vs. clobetasol - How do they compare?
- Halobetasol vs triamcinolone: which is better?
- Can halobetasol be used on the scalp?
- Can halobetasol propionate be bought over-the-counter (OTC)?
- Can halobetasol be used for poison ivy?
- Can halobetasol be used on the face / for acne?
- How long should you use Duobrii?
- What happens if you use too much Duobrii?
- Can you use adapalene and Duobrii together?
More about Halonate (halobetasol topical)
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: topical steroids
- Breastfeeding