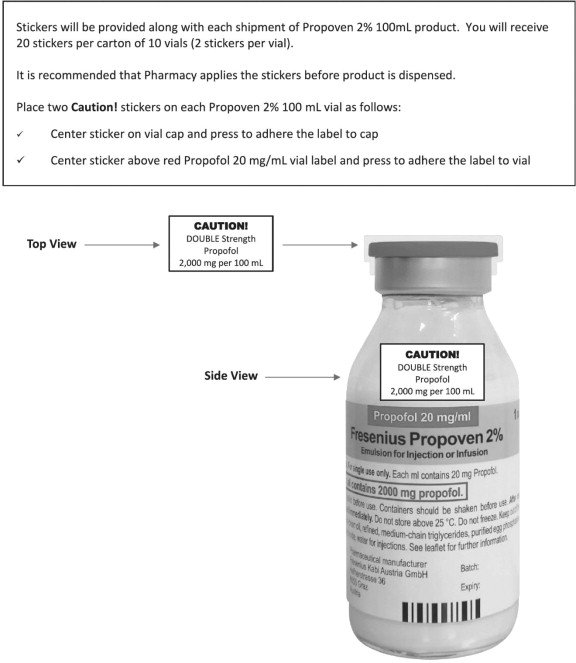
Propoven: Package Insert / Prescribing Info
Package insert / product label
Generic name: propofol
Dosage form: injection, emulsion
Drug class: General anesthetics
Medically reviewed by Drugs.com. Last updated on Jun 16, 2025.
EMERGENCY USE AUTHORIZATION (EUA) OF FRESENIUS
PROPOVEN 2% EMULSION
The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of the unapproved product, Fresenius Propoven 2% (propofol 20 mg/mL) Emulsion 100 mL, to maintain sedation via continuous infusion in patients greater than 16 years old who require mechanical ventilation in an intensive care unit (ICU) setting.
CAUTION: THERE IS A RISK OF UNINTENTIONAL OVERDOSE WITH THIS UNAPPROVED PRODUCT.
Fresenius Propoven 2% Emulsion contains the same active ingredient, propofol, as FDA-approved Diprivan Injectable Emulsion USP 10 mg/mL, but contains double the concentration. BECAUSE OF THIS DIFFERENCE IN CONCENTRATION BETWEEN THIS UNAPPROVED PRODUCT AND THE FDA-APPROVED PRODUCT, THERE IS A RISK OF UNINTENTIONAL OVERDOSE.
| NOTE IMPORTANT DIFFERENCES IN FORMULATION AND LABELING BETWEEN THE CURRENT U.S. MARKETED FDA-APPROVED DIPRIVAN INJECTABLE EMULSION, USP 10 MG/ML ® (DIPRIVAN®) PRODUCTS AND FRESENIUS PROPOVEN 2% EMULSION. SEE DETAILS BELOW BEFORE ADMINISTERING FRESENIUS PROPOVEN 2% EMULSION. |
Fresenius Propoven 2% Emulsion 100mL is not an FDA-approved drug in the United States. However, FDA has issued an EUA permitting the emergency use of Fresenius Propoven 2% Emulsion during the COVID-19 pandemic.
The scope of the EUA is limited as follows:
- Fresenius Propoven 2% Emulsion will be used only to maintain sedation via continuous infusion in patients greater than 16 years old who require mechanical ventilation.
- Fresenius Propoven 2% Emulsion will be administered only by a licensed healthcare provider in an ICU setting.
- Fresenius Propoven 2% Emulsion will NOT be administered to pregnant women, unless there are no FDA-approved products available to maintain sedation for these patients should they require mechanical ventilation in an ICU setting.
- Fresenius Propoven 2% Emulsion will be used only in accordance with the dosing regimens as detailed in the authorized Fact Sheets.
Product Description
Consistent with the EUA, Fresenius Kabi USA will offer the following presentation of Fresenius Propoven 2% (propofol 20 mg/mL) Emulsion:
|
*NOTE: This propofol 20 mg/mL product contains double the concentration of propofol compared to the FDA-approved and marketed Diprivan (propofol) Injectable Emulsion, USP 10 mg/mL product. |
|||
| Product Name And Description | MCT/LCT Concentration | Source/ Type of Oil | Size |
| Fresenius Propoven 2% Propofol Emulsion for Continuous Infusion 2,000 mg/100 mL (20 mg/mL) | Medium Chain Triglycerides (MCT) 50 mg/mL Long Chain Triglycerides (LCT) 50 mg/mL | soybean oil, refined; medium-chain triglycerides |
100 mL |
Fresenius Propoven 2% Emulsion 50 mL is approved in Europe and other international countries. The Fresenius Propoven 2% Emulsion 100mL product will be manufactured by Fresenius Kabi AG in the same FDA inspected facilities as DIPRIVAN® and other Propoven 2% fill sizes.
PLEASE READ IMPORTANT MESSAGE
|
Diprivan Injectable Emulsion, USP 10 mg/mL |
Fresenius Propoven 2% (propofol 20 mg/mL) Emulsion |
What does this mean to you as a healthcare professional? |
|
|
Concentration |
Contains propofol 10 mg/mL |
Contains propofol 20 mg/mL | THERE IS A RISK OF UNINTENTIONAL OVERDOSE WITH THIS UNAPPROVED
PRODUCT. Fresenius Propoven 2% (propofol 20 mg/mL) is DOUBLE THE CONCENTRATION of US marketed Diprivan 10 mg/mL (Diprivan 1%). Exercise caution and implement steps to ensure dosing calculations, infusion rates, and infusion pump settings are accurate. Addition of the new concentration (20 mg/mL) to the drug library of the respective pumps and to electronic health records (EHR) must be implemented. |
|
Indication |
General anesthesia, procedural sedation, ICU sedation |
ICU sedation ONLY | Fresenius Propoven 2% is only to maintain sedation via continuous infusion in patients greater than 16 years old who require mechanical ventilation in the ICU setting. |
|
Patient Population |
Greater than 3 years old (procedural sedation and general anesthesia) Greater than 16 years old (ICU sedation) |
Greater than 16 years old (ICU sedation) | Fresenius Propoven 2% is only indicated to maintain sedation via continuous infusion for patients greater than 16 years old who require mechanical ventilation in the ICU setting. Fresenius Propoven 2% should not be used in pregnant women unless there are no FDA-approved products available to maintain sedation for these patients who require mechanical ventilation in an ICU setting. |
|
Composition |
Contains long-chain triglycerides (LCT) |
Contains a combination of medium-chain triglycerides (MCT) and long-chain triglycerides (LCT) | Prolonged IV infusion of MCT to pregnant rabbits has been reported in the published literature to increase the RISK OF NEURAL TUBE DEFECTS.
Because it is not yet clear if there is differential risk for adverse developmental effects with Fresenius Propoven 2% compared to Diprivan 1%, Fresenius Propoven 2% SHOULD NOT BE USED IN PREGNANT WOMEN unless there are no FDA-approved products |
| available to maintain sedation in these patients who require mechanical ventilation in an ICU setting. | |||
|
Dosing |
See package insert | Administration rates of 0.3 to 4.0 mg propofol/kg bodyweight/h have been demonstrated to provide adequate sedation |
Infusion rates greater than 4.0 mg propofol/kg bodyweight/h are not recommended due to risk of Propofol Infusion Syndrome. The duration of administration must not exceed 7 days. |
|
Method of administration |
Bolus or infusion |
Infusion ONLY | Fresenius Propoven 2% Emulsion is administered undiluted intravenously by continuous infusion. Containers should be shaken before use. If two layers can be seen after shaking the emulsion should not be used. Do not admix with other medicinal products. Co-administration of other medicinal products or fluids added to the Fresenius Propoven 2% Emulsion infusion line must occur close to the cannula site using a Y-piece connector or a three-way valve. Fresenius Propoven 2% Emulsion must not be administered via a microbiological filter. |
|
Other Special Patient Populations |
See package insert |
Caution should be taken when treating patients with mitochondrial disease, epilepsy, and disorders of fat metabolism. | Patients with mitochondrial disease may be susceptible to exacerbations of their disorder when undergoing ICU care. Maintenance of normothermia, provision of carbohydrates and good hydration are recommended for such patients. The early presentations of mitochondrial disease exacerbation and of the ‘propofol infusion syndrome’ may be similar. Although several studies have demonstrated efficacy in treating status epilepticus, administration of propofol in epileptic patients may also increase the risk of seizure. For these patients, as well as for ARDS/respiratory failure and tetanus patients, sedation maintenance dosages were generally higher than those for other critically ill patient populations. Appropriate care should be applied in patients with disorders of fat metabolism and in other conditions where lipid emulsions must be used cautiously. It is recommended that blood lipid levels should be monitored if propofol is administered to patients |
| thought to be at particular risk of fat overload. Administration of propofol should be adjusted appropriately if the monitoring indicates that fat is being inadequately cleared from the body. If the patient is receiving other intravenous lipid concurrently, a reduction in quantity should be made in order to take account of the amount of lipid infused as part of the propofol formulation; 1.0 mL of Fresenius Propoven 2% Emulsion contains approximately 0.1 g of fat. | |||
|
Drug Interactions |
See package insert |
Drug interaction with fentanyl, cyclosporine and valproate | After administration of fentanyl, the blood level of propofol may be temporarily increased with an increase in the rate of apnea. Leukoencephalopathy has been reported with administration of lipid emulsions as used for Fresenius Propoven 2% Emulsion in patients receiving cyclosporine. A need for lower propofol doses has been observed in patients taking valproate. When used concomitantly, a dose reduction of propofol may be considered. |
|
Presence of antimicrobial retardant |
Yes |
NO | Fresenius Propoven 2% Emulsion does NOT contain an anti-microbial retardant such as ethylenediaminetetraacetic acid (EDTA), sodium metabisulfite, or benzyl alcohol/sodium benzoate. STRICT ASEPTIC TECHNIQUE MUST ALWAYS BE MAINTAINED DURING HANDLING. Each vial of Fresenius Propoven 2% Emulsion is intended only for single administration for an individual patient. Vials are not intended for multiple use. If propofol is to be aspirated, it must be drawn aseptically into a sterile syringe immediately after breaking the vial seal. Administration must commence without delay. The unused portion of a vial should be discarded immediately after opening. As with any propofol used in IV infusion, discard all product and infusion lines after 12 hours. |
|
Contraindications |
See package insert | Fresenius Propoven 2% Emulsion should not be used in patients who | Fresenius Propoven 2% Emulsion is contraindicated in patients with a known hypersensitivity to propofol or |
| are hypersensitive to peanut or soy. | any of the excipients: Soybean oil; Medium-chain triglycerides; Purified egg phosphatides; Glycerol; Oleic acid; Sodium hydroxide; Water for injections Fresenius Propoven 2% Emulsion should not be used in patients who are hypersensitive to peanut or soy. | ||
|
Bar code |
Unit of use barcode on individual vials |
No unit of use barcode | The barcode used on Fresenius Propoven 2% Emulsion may not register accurately on U.S. scanning systems. Institutions should manually input the product into their systems to confirm that barcode systems do not provide incorrect information when the product is scanned. Alternative procedures should be followed to assure that the correct drug product is being used and administered to individual patients. |
| For questions regarding Fresenius Propoven 2% Emulsion in the United States, please contact Fresenius Kabi USA Medical Affairs at 1-800-551-7176 Option 3, Monday – Friday, between the hours of 8 a.m. and 5 p.m. (CST), or e-mail medinfo.USA@fresenius-kabi.com. |
|||
What is an EUA?
The United States FDA has made the Fresenius Propoven 2% Emulsion available to treat patients in an ICU during the COVID-19 pandemic under an emergency access mechanism called an Emergency Use Authorization (EUA). The EUA is supported by a Secretary of Health and Human Service (HHS) declaration that circumstances exist to justify the emergency use of drugs and biological products during the COVID-19 pandemic.
Fresenius Propoven 2% Emulsion made available under an EUA have not undergone the same type of review as an FDA-approved product. FDA may issue an EUA when certain criteria are met, which includes that there are no adequate, approved, available alternatives. Based on the totality of scientific evidence available, it is reasonable to believe that the Fresenius Propoven 2% Emulsion has met certain criteria for safety, performance, and labeling and may be effective to maintain sedation via continuous infusion in patients greater than 16 years old who require mechanical ventilation in an ICU setting.
This EUA for Fresenius Propoven 2% Emulsion is in effect for the duration of the COVID-19 declaration justifying emergency use of the products, unless terminated or revoked (after which the products may no longer be needed). The EUA will end when the declaration is terminated or revoked or when there is a change in the approval status of the product such that an EUA is no longer needed.
This communication and product information is available on the Fresenius Kabi USA website https://www.fresenius-kabi.com/us/pharmaceutical-product-updates as well as the FDA webpage which includes links to patient fact sheet and manufacturer's instructions https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy- framework/emergency-use-authorization#covidtherapeutics.
Adverse Event Reporting
Report adverse events or quality problems experienced with the use of this product. Healthcare facilities and prescribing health care providers or their designee receiving Fresenius
Propoven 2% Emulsion will track all medication errors associated with the use of and all serious
adverse events that are considered to be potentially attributable to Fresenius Propoven 2% Emulsion use and must report these to FDA using one of the following methods:
- Complete and submit a MedWatch form online (www.fda.gov/medwatch/report.htm)
- Complete and submit FDA Form 3500 (health professional) by fax (1-800-FDA-0178) (this form can be found via link above).
Call 1-800-FDA-1088 for questions. Submitted reports should state, “use of Fresenius Propoven 2% Emulsion was under an EUA” at the beginning of the question “Describe Event” for further analysis.
Fresenius Kabi USA CONTACT NUMBERS: Please use the following contact numbers as appropriate:
|
*Call Fresenius Kabi USA Vigilance or Medical Affairs Monday – Friday, between the hours of |
||
| Reason To Call | Department | Number |
| ADE Reporting | Vigilance Department |
1-800-551-7176* |
| Clinical/Technical Info. Or Product Complaint | Medical Affairs Department | |
| Product Availability & Ordering | Customer Service Department | 1-888-386-1300 |
Fact Sheet for Patients And Parent/Caregivers
Emergency Use Authorization (EUA) of FRESENIUS PROPOVEN 2% (Propofol) Emulsion during Coronavirus Disease 2019 (COVID-19) Pandemic
Fresenius Propoven 2% Emulsion is used for sedation of patients who are on a machine that helps with breathing (ventilator). Fresenius Propoven 2% Emulsion is given through a vein by intravenous (IV) infusion in patients 16 years of age and older in the intensive care unit (ICU). Fresenius Propoven 2% Emulsion should be not be used in pregnant women unless there are no other FDA-approved medicines available for ICU sedation. This fact sheet contains information to help you understand the risks and benefits of Fresenius Propoven 2% Emulsion you have received or may receive.
There is currently a shortage of U.S. Food and Drug Administration (FDA)-approved propofol products that maintain sedation for patients who are on a machine that helps with breathing (ventilator) due to the COVID-19 pandemic. Fresenius Propoven 2% Emulsion is not an FDA-approved medicine in the United States. Fresenius Propoven 2% Emulsion contains the same active ingredient, propofol, as Diprivan Injectable Emulsion and other propofol products approved in the United States, but contains double the concentration. Fresenius Propoven 2% Emulsion is currently approved in Europe and other international countries. Read this Fact Sheet for information about Fresenius Propoven 2% Emulsion.
Talk to your healthcare provider if you have questions. It is your choice to take Fresenius Propoven 2% Emulsion or stop it at any time.
What is COVID-19?
COVID-19 is caused by a virus called a coronavirus. This type of coronavirus has not been seen before. This new coronavirus was first found in people in December 2019. You can get COVID-19 through contact with another person who has the virus.
COVID-19 illnesses have ranged from very mild (including some with no reported symptoms) to severe, including illness resulting in death. While information so far suggests that most COVID- 19 illness is mild, serious illness can happen and may cause some of your other medical conditions to become worse. Older people and people of all ages with severe, long lasting (chronic) medical conditions like heart disease, lung disease and diabetes, for example seem to be at higher risk of being hospitalized for COVID-19.
The symptoms of COVID-19 are fever, cough and shortness of breath, which may appear
2 to 14 days after exposure. Serious illness including breathing problems can occur and may cause your other medical conditions to become worse.
What is Fresenius Propoven 2% Emulsion?
Fresenius Propoven 2% Emulsion is the brand name for propofol which belongs to a group of medicines called sedative/hypnotics. It will be used to help calm (sedate) you if you need a tube inserted (intubation) and a machine to help you breathe (ventilator) while in an ICU.
What should I tell my healthcare provider before I receive Fresenius Propoven 2% Emulsion?
Tell your healthcare provider about all of your medical conditions, including if you:
- Have any allergies
-
Have kidney or liver problems
PLEASE READ
IMPORTANT MESSAGE -
Are pregnant or plan to become pregnant. DUE TO POSSIBILITY OF BIRTH DEFECTS, FRESENIUS PROPOVEN 2% EMULSION SHOULD BE USED IN PREGNANT WOMEN ONLY IF THERE ARE NO OTHER FDA-APPROVED MEDICINES AVAILABLE FOR YOUR MEDICAL CONDITION.
- Are breastfeeding or plan to breastfeed
- Are taking any medicines including prescription, over-the-counter, vitamins, or herbal products.
Who should not receive Fresenius Propoven 2% Emulsion?
Do not receive Fresenius Propoven 2% Emulsion if:
- You have ever received propofol before and have had an allergic reaction to it
- You have allergies to peanut or soy
- You are 16 years of age or younger
How will I receive Fresenius Propoven 2% Emulsion?
Fresenius Propoven 2% Emulsion is given to you through a vein (IV) under the direct supervision of an anesthesiologist or intensive care doctor who will closely control the amount of Fresenius Propoven 2% Emulsion given to you.
What are the important possible side effects of Fresenius Propoven 2% Emulsion?
The most common side effects are:
- Drop in blood pressure
- Changes in heart rate
- Hot flushes
- Changes in breathing, coughing and hiccups
Less common side effects are:
- Allergic reactions, for example swelling of the throat, difficulty breathing, reddening of the skin
- Inflammation of the pancreas
- Fluid in the lungs
- Skin or tissue damage if given or leaks outside of the vein
- Muscle damage
- Heart failure
- During recovery period, coughing or nausea, headache, dizziness, shivering, fever, epileptic movements, spasms, irregular heartbeat
- Change in color of your urine
What other treatment choices are there?
Your anesthesiologist or intensive care doctor may give you other sedation agents depending on your medical condition.
What if I am pregnant or breastfeeding?
It is not known if Fresenius Propoven 2% Emulsion is safe in pregnant women. It may harm your unborn baby. If you receive Fresenius Propoven 2% Emulsion during pregnancy, your baby may be at risk for birth defects that affect the brain or spinal cord (neural tube defects). Fresenius Propoven 2% Emulsion should be used in pregnant women only if there are no other FDA-approved medicines available for your medical condition. Tell your doctor if you are pregnant or think that you may be pregnant.
Fresenius Propoven 2% Emulsion may pass into your breast milk. You should stop breastfeeding and throw away (discard) breast milk for 24 hours after receiving Fresenius Propoven 2% Emulsion.
How do I report side effects with Fresenius Propoven 2% Emulsion?
Tell your healthcare provider right away if you have any side effect that bothers you or does not go away.
Report side effects to FDA MedWatch at www.fda.gov/medwatch or call 1-800-FDA-1088. You may also report the problem to Fresenius Kabi USA at 1-800-551-7176, Monday – Friday, between the hours of 8 a.m. and 5 p.m. (CST).
How can I learn more about COVID-19?
- Ask your healthcare provider
- Visit https://www.cdc.gov/COVID19
- Contact your local or state public health department
What is an Emergency Use Authorization (EUA)?
The United States FDA has made Fresenius Propoven 2% Emulsion available under an emergency access mechanism called an EUA. The EUA is supported by a Secretary of Health and Human Service (HHS) declaration that circumstances exist to justify the emergency use of drugs and biological products during the COVID-19 pandemic.
Fresenius Propoven 2% Emulsion has not undergone the same type of review as an FDA-approved product. FDA may issue an EUA when certain criteria are met, which includes that there are no adequate, approved, and available alternatives. In addition, the FDA decision is based on the totality of scientific evidence available showing that it is reasonable to believe that the product meets certain criteria for safety, performance, and labeling and may be effective in treatment of patients during the COVID-19 pandemic. All of these criteria must be met to allow for the product to be used in the treatment of patients during the COVID-19 pandemic.
The EUA for Fresenius Propoven 2% Emulsion is in effect for the duration of the COVID-19 declaration justifying emergency use of these products, unless terminated or revoked (after which the products may no longer be used).
| FRESENIUS PROPOVEN 2%
propofol emulsion |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Fresenius Kabi USA, LLC (013547657) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Fresenius Kabi Austria | 300206604 | ANALYSIS(65219-427) , MANUFACTURE(65219-427) , PACK(65219-427) | |
More about Propoven (propofol)
- Check interactions
- Compare alternatives
- Latest FDA alerts (10)
- Side effects
- Dosage information
- During pregnancy
- Drug class: general anesthetics
- Breastfeeding
- En español