Doxycycline Oral Suspension: Package Insert / Prescribing Info
Package insert / product label
Generic name: doxycycline monohydrate
Dosage form: powder, for oral suspension
Drug classes: Miscellaneous antimalarials, Tetracyclines
Medically reviewed by Drugs.com. Last updated on Jan 6, 2025.
On This Page
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Doxycycline and other antibacterial drugs, doxycycline should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
Doxycycline Oral Suspension Description
Doxycycline is an antibacterial drug synthetically derived from oxytetracycline, and is available as Doxycycline Hyclate Tablets and Capsules and Doxycycline for Oral Suspension; for oral administration.
The structural formula of doxycycline monohydrate is
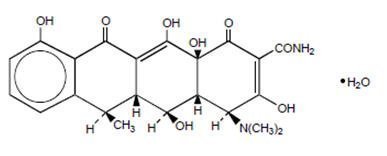
with a molecular formula of C22H24N2O8∙H2O and a molecular weight of 462.46. The chemical designation for doxycycline is 4-(Dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide monohydrate. The molecular formula for doxycycline hydrochloride hemiethanolate hemihydrate is (C22H24N2O8∙HCl)2∙C2H6O∙H2O and the molecular weight is 1025.89. Doxycycline is a light-yellow crystalline powder. Doxycycline hyclate is soluble in water, while doxycycline monohydrate is very slightly soluble in water.
Doxycycline has a high degree of lipoid solubility and a low affinity for calcium binding. It is highly stable in normal human serum. Doxycycline will not degrade into an epianhydro form.
Inactive ingredients in the capsule formulations are: colloidal silicon dioxide, lactose anhydrous, magnesium stearate, methylcellulose, microcrystalline cellulose, polyethylene glycol, sodium starch glycolate, and stearic acid. 50 mg gelatin capsule shell contains: FD&C Blue #1, D&C Yellow #10, titanium dioxide and gelatin. 100 mg gelatin capsule shell contains: FD&C Blue #1, titanium dioxide and gelatin. Black ink contains shellac glaze in SD-45, black iron oxide, propylene glycol, FD&C Blue #2, FD&C Red # 40, FD&C Blue # 1, D&C Yellow # 10, butyl alcohol, denatured alcohol. Each capsule, for oral administration, contains doxycycline hyclate equivalent to 50 mg or 100 mg of doxycycline.
Inactive ingredients for the oral suspension formulation are: confectioner's sugar, D&C red #27 aluminum lake, ethyl acetate, maltodextrin, methylparaben, microcrystalline cellulose/ sodium carboxymethylcellulose, modified cornstarch, natural flavoring; propylparaben, simethicone and sucrose. When reconstituted, each teaspoonful (5 mL) doxycycline suspension, for oral administration, contains doxycycline monohydrate equivalent to 25 mg of doxycycline.
Inactive ingredients for the tablet formulation are: anhydrous lactose, colloidal silicon dioxide, FD&C Red No. 40, FD&C Yellow No. 6, hypromellose, magnesium stearate, methylcellulose, microcrystalline cellulose, polyethylene glycol, sodium starch glycolate, stearic acid, and titanium dioxide. Each tablet, for oral administration, contains doxycycline hyclate equivalent to 100 mg doxycycline.
Doxycycline Oral Suspension - Clinical Pharmacology
Tetracyclines are readily absorbed and are bound to plasma proteins in varying degree. They are concentrated by the liver in the bile, and excreted in the urine and feces at high concentrations and in a biologically active form. Doxycycline is virtually completely absorbed after oral administration.
Following a 200 mg dose, normal adult volunteers averaged peak serum levels of 2.6 mcg/mL of doxycycline at 2 hours, decreasing to 1.45 mcg/mL at 24 hours. Excretion of doxycycline by the kidney is about 40%/72 hours in individuals with normal function (creatinine clearance about 75 mL/min.). This percentage excretion may fall as low as 1 to 5%/72 hours in individuals with severe renal insufficiency (creatinine clearance below 10 mL/min.). Studies have shown no significant difference in serum half-life of doxycycline (range 18–22 hours) in individuals with normal and severely impaired renal function.
Hemodialysis does not alter serum half-life.
Results of animal studies indicate that tetracyclines cross the placenta and are found in fetal tissues.
Microbiology
Mechanism of Action
Doxycycline inhibits bacterial protein synthesis by binding to the 30S ribosomal subunit. Doxycycline has bacteriostatic activity against a broad range of Gram-positive and Gram-negative bacteria.
Antimicrobial Activity
Doxycycline has been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section of the package insert.
Gram-Negative Bacteria
Acinetobacter species
Bartonella bacilliformis
Brucella species
Klebsiella species
Klebsiella granulomatis
Campylobacter fetus
Enterobacter aerogenes
Escherichia coli
Francisella tularensis
Haemophilus ducreyi
Haemophilus influenzae
Neisseria gonorrhoeae
Shigella species
Vibrio cholerae
Yersinia pestis
Indications and Usage for Doxycycline Oral Suspension
To reduce the development of drug-resistant bacteria and maintain effectiveness of doxycycline and other antibacterial drugs, doxycycline should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Treatment
Doxycycline is indicated for the treatment of the following infections
- Rocky Mountain spotted fever, typhus fever and the typhus group, Q fever, rickettsialpox, and tick fevers caused by Rickettsiae.
- Respiratory tract infections caused by Mycoplasma pneumoniae.
- Lymphogranuloma venereum caused by Chlamydia trachomatis.
- Psittacosis (ornithosis) caused by Chlamydophila psittaci.
- Trachoma caused by Chlamydia trachomatis, although the infectious agent is not always eliminated, as judged by immunofluorescence.
- Inclusion conjunctivitis caused by Chlamydia trachomatis.
- Uncomplicated urethral, endocervical, or rectal infections in adults caused by Chlamydia trachomatis.
- Nongonococcal urethritis caused by Ureaplasma urealyticum.
- Relapsing fever due to Borrelia recurrentis.
Doxycycline is also indicated for the treatment of infections caused by the following gram-negative microorganisms:
- Chancroid caused by Haemophilus ducreyi.
- Plague due to Yersinia pestis.
- Tularemia due to Francisella tularensis.
- Cholera caused by Vibrio cholerae.
- Campylobacter fetus infections caused by Campylobacter fetus.
- Brucellosis due to Brucella species (in conjunction with streptomycin).
- Bartonellosis due to Bartonella bacilliformis.
- Granuloma inguinale caused by Klebsiella granulomatis.
Because many strains of the following groups of microorganisms have been shown to be resistant to doxycycline, culture and susceptibility testing are recommended.
Doxycycline is indicated for treatment of infections caused by the following gram-negative bacteria when bacteriologic testing indicates appropriate susceptibility to the drug:
- Escherichia coli.
- Enterobacter aerogenes.
- Shigella species.
- Acinetobacter species.
- Respiratory tract infections caused by Haemophilus influenzae.
- Respiratory tract and urinary tract infections caused by Klebsiella species.
Doxycycline is indicated for treatment of infections caused by the following gram-positive microorganisms when bacteriologic testing indicates appropriate susceptibility to the drug:
- Upper respiratory infections caused by Streptococcus pneumoniae.
- Anthrax due to Bacillus anthracis, including inhalational anthrax (post-exposure): to reduce the incidence or progression of disease following exposure to aerosolized Bacillus anthracis.
When penicillin is contraindicated, doxycycline is an alternative drug in the treatment of the following infections:
- Uncomplicated gonorrhea caused by Neisseria gonorrhoeae.
- Syphilis caused by Treponema pallidum.
- Yaws caused by Treponema pallidum subspecies pertenue.
- Listeriosis due to Listeria monocytogenes.
- Vincent's infection caused by Fusobacterium fusiforme.
- Actinomycosis caused by Actinomyces israelii.
- Infections caused by Clostridium species.
In acute intestinal amebiasis, doxycycline may be a useful adjunct to amebicides.
In severe acne, doxycycline may be useful adjunctive therapy.
Prophylaxis
Doxycycline is indicated for the prophylaxis of malaria due to Plasmodium falciparum in short-term travelers (<4 months) to areas with chloroquine and/or pyrimethamine-sulfadoxine resistant strains. (See DOSAGE AND ADMINISTRATION section and Information for Patients subsection of the PRECAUTIONS section.)
Contraindications
This drug is contraindicated in persons who have shown hypersensitivity to any of the tetracyclines.
Warnings
The use of drugs of the tetracycline class during tooth development (last half of pregnancy, infancy and childhood to the age of 8 years) may cause permanent discoloration of the teeth (yellow-gray-brown). This adverse reaction is more common during long-term use of the drugs, but it has been observed following repeated short-term courses. Enamel hypoplasia has also been reported. Use doxycycline in pediatric patients 8 years of age or less only when the potential benefits are expected to outweigh the risks in severe or life-threatening conditions (e.g., anthrax, Rocky Mountain spotted fever), particularly when there are no alternative therapies.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including doxycycline, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following the use of antibacterial drugs. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing use of antibacterial drugs not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Severe skin reactions, such as exfoliative dermatitis, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported in patients receiving doxycycline. (See ADVERSE REACTIONS.) If severe skin reactions occur, doxycycline should be discontinued immediately and appropriate therapy should be instituted.
Intracranial hypertension (IH, pseudotumor cerebri) has been associated with the use of tetracyclines including doxycycline. Clinical manifestations of IH include headache, blurred vision, diplopia, and vision loss; papilledema can be found on fundoscopy. Women of childbearing age who are overweight or have a history of IH are at greater risk for developing tetracycline associated IH. Concomitant use of isotretinoin and doxycycline should be avoided because isotretinoin is also known to cause pseudotumor cerebri.
Although IH typically resolves after discontinuation of treatment, the possibility for permanent visual loss exists. If visual disturbance occurs during treatment, prompt ophthalmologic evaluation is warranted. Since intracranial pressure can remain elevated for weeks after drug cessation patients should be monitored until they stabilize.
All tetracyclines form a stable calcium complex in any bone-forming tissue. A decrease in fibula growth rate has been observed in prematures given oral tetracycline in doses of 25 mg/kg every 6 hours. This reaction was shown to be reversible when the drug was discontinued.
Results of animal studies indicate that tetracyclines cross the placenta, are found in fetal tissues, and can have toxic effects on the developing fetus (often related to retardation of skeletal development). Evidence of embryotoxicity has also been noted in animals treated early in pregnancy. If any tetracycline is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
The antianabolic action of the tetracyclines may cause an increase in BUN. Studies to date indicate that this does not occur with the use of doxycycline in patients with impaired renal function.
Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines. Patients apt to be exposed to direct sunlight or ultraviolet light should be advised that this reaction can occur with tetracycline drugs, and treatment should be discontinued at the first evidence of skin erythema.
Precautions
General
As with other antibacterial drugs, use of doxycycline may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs, doxycycline should be discontinued and appropriate therapy instituted.
Incision and drainage or other surgical procedures should be performed in conjunction with antibacterial therapy, when indicated.
Doxycycline offers substantial but not complete suppression of the asexual blood stages of Plasmodium strains.
Doxycycline does not suppress P. falciparum's sexual blood stage gametocytes. Subjects completing this prophylactic regimen may still transmit the infection to mosquitoes outside endemic areas.
Prescribing doxycycline in the absence of proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Information For Patients
Patients taking doxycycline for malaria prophylaxis should be advised:
- –
- that no present-day antimalarial agent, including doxycycline, guarantees protection against malaria.
- –
- to avoid being bitten by mosquitoes by using personal protective measures that help avoid contact with mosquitoes, especially from dusk to dawn (e.g., staying in well-screened areas, using mosquito nets, covering the body with clothing, and using an effective insect repellent).
- –
- that doxycycline prophylaxis:
- –
- should begin 1–2 days before travel to the malarious area,
- –
- should be continued daily while in the malarious area and after leaving the malarious area,
- –
- should be continued for 4 further weeks to avoid development of malaria after returning from an endemic area,
- –
- should not exceed 4 months.
All patients taking doxycycline should be advised:
- –
- to avoid excessive sunlight or artificial ultraviolet light while receiving doxycycline and to discontinue therapy if phototoxicity (e.g., skin eruption, etc.) occurs. Sunscreen or sunblock should be considered. (See WARNINGS.)
- –
- to drink fluids liberally along with doxycycline to reduce the risk of esophageal irritation and ulceration. (See ADVERSE REACTIONS.)
- –
- that the absorption of tetracyclines is reduced when taken with foods, especially those which contain calcium. However, the absorption of doxycycline is not markedly influenced by simultaneous ingestion of food or milk. (See DRUG INTERACTIONS.)
- –
- that the absorption of tetracyclines is reduced when taking bismuth subsalicylate. (See DRUG INTERACTIONS.)
- –
- that the use of doxycycline might increase the incidence of vaginal candidiasis.
Patients should be counseled that antibacterial drugs, including doxycycline should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When doxycycline is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by doxycycline or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibacterial drugs, which usually ends when the antibacterials are discontinued. Sometimes after starting treatment with antibacterial drugs, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial drug. If this occurs, patients should contact their physician as soon as possible.
Laboratory Tests
In venereal disease, when co-existent syphilis is suspected, dark field examinations should be done before treatment is started and the blood serology repeated monthly for at least 4 months.
In long-term therapy, periodic laboratory evaluation of organ systems, including hematopoietic, renal, and hepatic studies, should be performed.
Drug Interactions
Because tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage.
Since bacteriostatic drugs may interfere with the bactericidal action of penicillin, it is advisable to avoid giving tetracyclines in conjunction with penicillin.
Absorption of tetracyclines is impaired by antacids containing aluminum, calcium, or magnesium, and iron-containing preparations.
Absorption of tetracyclines is impaired by bismuth subsalicylate.
Barbiturates, carbamazepine, and phenytoin decrease the half-life of doxycycline.
The concurrent use of tetracycline and Penthrane® (methoxyflurane) has been reported to result in fatal renal toxicity.
Concurrent use of tetracycline may render oral contraceptives less effective.
Drug/Laboratory Test Interactions
False elevations of urinary catecholamine levels may occur due to interference with the fluorescence test.
Carcinogenesis, Mutagenesis, Impairment Of Fertility
Long-term studies in animals to evaluate carcinogenic potential of doxycycline have not been conducted. However, there has been evidence of oncogenic activity in rats in studies with the related antibacterial drugs, oxytetracycline (adrenal and pituitary tumors), and minocycline (thyroid tumors).
Likewise, although mutagenicity studies of doxycycline have not been conducted, positive results in in vitro mammalian cell assays have been reported for related antibacterial drugs (tetracycline, oxytetracycline).
Doxycycline administered orally at dosage levels as high as 250 mg/kg/day had no apparent effect on the fertility of female rats. Effect on male fertility has not been studied.
Pregnancy
Teratogenic Effects
There are no adequate and well-controlled studies on the use of doxycycline in pregnant women. The vast majority of reported experience with doxycycline during human pregnancy is short-term, first trimester exposure. There are no human data available to assess the effects of long-term therapy of doxycycline in pregnant women, such as that proposed for treatment of anthrax exposure. An expert review of published data on experiences with doxycycline use during pregnancy by TERIS – the Teratogen Information System – concluded that therapeutic doses during pregnancy are unlikely to pose a substantial teratogenic risk (the quantity and quality of data were assessed as limited to fair), but the data are insufficient to state that there is no risk.1 A case-control study (18,515 mothers of infants with congenital anomalies and 32,804 mothers of infants with no congenital anomalies) shows a weak but marginally statistically significant association with total malformations and use of doxycycline anytime during pregnancy. Sixty-three (0.19%) of the controls and fifty-six (0.30%) of the cases were treated with doxycycline. This association was not seen when the analysis was confined to maternal treatment during the period of organogenesis (i.e., in the second and third months of gestation) with the exception of a marginal relationship with neural tube defect based on only two exposed cases.2
A small prospective study of 81 pregnancies describes 43 pregnant women treated for 10 days with doxycycline during early first trimester. All mothers reported their exposed infants were normal at 1 year of age.3
Nursing Mothers
Tetracyclines are excreted in human milk; however, the extent of absorption of tetracyclines, including doxycycline, by the breastfed infant is not known. Short-term use by lactating women is not necessarily contraindicated; however, the effects of prolonged exposure to doxycycline in breast milk are unknown.4 Because of the potential for serious adverse reactions in nursing infants from doxycycline, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. (See WARNINGS.)
Pediatric Use
Because of the effects of drugs of the tetracycline-class on tooth development and growth, use doxycycline in pediatric patients 8 years of age or less only when the potential benefits are expected to outweigh the risks in severe or life-threatening conditions (e.g., anthrax, Rocky Mountain spotted fever), particularly when there are no alternative therapies. (See WARNINGS and DOSAGE AND ADMINISTRATION.)
Adverse Reactions/Side Effects
Due to oral doxycycline's virtually complete absorption, side effects of the lower bowel, particularly diarrhea, have been infrequent. The following adverse reactions have been observed in patients receiving tetracyclines:
Gastrointestinal: anorexia, nausea, vomiting, diarrhea, glossitis, dysphagia, enterocolitis, inflammatory lesions (with monilial overgrowth) in the anogenital region, and pancreatitis. Hepatotoxicity has been reported rarely. These reactions have been caused by both the oral and parenteral administration of tetracyclines. Superficial discoloration of the adult permanent dentition, reversible upon drug discontinuation and professional dental cleaning has been reported. Permanent tooth discoloration and enamel hypoplasia may occur with drugs of the tetracycline class when used during tooth development. (See WARNINGS.) Rare instances of esophagitis and esophageal ulcerations have been reported in patients receiving capsule and tablet forms of the drugs in the tetracycline class. Most of these patients took medications immediately before going to bed. (See DOSAGE AND ADMINISTRATION).
Skin: toxic epidermal necrolysis, Stevens-Johnson syndrome, erythema multiforme, skin hyperpigmentation, maculopapular and erythematous rashes. Exfoliative dermatitis has been reported but is uncommon. Photosensitivity is discussed above. (See WARNINGS.)
Renal toxicity: Rise in BUN has been reported and is apparently dose related. (See WARNINGS.)
Immune: Hypersensitivity reactions including urticaria, angioneurotic edema, anaphylaxis, anaphylactoid purpura, serum sickness, pericarditis, exacerbation of systemic lupus erythematosus, drug reaction with eosinophilia and systemic symptoms (DRESS), and Jarisch-Herxheimer reaction has been reported in the setting of spirochete infections treated with doxycycline.
Blood: Hemolytic anemia, thrombocytopenia, neutropenia, and eosinophilia have been reported.
Other: Bulging fontanels in infants and intracranial hypertension in adults. (See WARNINGS).
When given over prolonged periods, tetracyclines have been reported to produce brown-black microscopic discoloration of the thyroid gland. No abnormalities of thyroid function studies are known to occur.
To report suspected ADVERSE REACTIONS, contact Chartwell RX, LLC. at 1-845-232-1683 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Related/similar drugs
Overdosage
In case of overdosage, discontinue medication, treat symptomatically and institute supportive measures. Dialysis does not alter serum half-life and thus would not be of benefit in treating cases of overdosage.
Doxycycline Oral Suspension Dosage and Administration
The usual dosage and frequency of administration of doxycycline differs from that of the other tetracyclines. Exceeding the recommended dosage may result in an increased incidence of side effects.
Adults
The usual dose of oral doxycycline is 200 mg on the first day of treatment (administered 100 mg every 12 hours) followed by a maintenance dose of 100 mg/day. In the management of more severe infections (particularly chronic infections of the urinary tract), 100 mg every 12 hours is recommended.
Pediatric Patients
For all pediatric patients weighing less than 45 kg with severe or life-threatening infections (e.g., anthrax, Rocky Mountain spotted fever), the recommended dosage is 2.2 mg/kg of body weight administered every 12 hours. Children weighing 45 kg or more should receive the adult dose. (See WARNINGS and PRECAUTIONS.)
For pediatric patients with less severe disease (greater than 8 years of age and weighing less than 45 kg), the recommended dosage schedule is 4.4 mg/kg of body weight divided into two doses on the first day of treatment, followed by a maintenance dose of 2.2 mg/kg of body weight (given as a single daily dose or divided into twice daily doses). For pediatric patients weighing over 45 kg, the usual adult dose should be used.
The therapeutic antibacterial serum activity will usually persist for 24 hours following recommended dosage.
When used in streptococcal infections, therapy should be continued for 10 days.
Administration of adequate amounts of fluid along with capsule and tablet forms of drugs in the tetracycline class is recommended to wash down the drugs and reduce the risk of esophageal irritation and ulceration. (See ADVERSE REACTIONS.)
If gastric irritation occurs, it is recommended that doxycycline be given with food or milk. The absorption of doxycycline is not markedly influenced by simultaneous ingestion of food or milk.
Studies to date have indicated that administration of doxycycline at the usual recommended doses does not lead to excessive accumulation of doxycycline in patients with renal impairment.
Uncomplicated gonococcal infections in adults (except anorectal infections in men): 100 mg, by mouth, twice a day for 7 days. As an alternate single visit dose, administer 300 mg stat followed in one hour by a second 300 mg dose. The dose may be administered with food, including milk or carbonated beverage, as required.
Uncomplicated urethral, endocervical, or rectal infection in adults caused by Chlamydia trachomatis: 100 mg, by mouth twice a day for 7 days.
Nongonococcal urethritis (NGU) caused by C. trachomatis or U. urealyticum: 100 mg by mouth, twice a day for 7 days.
Syphilis – early: Patients who are allergic to penicillin should be treated with doxycycline 100 mg, by mouth, twice a day for 2 weeks.
Syphilis of more than one year's duration: Patients who are allergic to penicillin should be treated with doxycycline 100 mg, by mouth, twice a day for 4 weeks.
Acute epididymo-orchitis caused by N. gonorrhoeae: 100 mg, by mouth, twice a day for at least 10 days.
Acute epididymo-orchitis caused by C. trachomatis: 100 mg, by mouth, twice a day for at least 10 days.
For prophylaxis of malaria: For adults, the recommended dose is 100 mg daily. For children over 8 years of age, the recommended dose is 2 mg/kg given once daily up to the adult dose. Prophylaxis should begin 1–2 days before travel to the malarious area. Prophylaxis should be continued daily during travel in the malarious area and for 4 weeks after the traveler leaves the malarious area.
Doxycycline for Oral Suspension Mixing Directions
Tap bottle lightly to loosen powder. Add 47.6 mL of water to the bottle to make a total volume of 60 mL. Shake well. This prescription, when in suspension, will maintain its potency for two weeks when kept at room temperature.
Discard Unused Portion After Two Weeks.
How is Doxycycline Oral Suspension supplied
Doxycycline Hyclate Capsules, USP 50 mg are available as a blue and white capsule filled with yellow powder, imprinted with "2984", containing doxycycline hyclate, equivalent to 50 mg doxycycline; 100 mg are available as a light blue capsule filled with yellow powder imprinted with "2985" containing doxycycline hyclate, equivalent to 100 mg of doxycycline.
NDC 62135-984-50 50 mg capsules - Bottles of 50 capsules
NDC 62135-985-50 100 mg capsules -Bottles of 50 capsules
NDC 62135-985-05 100 mg capsules -Bottles of 500 capsules
Doxycycline Hyclate Tablets, USP equivalent to 100 mg doxycycline: Round, orange film-coated tablet engraved with "3626" on one side, and plain on the other side.
NDC 62135-626-20 Bottles of 20 tablets
NDC 62135-626-28 Bottles of 28 tablets
NDC 62135-626-50 Bottles of 50 tablets
NDC 62135-626-12 Bottles of 120 tablets
NDC 62135-626-23 Bottles of 210 tablets
NDC 62135-626-31 Bottles of 300 tablets
NDC 62135-626-40 Bottles of 400 tablets
NDC 62135-626-05 Bottles of 500 tablets
NDC 62135-626-10 Bottles of 1000 tablets
NDC 62135-626-96 Bottles of 2000 tablets
Doxycycline for Oral Suspension USP is available as a raspberry-flavored, pink dry powder for oral suspension. When reconstituted, each teaspoonful (5 mL) contains doxycycline monohydrate equivalent to 25 mg of doxycycline in a 2 oz (60 mL) bottle.
NDC 62135-417-46
ANIMAL PHARMACOLOGY AND ANIMAL TOXICOLOGY
Hyperpigmentation of the thyroid has been produced by members of the tetracycline class in the following species: in rats by oxytetracycline, doxycycline, tetracycline PO4, and methacycline; in minipigs by doxycycline, minocycline, tetracycline PO4, and methacycline; in dogs by doxycycline and minocycline; in monkeys by minocycline.
Minocycline, tetracycline PO4, methacycline, doxycycline, tetracycline base, oxytetracycline HCl, and tetracycline HCl were goitrogenic in rats fed a low iodine diet. This goitrogenic effect was accompanied by high radioactive iodine uptake. Administration of minocycline also produced a large goiter with high radioiodine uptake in rats fed a relatively high iodine diet.
Treatment of various animal species with this class of drugs has also resulted in the induction of thyroid hyperplasia in the following: in rats and dogs (minocycline); in chickens (chlortetracycline); and in rats and mice (oxytetracycline). Adrenal gland hyperplasia has been observed in goats and rats treated with oxytetracycline.
References
- Friedman JM and Polifka JE. Teratogenic Effects of Drugs. A Resource for Clinicians (TERIS). Baltimore, MD: The Johns Hopkins University Press, 2000: 149–195.
- Cziezel AE and Rockenbauer M. Teratogenic study of doxycycline. Obstet Gynecol 1997; 89: 524–528.
- Horne HW Jr and Kundsin RB. The role of mycoplasma among 81 consecutive pregnancies: a prospective study. Int J Fertil 1980; 25: 315–317.
- Hale T. Medications and Mothers Milk. 9th edition. Amarillo, TX: Pharmasoft Publishing, 2000: 225–226.
This product's label may have been updated. For current full prescribing information, please call 845-232-1683.
® Penthrane is a trademark of Abbott Laboratories.
Manufactured by:
Chartwell Pharmaceuticals, LLC.
Congers, NY 10920
Manufactured for:
Chartwell RX, LLC.
Congers, NY 10920
Made in USA
L70282
Rev. 11/2019
| DOXYCYCLINE
doxycycline powder, for suspension |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Chartwell RX LLC (079394054) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Chartwell Pharmaceuticals LLC | 962619982 | MANUFACTURE(62135-417) | |
Frequently asked questions
- Is Doxycycline safe for dogs? Uses, dosage, side effects
- What are the most common skin conditions? (with photos)
- Doxycycline for STDs: Effectiveness, Dosage and Side Effects
- Should doxycycline be taken with food?
- Does doxycycline work for acne?
- What are the best antibiotics for pneumonia?
- What is the best antibiotic to treat a sinus infection?
- Does Doxycycline make you tired?
- Can I take doxycycline for a strep throat?
More about doxycycline
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (1,646)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: miscellaneous antimalarials
- Breastfeeding
- En español
Patient resources
Professional resources
- Doxycycline monograph
- Doxycycline (Topical) (AHFS Monograph)
- Doxycycline Hyclate (EENT) (AHFS Monograph)
- Doxycycline Delayed Release Capsule (FDA)
- Doxycycline Hyclate (FDA)
Other brands
Vibramycin, Monodox, Doryx, Oracea, ... +7 more


