Cetirizine and Pseudoephedrine: Package Insert / Prescribing Info
Package insert / product label
Generic name: cetirizine hydrochloride, pseudoephedrine hydrochloride
Dosage form: tablet, film coated, extended release
Drug class: Upper respiratory combinations
Medically reviewed by Drugs.com. Last updated on Mar 24, 2025.
Indications and Usage for Cetirizine and Pseudoephedrine
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- nasal congestion
- sneezing
- itching of the nose or throat
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily restores freer breathing through the nose
Warnings
Do not use
- if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- heart disease
- thyroid disease
- diabetes
- glaucoma
- high blood pressure
- trouble urinating due to an enlarged prostate gland
- liver or kidney disease.Your doctor should determine if you need a different dose.
When using this product
- do not use more than directed
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if
- an allergic reaction to this product occurs. Seek medical help right away.
- you get nervous, dizzy, or sleepless
- symptoms do not improve within 7 days or are accompanied by fever
Cetirizine and Pseudoephedrine Dosage and Administration
- do not break or chew tablet; swallow tablet whole
| adults and children 12 years and over | take 1 tablet every 12 hours; do not take more than 2 tablets in 24 hours. |
| adults 65 years and over | ask a doctor |
| children under 12 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information
- Safety sealed: do not use if the imprinted bottle seal is open or torn (for bottle only).
- DO NOT USE IF BLISTER UNIT IS BROKEN OR TORN (for blister package only).
Related/similar drugs
Inactive Ingredients
Colloidal silicon dioxide, croscarmellose sodium, D & C yellow aluminum lake, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate, povidone, and titanium dioxide.
5 mg/120 mg Label
NDC 0781-5285-31
Cetirizine HCl and
Pseudoephedrine HCl
Extended Release Tablets
5 mg/120 mg
antihistamine/
nasal decongestant
Indoor & Outdoor Allergies
ALLERGY & CONGESTION
12 hour Relief of
Runny Nose Itchy, Watery Eyes Sinus Pressure
Sneezing Itchy Throat or Nose Nasal Congestion
SANDOZ
30 Tablets

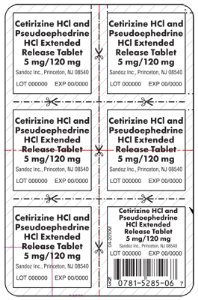
5 mg/120 mg Blister Pack
Cetirizine HCl and
Pseudoephedrine
HCl Extended
Release Tablet
5 mg/120 mg
Sandoz Inc., Princeton, NJ 08540
5 mg/120 mg Blister Pack Carton
NDC 0781-5285-40
Cetirizine HCl and
Pseudoephedrine HCl
Extended Release Tablets
5 mg/120 mg
antihistamine/nasal decongestant
24 Tablets (4 x 6)
SANDOZ
ALLERGY &
CONGESTION
12 hour Relief of
Runny Nose
Itchy, Watery Eyes
Sinus Pressure
Sneezing
Itchy Throat or Nose
Nasal Congestion
Indoor &
Outdoor
Allergies

| CETIRIZINE HCL AND PSEUDOEPHEDRINE HCL
ER
cetirizine hcl and pseudoephedrine hcl tablet, film coated, extended release |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Sandoz Inc (110342024) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sandoz Inc | 110342024 | MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Thermo-Pak, Inc | 161049044 | PACK, LABEL | |
More about cetirizine / pseudoephedrine
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (8)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: upper respiratory combinations
- En español
Patient resources
Professional resources
Other brands
Zyrtec-D 12 Hour, Allergy Relief D, Wal-Zyr D, All Day Allergy-D

