Triptodur: Package Insert / Prescribing Info
Package insert / product label
Generic name: triptorelin
Dosage form: extended-release injection
Drug class: Gonadotropin releasing hormones
J Code (medical billing code): J3315 (3.75 mg, intramuscular)
Medically reviewed by Drugs.com. Last updated on May 16, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
- Medication Guide
Highlights of Prescribing Information
TRIPTODUR (triptorelin) for extended-release injectable suspension, for intramuscular use
Initial U.S. Approval: 2000
Indications and Usage for Triptodur
TRIPTODUR is a gonadotropin releasing hormone (GnRH) agonist indicated for the treatment of pediatric patients 2 years and older with central precocious puberty. ( 1)
Triptodur Dosage and Administration
- Must only be administered by a healthcare provider. ( 2.1)
- Administer TRIPTODUR as a single intramuscular injection of 22.5 mg once every 24 weeks. ( 2.1)
- Monitor response with LH levels after a GnRH or GnRH agonist stimulation test, basal LH, or serum concentration of sex steroid levels beginning 1 to 2 months following initiation of therapy, during therapy as necessary to confirm maintenance of efficacy, and with each subsequent dose. ( 2.2)
- Measure height every 3-6 months and monitor bone age periodically. ( 2.2)
- See FPI for complete reconstitution and administration instructions. ( 2.3)
Dosage Forms and Strengths
For extended-release injectable suspension: 22.5 mg of triptorelin as a powder cake for reconstitution with the co-packaged 2 mL of diluent Sterile Water for Injection. ( 3)
Warnings and Precautions
- Initial Rise of Gonadotropins and Sex Steroid Levels:An increase in clinical signs and symptoms of puberty may be observed during the first 2- 4 weeks of therapy since gonadotropins and sex steroids rise above baseline because of the initial stimulatory effect of the drug. ( 5.1)
- Psychiatric events:Have been reported in patients taking GnRH agonists. Events include emotional lability, such as crying, irritability, impatience, anger, and aggression. Monitor for development or worsening of psychiatric symptoms. ( 5.2)
- Convulsions:Have been observed in patients with or without a history of seizures, epilepsy, cerebrovascular disorders, central nervous system anomalies or tumors, and in patients on concomitant medications that have been associated with convulsions. ( 5.3)
- Pseudotumor Cerebri (Idiopathic Intracranial Hypertension):Have been reported in pediatric patients receiving GnRH agonists, including triptorelin. Monitor patients for headache, papilledema, and blurred vision. ( 5.4)
Adverse Reactions/Side Effects
In clinical trials for TRIPTODUR, the most common adverse reactions (≥4.5%) are injection site reactions, menstrual (vaginal) bleeding, hot flush, headache, cough, and infections (bronchitis, gastroenteritis, influenza, nasopharyngitis, otitis externa, pharyngitis, sinusitis, and upper respiratory tract infection). ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Azurity Pharmaceuticals, Inc. at 1-800-461-7449 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2023
Full Prescribing Information
1. Indications and Usage for Triptodur
TRIPTODUR is indicated for the treatment of pediatric patients 2 years of age and older with central precocious puberty (CPP).
2. Triptodur Dosage and Administration
2.1 Dosing Information
TRIPTODUR must only be administered by a healthcare provider.
The dosage of TRIPTODUR is 22.5 mg reconstituted with accompanying diluent (Sterile Water) 2 mL, and administered as a single intramuscular injection once every 24 weeks.
TRIPTODUR treatment should be discontinued at the appropriate age of onset of puberty at the discretion of the physician.
2.2 Monitoring
Monitor response to TRIPTODUR with LH levels after a GnRH or GnRH agonist stimulation test, basal LH, or serum concentration of sex steroid levels beginning 1 to 2 months following initiation of therapy, during therapy as necessary to confirm maintenance of efficacy, and with each subsequent dose.
Measure height (for calculation of growth rate) every 3-6 months and monitor bone age periodically.
Noncompliance with drug regimen or inadequate dosing may result in inadequate control of the pubertal process with gonadotropins and/or sex steroids increasing above prepubertal levels. If the dose of TRIPTODUR is not adequate switching to an alternative GnRH agonist for the treatment of CPP with the ability for dose adjustment may be necessary.
2.3 Reconstitution and Administration Instructions
Read these instructions completely before you begin.
• Triptodur suspension will sediment very quickly and should be injected immediately after reconstitution in accordance with the detailed instructions below.
• If the sequence of steps to prepare the suspension is interrupted and/or the vial is put aside, the suspension will start to separate into diluent and microgranules.
• To minimize the risk of needle blockage during the injection, ensure that the preparation of the injection is not interrupted and/or the mixed suspension syringe is not put aside because the suspension will sediment quickly.
1. Use appropriate aseptic technique for preparation and administration.
2. Screw the plunger rod into the barrel end of the prefilled sterile water diluent syringe.
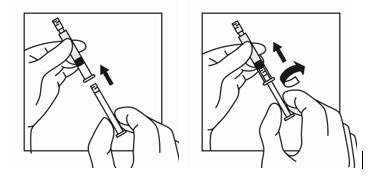
3. To remove the cap, twist counterclockwise to separate from the Luer lock on the syringe barrel.
4.
Firmly attach one of the 21-gauge sterile safety needles onto the prefilled sterile
water diluent syringe with a push and clockwise twist. This 21-gauge needle will only be used for reconstitution of the product.
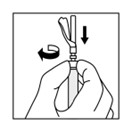
a. Remove the plastic Flip-off from the vial. Disinfect the visible part of the stopper
b. Pull back on the safety cover towards the syringe and away from the 21-gauge needle. Then pull the clear needle shield off.
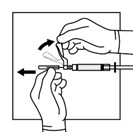
5. Insert the 21-gauge needle through the stopper. Inject the Sterile Water diluent into the vial, ensuring the diluent rinses the sides of the vial. Do not release the plunger rod.
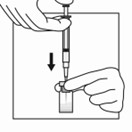
6. If the syringe plunger is not maintained in position, it will naturally withdraw product into the syringe. Thoroughly mix the vial with agitation for 30 to 60 seconds, ensuring the diluent rinses the sides of the vial.
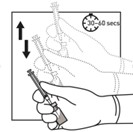
7. Before moving on to the next step, check visually that the suspension appears milky and homogeneous without any visible aggregates or precipitates.
a. If the suspension DOES NOT appear milky and homogeneous without any visible aggregates or precipitates, continue with the agitation. An up and down agitation can also be used to help eliminate aggregates or precipitates. The complete and homogeneous (milky) suspension of the product may require up to 60 seconds of agitation.
Important: Once mixed, proceed to the next steps and administer without delay.
8. The suspension will sediment very quickly so it is imperative to withdraw the suspension into the syringe directly after suspending the product in the vial.
9. Invert the vial and move back the syringe in order to position the end of the 21-gauge needle very near the level of the stopper, making sure the needle lumen is still completely in the vial.
10. Pull back the plunger rod slowly to withdraw the reconstituted product into the syringe, withdrawing as much of the reconstituted product into the syringe as possible. Move the tip of the needle at the level of the stopper so as to be able to withdraw a maximum amount of suspension.
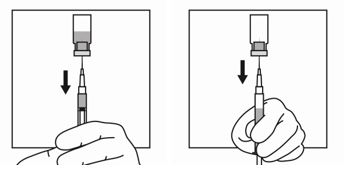
11. Withdraw the needle from the vial and push the safety cover forward toward the needle until you hear and/or feel it lock. Then remove the first 21-gauge needle by grasping the needle hub to disconnect the needle from the syringe and discard it.
This (first) 21-gauge needle will no longer be used.
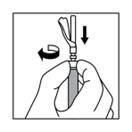
12.
Firmly attach the
second sterile needle
onto the syringe with a push and clockwise twist and pull back the safety cover towards the syringe. This 21-gauge needle will be used for administration. Triptodur must
only be administered with a thin-wall 21 -gauge needle.
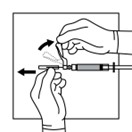
13. Do not prime the needle. Inspect the suspension visually for particulate matter and discoloration.
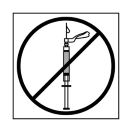
a. If the suspension does not appear milky and homogeneous, continue with an up and down agitation.
b. If the suspension appears milky and homogeneous without visible aggregates or precipitates, administer the suspension immediately.
14. Inject the patient intramuscularly, preferably in either buttock or thigh using the entire contents of the syringe. The injection of the suspension should be performed rapidly and in a steady and uninterrupted manner in order to avoid any potential blockage of the needle.
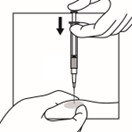
15. After administering the injection, immediately activate the safety cover:
a. Center your thumb or forefinger on the textured finger pad area of the safety cover and push it forward over the needle until you hear or feel it lock.
b. Use the one-handed technique and activate the mechanism away from yourself and others.
c. Immediately discard the syringe assembly into a suitable sharps container.
3. Dosage Forms and Strengths
For extended-release injectable suspension: 22.5 mg of triptorelin as a lyophilized white to slightly yellow powder cake in a single-dose vial for reconstitution with the co-packaged 2 mL of diluent (Sterile Water) for Injection.
4. Contraindications
- Hypersensitivity: TRIPTODUR is contraindicated in individuals with a known hypersensitivity to triptorelin, any other component of the product, or other GnRH agonists or GnRH [see Adverse Reactions (6.2)] .
- Pregnancy: TRIPTODUR may cause fetal harm [see Use in Specific Populations (8.1)] .
5. Warnings and Precautions
5.1 Initial Rise of Gonadotropins and Sex Steroid Levels
During the early phase of initial therapy or after subsequent doses, gonadotropins and sex steroids may rise above baseline because of a transient stimulatory effect of the drug [see Clinical Pharmacology (12.2)] . Therefore, a transient increase in clinical signs and symptoms of puberty, including vaginal bleeding, may be observed during the first weeks of therapy or after subsequent doses.
5.2 Psychiatric Events
Psychiatric events have been reported in patients taking GnRH agonists, including triptorelin. Post-marketing reports with this class of drugs include symptoms of emotional lability, such as crying, irritability, impatience, anger, and aggression. Monitor for development or worsening of psychiatric symptoms during treatment with TRIPTODUR [see Adverse Reactions (6)] .
5.3 Convulsions
Post-marketing reports of convulsions have been observed in patients receiving GnRH agonists, including triptorelin. These included patients with a history of seizures, epilepsy, cerebrovascular disorders, central nervous system anomalies or tumors, and patients on concomitant medications that have been associated with convulsions such as bupropion and SSRIs. Convulsions have also been reported in patients in the absence of any of the conditions mentioned above [ see Adverse Reactions (6)].
5.4 Pseudotumor Cerebri (Idiopathic Intracranial Hypertension)
Pseudotumor cerebri (idiopathic intracranial hypertension) has been reported in pediatric patients receiving GnRH agonists, including triptorelin. Monitor patients for signs and symptoms of pseudotumor cerebri, including headache, papilledema, blurred vision, diplopia, loss of vision, pain behind the eye or pain with eye movement, tinnitus, dizziness, and nausea.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are described here and elsewhere in the label:
- Initial Rise of Gonadotropins and Sex Steroid Levels [see Warnings and Precautions (5.1)]
- Psychiatric Events [see Warnings and Precautions (5.2)]
- Convulsions [see Warnings and Precautions (5.3)]
- Pseudotumor Cerebri (Idiopathic Intracranial Hypertension) [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of TRIPTODUR was evaluated in one uncontrolled, open-label single-arm clinical trial in which 44 children with central precocious puberty received two doses of TRIPTODUR and were observed for 12 months. The median age of the study population was 8 years (range 2-9 years) at treatment start; 88.6% of subjects were female, 59.1% were White, 27.3% were Black and 4.5% were Asian. Table 1 shows all the adverse reactions that occurred in at least 2 patients (≥4.5%) during the open-label single-arm trial.
| Adverse Reactions | Number of Patients Reporting Event (%)
(Total N=44) |
|---|---|
| Infections & Infestations | |
| Bronchitis | 2 (4.5) |
| Gastroenteritis | 3 (6.8) |
| Influenza | 2 (4.5) |
| Nasopharyngitis | 6 (13.6) |
| Otitis externa | 2 (4.5) |
| Pharyngitis | 2 (4.5) |
| Sinusitis | 2 (4.5) |
| Upper respiratory tract infection | 4 (9.1) |
| Nervous System Disorders | |
| Headache | 6 (13.6) |
| Reproductive System & Breast Disorders | |
| Menstrual (Vaginal bleeding) † | 3 (7.7) |
| Respiratory, Thoracic & Mediastinal Disorder | |
| Cough | 3 (6.8) |
| Vascular Disorders | |
| Hot flush | 2 (4.5) |
6.2 Post-marketing Experience
The following adverse reactions have been identified during post-approval use of triptorelin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity Reactions:Anaphylactic shock, anaphylactoid reaction, angioedema, urticaria.
Cardiovascular:Hypertension.
Psychiatric:Emotional lability, such as crying, irritability, impatience, anger, and aggression .Depression, including rare reports of suicidal ideation and attempt. Many, but not all, of these patients had a history of psychiatric illness or other comorbidities with an increased risk of depression.
Nervous System:Convulsions ,pseudotumor cerebri (idiopathic intracranial hypertension)
Vision Disorders:Visual impairment, visual disturbance
Related/similar drugs
7. Drug Interactions
7.1 Drug-Drug Interactions
Results of in vitro studies show that drug-drug interactions with triptorelin are unlikely [see Clinical Pharmacology (12.3)]. However, in the absence of relevant data and as a precaution, hyperprolactinemic drugs should not be used concomitantly with triptorelin since hyperprolactinemia reduces the number of pituitary GnRH receptors.
7.2 Drug-Laboratory Test Interactions
Administration of TRIPTODUR results in suppression of the pituitary-gonadal system.
The effect of TRIPTODUR on pituitary and gonadal function is expected to disappear within six to twelve months after treatment discontinuation. Therefore, diagnostic tests of pituitary gonadotropic and gonadal functions conducted during treatment or after discontinuation of treatment may be affected.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
TRIPTODUR is contraindicated in women who are pregnant [see Contraindications (4)] since expected hormonal changes that occur with TRIPTODUR treatment increase the risk for pregnancy loss. Available data with triptorelin use in pregnant women are insufficient to determine a drug-associated risk of adverse developmental outcomes. Based on mechanism of action in humans and findings of increased pregnancy loss in animal studies, TRIPTODUR may cause fetal harm when administered to pregnant women. Advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% - 4% and 15% -20%, respectively.
Data
Animal Data
In pregnant rats administered triptorelin at doses of 2, 10, and 100 mcg/kg/day during the period of organogenesis, maternal toxicity (decrease in body weight) and embryo-fetal toxicities (pre-implantation loss, increased resorption, and reduced number of viable fetuses) were observed at 100 mcg/kg, approximately 4 times the clinical dose based on body surface area. No embryonic and fetal developmental toxicities were observed in mice at doses up to 4 times the clinical dose. Teratogenic effects were not observed in viable fetuses in rats or mice.
8.2 Lactation
Risk Summary
There are no data on the presence of triptorelin in human milk, or the effects of the drug on the breastfed infant, or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for TRIPTODUR and any potential adverse effects on the breastfed child from TRIPTODUR or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of TRIPTODUR have been established in pediatric patients 2 years of age and older based on a single-arm open-label study of 44 children 2-9 years of age with CPP [see Clinical Studies (14)] . The safety and effectiveness of TRIPTODUR have not been established in pediatric patients less than 2 years old.
8.6 Renal Impairment
TRIPTODUR has not been studied in children with renal impairment. Adult subjects with renal impairment had higher exposure than young healthy adult males [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
TRIPTODUR has not been studied in children with hepatic impairment. Adult subjects with hepatic impairment had higher exposure than young healthy adult males [see Clinical Pharmacology (12.3)].
10. Overdosage
There is no experience with overdosage in clinical trials of triptorelin. If overdosage occurs, therapy should be discontinued and appropriate supportive and symptomatic treatment administered.
11. Triptodur Description
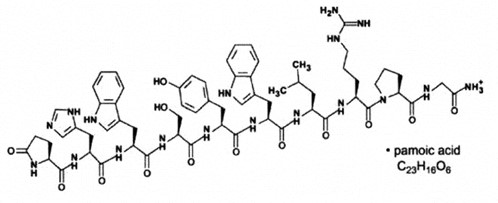
TRIPTODUR contains the pamoate salt of triptorelin, a synthetic decapeptide analog of naturally occurring gonadotropin-releasing hormone (GnRH or LHRH). The chemical name of triptorelin pamoate is 5-oxo-L-prolyl-L-histidyl-L-tryptophyl-L-seryl-L-tyrosyl-D-tryptophyl-L-leucyl-L-arginyl-L-prolylglycine amide (pamoate salt). The molecular weight is 1699.9 and the structural formula is:
TRIPTODUR for extended release injectable suspension for intramuscular use is provided as a sterile, lyophilized, biodegradable microgranule formulation in a single-dose vial, co-packaged with a syringe containing 2 mL Sterile Water for Injection for reconstitution of the lyophilisate. The triptorelin formulation is comprised of 22.5 mg triptorelin (equivalent to 31 mg triptorelin pamoate), carboxymethylcellulose sodium (26 mg), mannitol (74 mg), poly- d,l-lactide-co-glycolide (183 mg), and polysorbate 80 (1.7 mg). When 2 mL Sterile Water for Injection is added to the vial containing TRIPTODUR and mixed, a suspension is formed which is intended as a single intramuscular injection.
12. Triptodur - Clinical Pharmacology
12.2 Pharmacodynamics
Following the first administration, there is a transient surge in circulating levels of LH, FSH, testosterone, and estradiol [see Warnings and Precautions (5.2)] . After chronic and continuous administration, by 4 weeks after initiation of therapy, a sustained decrease in LH and FSH secretion and marked reduction in sex steroids are observed.
12.3 Pharmacokinetics
Absorption
After an initial intramuscular TRIPTODUR 22.5 mg injection and a second 22.5 mg intramuscular injection 24 weeks later in children 2 to 9 years old with CPP, triptorelin peaked 4 hours postdose with a geometric mean C maxof 39.9 and 36.5 ng/mL, respectively. No apparent accumulation of triptorelin occurred after the second injection. Absorption occurred in two phases, a burst phase followed by a maintenance release phase. In children with CPP, following the burst phase after the first 22.5 mg injection, geometric mean serum triptorelin levels were 0.11, 0.17, 0.05 and 0.03 ng/mL at Months 1, 2, 3, and 6, respectively.
Distribution
There is no evidence that triptorelin, at clinically relevant concentrations, binds to plasma proteins.
Elimination
Metabolism
The metabolism of triptorelin in humans is unknown, but is unlikely to involve hepatic microsomal enzymes (cytochrome P450). Thus far no metabolites of triptorelin have been identified. Pharmacokinetic data suggest that C-terminal fragments produced by tissue degradation are either completely degraded in the tissues, or rapidly degraded in plasma, or cleared by the kidneys.
Excretion
Triptorelin is eliminated by both the liver and the kidneys. Following intravenous administration of 0.5 mg triptorelin peptide to six healthy male volunteers with a creatinine clearance of 149.9 mL/min, 41.7% of the dose was excreted in urine as intact peptide with a total triptorelin clearance of 211.9 mL/min. This percentage increased to 62.3% in patients with liver disease who have a lower creatinine clearance (89.9 mL/min). It has also been observed that the nonrenal clearance of triptorelin (patient anuric, Cl creat= 0) was 76.2 mL/min, thus indicating that the nonrenal elimination of triptorelin is mainly dependent on the liver.
Specific Populations
Renal Impairment
After intravenous bolus injection of 0.5 mg triptorelin in adults, the two distribution half-lives were unaffected by renal impairment. However, renal insufficiency led to a decrease in total triptorelin clearance proportional to the decrease in creatinine clearance as well as increases in volume of distribution and consequently, an increase in the elimination half-life. Adult male subjects with moderate or severe renal impairment had approximately 2-fold higher exposure (AUC values) than young healthy adult males (see Table 1) [see Use in Specific Populations (8.6)].
Hepatic Impairment
After intravenous bolus injection of 0.5 mg triptorelin in adults, the two distribution half-lives were unaffected by hepatic impairment. In adult males with hepatic insufficiency, triptorelin clearance was reduced and exposure (AUC) was increased 3.7-fold compared to young healthy adult males (Table 2) [see Use in Specific Populations (8.7)] .
| Group | C
max
(ng/mL) | AUC
inf
(h∙ng/mL) | Cl
p
(mL/min) | Cl
renal
(mL/min) | t 1/2(h) | Cl
creat
(mL/min) |
|---|---|---|---|---|---|---|
| 6 healthy male volunteers | 48.2 ±11.8 | 36.1 ±5.8 | 211.9 ±31.6 | 90.6 ±35.3 | 2.81 ±1.21 | 149.9 ±7.3 |
| 6 males with moderate renal impairment | 45.6 ±20.5 | 69.9 ±24.6 | 120.0 ±45.0 | 23.3 ±17.6 | 6.56 ±1.25 | 39.7 ±22.5 |
| 6 males with severe renal impairment | 46.5 ±14.0 | 88.0 ±18.4 | 88.6 ±19.7 | 4.3 ±2.9 | 7.65 ±1.25 | 8.9 ±6.0 |
| 6 males with liver disease | 54.1 ±5.3 | 131.9 ±18.1 | 57.8 ±8.0 | 35.9 ±5.0 | 7.58 ±1.17 | 89.9 ±15.1 |
Drug-Drug Interactions
In Vitro Assessment of Drug Interactions
Drug Metabolizing Enzyme Inhibition
Triptorelin did not inhibit CYP1A2, 2B6, 2C8, 2C9, 2C19 or 2D6, or CYP 3A4/5 at clinically relevant concentrations.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis was evaluated in an 18-month study in mice and a 24-month study in rats. In rats, triptorelin doses of 120, 600, and 3000 mcg/kg given every 28 days (approximately 0.2, 0.8, and 4 times the estimated human monthly dose based on body surface area) resulted in increased mortality with a drug treatment period of 13 to 19 months. The incidences of benign and malignant pituitary tumors and histosarcomas were increased in a dose-related manner. There were no treatment-related tumors in mice at exposure up to 4-fold higher than the estimated human monthly dose based on body surface area.
Mutagenicity studies performed with triptorelin using bacterial and mammalian systems ( in vitroAmes test and chromosomal aberration test in CHO cells and an in vivomouse micronucleus test) provided no evidence of mutagenic potential.
After 60 days of subcutaneous treatment followed by a minimum of four estrus cycles prior to mating, triptorelin at doses of 2, 20, and 200 mcg/kg (approximately 0.07, 0.7, and 7 times the estimated human daily dose based on body surface area) or two monthly injections as slow release microspheres (~20 mcg/kg/day) had no effect on the fertility or general reproductive function of female rats.
No studies were conducted to assess the effect of triptorelin on male fertility.
14. Clinical Studies
In a single-arm open-label study, 44 children 2 to 9 years of age with CPP, 39 females and 5 males, all naïve to previous GnRH agonist treatment, were administered TRIPTODUR 22.5 mg at a dosing interval of 24 weeks. Subjects were evaluated over two dosing intervals for a total of 12 months.
TRIPTODUR 22.5 mg suppressed pituitary release of LH and FSH and, consequently, gonadal secretion of estradiol in girls and testosterone in boys (Table 3). At all timepoints evaluated, ≥93% of children achieved LH suppression to prepubertal levels (i.e., serum LH ≤5 IU/L 30 minutes after GnRH agonist stimulation), ≥79% of girls achieved prepubertal levels of estradiol (i.e., <20 pg/mL), and ≥80% of boys achieved prepubertal levels of testosterone (i.e., <30 ng/dL). TRIPTODUR arrested or reversed progression of clinical signs of puberty with 95% of children showing no increase in the bone age/chronological age ratio, and 89% showing stabilization of sexual maturation at Month 12.
| Endpoint | % (n/N) of Children Achieving Endpoint | |||||
|---|---|---|---|---|---|---|
| Month 1 | Month 2 | Month 3 | Month 6 | Month 9 | Month 12 | |
| % with prepubertal LH (GnRH-stim LH ≤5 IU/L) | 95%
(42/44) | 95%
(42/44) | 95%
(42/44) | 93%
†
(41/44) | 95%
(42/44) | 98%
(43/44) |
| % girls with prepubertal estradiol (<20 pg/mL) | 87%
(34/39) | 89%
(34/38) | 92%
(36/39) | 79%
(31/39) | 82%
(32/39) | 79%
(31/39) |
| % boys with prepubertal testosterone (<30 ng/dL) | 80%
(4/5) | 80%
(4/5) | 100%
(5/5) | 100%
(5/5) | 80%
(4/5) | 80%
(4/5) |
| % with no increase in BA/CA ‡ratio vs. baseline | 64%
(28/44) | 95%
(42/44) |
||||
| % achieving stabilization of sexual maturation | 91%
(40/44) | 89%
(39/44) |
||||
| % girls with regression of uterine length | 69%
(27/39) | 77%
(30/39) |
||||
| % boys with no progression in testis volumes | 100%
(5/5) | 100%
(5/5) |
||||
Following the second TRIPTODUR injection, 22 children (all girls) were assessed for evidence of an acute-on-chronic phenomenon (i.e., increase in basal LH >5 IU/L or serum estradiol level >20 pg/mL 48 hours after the second triptorelin injection). Of these, one girl who achieved prepubertal hormone levels prior to the second injection showed biochemical evidence of acute-on-chronic phenomenon [see Warnings and Precautions (5.2)and Adverse Reactions (6.1)] .
16. How is Triptodur supplied
Each TRIPTODUR 22.5 mg single-use kit (NDC 24338-150-20) contains:
- One single-dose vial of TRIPTODUR 22.5 mg (NDC 24338-150-01) with a Flip-Off seal containing sterile lyophilized white to slightly yellow powder cake
- One sterile, glass syringe with Luer Lock prefilled with 2 mL of Sterile Water for Injection (NDC 24338-150-02)
- Two sterile 21 gauge, 1½" needles ( thin-wall) with safety cover
- One Package Insert
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Medication Guide).
Hypersensitivity Reactions
Inform caregivers that anaphylactic shock, hypersensitivity, and angioedema have been reported with triptorelin use and to immediately seek medical attention if any hypersensitivity reaction occurs.
Symptoms after Initial TRIPTODUR Administration
Inform caregivers that during the first weeks after the first TRIPTODUR injection, signs of puberty may occur such as vaginal bleeding [see Warnings and Precautions (5.1)and Adverse Reactions (6.1)] . Caregivers should notify the physician if these symptoms continue beyond the second month after TRIPTODUR administration.
Psychiatric Events
Inform caregivers that symptoms of emotional lability, such as crying, irritability, impatience, anger, and aggression have been observed in patients receiving GnRH agonists, including triptorelin. Alert caregivers to the possibility of development or worsening of psychiatric symptoms, including depression, during treatment with TRIPTODUR [see Warnings and Precautions (5.2)and Adverse Reactions (6.2)].
Convulsions
Inform caregivers that reports of convulsions have been observed in patients receiving GnRH agonists, including triptorelin. Patients with a history of seizures, epilepsy, cerebrovascular disorders, central nervous system anomalies or tumors, and patients on concomitant medications that have been associated with convulsions may be at increased risk [see Warnings and Precautions (5.3)].
Pseudotumor Cerebri (Idiopathic Intracranial Hypertension)
Inform patients and caregivers that reports of pseudotumor cerebri (idiopathic intracranial hypertension) have been observed in pediatric patients receiving GnRH agonists, including triptorelin. Monitor patients for signs and symptoms of pseudotumor cerebri, including headache, and vision issues such as blurred vision, double vision, loss of vision, pain behind the eye or pain with eye movement, ringing in the ears, dizziness, and nausea. Advise patients and caregivers to contact their healthcare provider if the patient develops any of these symptoms. [see Warnings and Precautions (5.4)] .
Pregnancy is Contraindicated
TRIPTODUR is contraindicated in pregnancy. If the patient becomes pregnant while taking the drug, the patient should be informed of the potential risk to fetus [see Use in Specific Populations (8.1)].
Manufactured for:
Azurity Pharmaceuticals, Inc.
Woburn, MA 01801
Manufactured by:
Debiopharm Research & Manufacturing SA
CH-1920 Martigny, Switzerland
TRIPTODUR is a registered trademark of Debiopharm International SA.
Patent: https://azurity.com/patents_and_trademarks/
This product's labeling may have been updated. For current Full Prescribing Information, please visit www.triptodur.com
TRIP-PI-08 Rev. NOV 2023
| MEDICATION GUIDE
TRIPTODUR ®[TRIP-toe-der] (triptorelin) for extended-release injectable suspension, for intramuscular use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Rev: 12/2022 | ||
What is the most important information I should know about TRIPTODUR?
Call your child's doctor right away if your child has a seizure while taking TRIPTODUR. Increased pressure in the fluid around the brain can happen in children taking GnRH agonists medicines including TRIPTODUR.
|
|||
|
| ||
What is TRIPTODUR?
|
|||
TRIPTODUR should not be taken if your child is:
|
|||
|
|
|
|
|
|||
Before your child receives TRIPTODUR, tell your child's doctor about all of your child's medical conditions, including if they:
|
|||
How will your child receive TRIPTODUR?
|
|||
| What are the possible side effects of TRIPTODUR?
TRIPTODUR may cause serious side effects. See " What is the most important information I should know about TRIPTODUR?" The most common side effects of TRIPTODUR include injection site reactions, menstrual (vaginal) bleeding, hot flush, headache, cough, and infections (bronchitis, gastroenteritis, influenza, nasopharyngitis, otitis externa, pharyngitis, sinusitis, and upper respiratory tract infection) . These are not all the possible side effects of TRIPTODUR. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
| What are the ingredients in TRIPTODUR?
Active ingredient: triptorelin Inactive ingredients:carboxymethylcellulose sodium, mannitol, poly- d,l-lactide-co-glycolide, and polysorbate 80 Manufactured for: Azurity Pharmaceuticals, Inc., Woburn, MA 01801 Manufactured by: Debiopharm Research & Manufacturing SA, CH-1920 Martigny, Switzerland For more information about TRIPTODUR, please contact Azurity Pharmaceuticals, Inc. at 1-800-461-7449. |
|||
TRIPTODUR is a registered trademark of Debiopharm International SA.
TRIP-MG-06 Rev. 04/2023
PRINCIPAL DISPLAY PANEL - Kit Carton
NDC 24338-150-20
Rx Only
Triptodur
®
(triptorelin)
for extended-release injectable suspension
22.5 mg
KIT
22.5 mg every 24 weeks
FOR INTRAMUSCULAR USE
Must only be administered by
a healthcare professional.
Give once every 24 weeks.
Single-Dose Delivery System
See package insert for full prescribing information.
Contents:
–One Single-Dose Vial
–One Pre-filled Syringe of Diluent (sterile water)
for Triptodur, 2 mL
–Two thin-walled, 21-gauge, 1½" Needles
Reconstitute With Accompanying Diluent Before Use.
After reconstitution, ensure there are no visible
precipitates in the vial and administer immediately.
Dispense the accompanying
Medication Guide to each patient.
azurity
®
pharmaceuticals

| TRIPTODUR
triptorelin kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Azurity Pharmaceuticals, Inc. (117505635) |
Frequently asked questions
- What is Triptodur used to treat?
- How long can I take Trelstar (triptorelin) for prostate cancer?
- Why is triptorelin used for prostate cancer?
More about Triptodur (triptorelin)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Drug images
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: gonadotropin releasing hormones
- En español