Cefazolin Injection: Package Insert / Prescribing Info
Package insert / product label
Dosage form: injection, powder, for solution
Drug class: First generation cephalosporins
J Code (medical billing code): J0690 (Up to 500 mg, injection)
Medically reviewed by Drugs.com. Last updated on Jul 31, 2025.
On This Page
Rx Only
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin for Injection and other antibacterial drugs, Cefazolin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
Cefazolin Injection Description
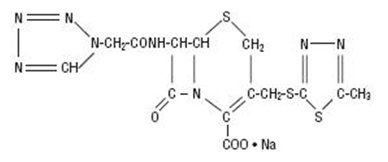
Cefazolin for Injection, USP is a semi-synthetic cephalosporin for parenteral administration. It is the sodium salt of 3-{[(5-methyl-1,3,4-thiadiazol-2-yl)thio]-methyl}-8-oxo-7-[2-(1H-tetrazol-1-yl) acetamido]-5-thia-1-azabicyclo [4.2.0]oct-2-ene-2-carboxylic acid. The molecular formula is C14H13N8NaO4S3 and molecular weight is 476.49.
Structural Formula:
Each vial contains 48 mg of sodium/1 gram of cefazolin sodium.
Cefazolin for Injection, USP is white to off-white crystalline powder, supplied in vials equivalent to 1 gram of cefazolin.
Cefazolin Injection - Clinical Pharmacology
After intramuscular administration of Cefazolin for Injection to normal volunteers, the mean serum concentrations were 37 mcg/mL at 1 hour and 3 mcg/mL at 8 hours following a 500 mg dose, and 64 mcg/mL at 1 hour and 7 mcg/mL at 8 hours following a 1 gram dose.
Studies have shown that following intravenous administration of Cefazolin for Injection to normal volunteers, mean serum concentrations peaked at approximately 185 mcg/mL and were approximately 4 mcg/mL at 8 hours for a 1 gram dose.
The serum half-life for Cefazolin for Injection is approximately 1.8 hours following IV administration and approximately 2 hours following IM administration.
In a study (using normal volunteers) of constant intravenous infusion with dosages of 3.5 mg/kg for 1 hour (approximately 250 mg) and 1.5 mg/kg the next 2 hours (approximately 100 mg), Cefazolin for Injection produced a steady serum level at the third hour of approximately 28 mcg/mL.
Studies in patients hospitalized with infections indicate that Cefazolin for Injection produces mean peak serum levels approximately equivalent to those seen in normal volunteers.
Bile levels in patients without obstructive biliary disease can reach or exceed serum levels by up to five times; however, in patients with obstructive biliary disease, bile levels of Cefazolin for Injection are considerably lower than serum levels (< 1 mcg/mL).
In synovial fluid, the level of Cefazolin for Injection becomes comparable to that reached in serum at about 4 hours after drug administration. Studies of cord blood show prompt transfer of Cefazolin for Injection across the placenta. Cefazolin for Injection is present in very low concentrations in the milk of nursing mothers.
Cefazolin for Injection is excreted unchanged in the urine. In the first 6 hours approximately 60% of the drug is excreted in the urine and this increases to 70% to 80% within 24 hours. Cefazolin for Injection achieves peak urine concentrations of approximately 2,400 mcg/mL and 4,000 mcg/mL respectively following 500 mg and 1 gram intramuscular doses.
In patients undergoing peritoneal dialysis (2 L/hr.), Cefazolin for Injection produced mean serum levels of approximately 10 and 30 mcg/mL after 24 hours’ instillation of a dialyzing solution containing 50 mg/L and 150 mg/L, respectively. Mean peak levels were 29 mcg/mL (range 13 to 44 mcg/mL) with 50 mg/L (3 patients), and 72 mcg/mL (range 26 to 142 mcg/mL) with 150 mg/L (6 patients). Intraperitoneal administration of Cefazolin for Injection is usually well tolerated.
Controlled studies on adult normal volunteers, receiving 1 gram 4 times a day for 10 days, monitoring CBC, SGOT, SGPT, bilirubin, alkaline phosphatase, BUN, creatinine and urinalysis, indicated no clinically significant changes attributed to Cefazolin for Injection.
Microbiology
Mechanism of Action
Cefazolin is a bactericidal agent that acts by inhibition of bacterial cell wall synthesis.
Resistance
Predominant mechanisms of bacterial resistance to cephalosporins include the presence of extended-spectrum beta-lactamases and enzymatic hydrolysis.
Antimicrobial Activity
Cefazolin has been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE (1) section:
Gram-Positive Bacteria:
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus agalactiae
Streptococcus pneumoniae
Streptococcus pyogenes
Methicillin-resistant staphylococci are uniformly resistant to cefazolin.
Gram-Negative Bacteria:
Escherichia coli
Proteus mirabilis
Most isolates of indole positive Proteus (Proteus vulgaris), Enterobacter spp., Morganella morganii, Providencia rettgeri, Serratia spp., and Pseudomonas spp. are resistant to cefazolin.
Indications and Usage for Cefazolin Injection
Cefazolin for Injection, USP is indicated in the treatment of the following serious infections due to susceptible organisms:
Respiratory Tract Infections: Due to S. pneumoniae, Klebsiella species, H. influenzae, S. aureus (penicillin-sensitive and penicillin-resistant), and group A beta-hemolytic streptococci. Injectable benzathine penicillin is considered to be the drug of choice in treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever.
Cefazolin for Injection is effective in the eradication of streptococci from the nasopharynx; however, data establishing the efficacy of Cefazolin for Injection in the subsequent prevention of rheumatic fever are not available at present.
Urinary Tract Infections: Due to E. coli, P. mirabilis, Klebsiella species, and some strains of enterobacter and enterococci.
Skin and Skin Structure Infections: Due to S. aureus (penicillin-sensitive and penicillin-resistant), group A beta-hemolytic streptococci, and other strains of streptococci.
Biliary Tract Infections: Due to E. coli, various strains of streptococci, P. mirabilis, Klebsiella species, and S. aureus.
Bone and Joint Infections: Due to S. aureus.
Genital Infections: (i.e., prostatitis, epididymitis) due to E. coli, P. mirabilis, Klebsiella species, and some strains of enterococci.
Septicemia: Due to S. pneumoniae, S. aureus (penicillin-sensitive and penicillin-resistant), P. mirabilis, E. coli and Klebsiella species.
Endocarditis: Due to S. aureus (penicillin-sensitive and penicillin-resistant) and group A beta-hemolytic streptococci.
Perioperative Prophylaxis: The prophylactic administration of Cefazolin for Injection preoperatively, intraoperatively and postoperatively may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures which are classified as contaminated or potentially contaminated (e.g., vaginal hysterectomy, and cholecystectomy in high-risk patients such as those older than 70 years, with acute cholecystitis, obstructive jaundice, or common duct bile stones).
The perioperative use of Cefazolin for Injection may also be effective in surgical patients in whom infection at the operative site would present a serious risk (e.g., during open-heart surgery and prosthetic arthroplasty).
The prophylactic administration of Cefazolin for Injection should usually be discontinued within a 24 hour period after the surgical procedure. In surgery where the occurrence of infection may be particularly devastating (e.g., open-heart surgery and prosthetic arthroplasty), the prophylactic administration of Cefazolin for Injection may be continued for 3 to 5 days following the completion of surgery.
If there are signs of infection, specimens for cultures should be obtained for the identification of the causative organism so that appropriate therapy may be instituted (see DOSAGE AND ADMINISTRATION).
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin for Injection, USP and other antibacterial drugs, Cefazolin for Injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Contraindications
Cefazolin for Injection IS CONTRAINDICATED IN PATIENTS WITH KNOWN ALLERGY TO THE CEPHALOSPORIN GROUP OF ANTIBIOTICS.
Warnings
BEFORE THERAPY WITH CEFAZOLIN FOR INJECTION IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEFAZOLIN, CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. IF THIS PRODUCT IS GIVEN TO PENICILLIN-SENSITIVE PATIENTS, CAUTION SHOULD BE EXERCISED BECAUSE CROSS-HYPERSENSITIVITY AMONG BETA-LACTAM ANTIBIOTICS HAS BEEN CLEARLY DOCUMENTED AND MAY OCCUR IN UP TO 10% OF PATIENTS WITH A HISTORY OF PENICILLIN ALLERGY. IF AN ALLERGIC REACTION TO CEFAZOLIN FOR INJECTION OCCURS, DISCONTINUE TREATMENT WITH THE DRUG. SERIOUS ACUTE HYPERSENSITIVITY REACTIONS MAY REQUIRE TREATMENT WITH EPINEPHRINE AND OTHER EMERGENCY MEASURES, INCLUDING OXYGEN, IV FLUIDS, IV ANTIHISTAMINES, CORTICOSTEROIDS, PRESSOR AMINES, AND AIRWAY MANAGEMENT, AS CLINICALLY INDICATED.
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including cefazolin, and may range in severity from mild to life-threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that a toxin produced by Clostridium difficile is a primary cause of “antibiotic-associated colitis”.
After the diagnosis of pseudomembranous colitis has been established, therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation, and treatment with an oral antibacterial drug clinically effective against C. difficile colitis.
Precautions
General
Prolonged use of Cefazolin for Injection may result in the overgrowth of nonsusceptible organisms. Careful clinical observation of the patient is essential.
When Cefazolin for Injection is administered to patients with low urinary output because of impaired renal function, lower daily dosage is required (see DOSAGE AND ADMINISTRATION).
As with other β-lactam antibiotics, seizures may occur if inappropriately high doses are administered to patients with impaired renal function (see DOSAGE AND ADMINISTRATION).
Cefazolin for Injection, as with all cephalosporins, should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Cephalosporins may be associated with a fall in prothrombin activity. Those at risk include patients with renal or hepatic impairment or poor nutritional state, as well as patients receiving a protracted course of antimicrobial therapy, and patients previously stabilized on anticoagulant therapy. Prothrombin time should be monitored in patients at risk and exogenous vitamin K administered as indicated.
Prescribing Cefazolin for Injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Drug Interactions
Probenecid may decrease renal tubular secretion of cephalosporins when used concurrently, resulting in increased and more prolonged cephalosporin blood levels.
Drug/Laboratory Test Interactions
A false positive reaction for glucose in the urine may occur with Benedict's solution, Fehling’s solution or with CLINITEST® tablets, but not with enzyme-based tests such as CLINISTIX®.
Positive direct and indirect antiglobulin (Coombs) tests have occurred; these may also occur in neonates whose mothers received cephalosporins before delivery.
Information for Patients
Patients should be counseled that antibacterial drugs including Cefazolin for Injection, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Cefazolin for Injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may: (1) decrease the effectiveness of the immediate treatment, and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Cefazolin for Injection or other antibacterial drugs in the future.
Carcinogenesis/Mutagenesis
Mutagenicity studies and long-term studies in animals to determine the carcinogenic potential of Cefazolin for Injection have not been performed.
Pregnancy
Teratogenic Effects
Reproduction studies have been performed in rats, mice and rabbits at doses up to 25 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to Cefazolin for Injection. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Labor and Delivery
When cefazolin has been administered prior to caesarean section, drug levels in cord blood have been approximately one quarter to one third of maternal drug levels. The drug appears to have no adverse effect on the fetus.
Nursing Mothers
Cefazolin for Injection is present in very low concentrations in the milk of nursing mothers. Caution should be exercised when Cefazolin for Injection is administered to a nursing woman.
Pediatric Use
Safety and effectiveness for use in premature infants and neonates have not been established. See DOSAGE AND ADMINISTRATION for recommended dosage in pediatric patients older than 1 month.
Geriatric Use
Of the 920 subjects who received Cefazolin for Injection in clinical studies, 313 (34%) were 65 years and over, while 138 (15%) were 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see PRECAUTIONS, General and DOSAGE AND ADMINISTRATION).
Adverse Reactions/Side Effects
The following reactions have been reported:
Gastrointestinal
Diarrhea, oral candidiasis (oral thrush), vomiting, nausea, stomach cramps, anorexia, and pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment (see WARNINGS). Nausea and vomiting have been reported rarely.
Allergic
Anaphylaxis, eosinophilia, itching, drug fever, skin rash, Stevens-Johnson syndrome.
Hematologic
Neutropenia, leukopenia, thrombocytopenia, thrombocythemia.
Hepatic
Transient rise in SGOT, SGPT, and alkaline phosphatase levels has been observed. As with other cephalosporins, reports of hepatitis have been received.
Renal
As with other cephalosporins, reports of increased BUN and creatinine levels, as well as renal failure, have been received.
Local Reactions
Rare instances of phlebitis have been reported at site of injection. Pain at the site of injection after intramuscular administration has occurred infrequently. Some induration has occurred.
Other Reactions
Genital and anal pruritus (including vulvar pruritus, genital moniliasis, and vaginitis).
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Pharmaceuticals USA Inc. at 1-877-845-0689, or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Related/similar drugs
Cefazolin Injection Dosage and Administration
Usual Adult Dosage
|
||
| Type of Infection | Dose | Dose Frequency |
| Moderate to severe infections | 500 mg to 1 gram | every 6 to 8 hours |
| Mild infections caused by susceptible gram–positive cocci | 250 mg to 500 mg | every 8 hours |
| Acute, uncomplicated urinary tract infections | 1 gram | every 12 hours |
| Pneumococcal pneumonia | 500 mg | every 12 hours |
| Severe, life-threatening infections (e.g., endocarditis, septicemia)* | 1 gram to 1.5 grams | every 6 hours |
Perioperative Prophylactic Use
To prevent postoperative infection in contaminated or potentially contaminated surgery, recommended doses are:
a. 1 gram IV or IM administered 1/2 hour to 1 hour prior to the start of surgery.
b. For lengthy operative procedures (e.g., 2 hours or more), 500 mg to 1 gram IV or IM during surgery (administration modified depending on the duration of the operative procedure).
c. 500 mg to 1 gram IV or IM every 6 to 8 hours for 24 hours postoperatively.
It is important that (1) the preoperative dose be given just (1/2 to 1 hour) prior to the start of surgery so that adequate antibiotic levels are present in the serum and tissues at the time of initial surgical incision; and (2) Cefazolin for Injection be administered, if necessary, at appropriate intervals during surgery to provide sufficient levels of the antibiotic at the anticipated moments of greatest exposure to infective organisms.
In surgery where the occurrence of infection may be particularly devastating (e.g., open-heart surgery and prosthetic arthroplasty), the prophylactic administration of Cefazolin for Injection may be continued for 3 to 5 days following the completion of surgery.
Dosage Adjustment for Patients with Reduced Renal Function
Cefazolin for Injection may be used in patients with reduced renal function with the following dosage adjustments: Patients with a creatinine clearance of 55 mL/min. or greater or a serum creatinine of 1.5 mg % or less can be given full doses. Patients with creatinine clearance rates of 35 to 54 mL/min. or serum creatinine of 1.6 to 3 mg % can also be given full doses but dosage should be restricted to at least 8 hour intervals. Patients with creatinine clearance rates of 11 to 34 mL/min. or serum creatinine of 3.1 to 4.5 mg % should be given 1/2 the usual dose every 12 hours. Patients with creatinine clearance rates of 10 mL/min. or less or serum creatinine of 4.6 mg % or greater should be given 1/2 the usual dose every 18 to 24 hours. All reduced dosage recommendations apply after an initial loading dose appropriate to the severity of the infection. Patients undergoing peritoneal dialysis: See CLINICAL PHARMACOLOGY.
Pediatric Dosage
In pediatric patients, a total daily dosage of 25 to 50 mg per kg (approximately 10 to 20 mg per pound) of body weight, divided into 3 or 4 equal doses, is effective for most mild to moderately severe infections. Total daily dosage may be increased to 100 mg per kg (45 mg per pound) of body weight for severe infections. Since safety for use in premature infants and in neonates has not been established, the use of Cefazolin for Injection in these patients is not recommended.
| Weight | 25 mg/kg/day Divided into 3 Doses | 25 mg/kg/day Divided into 4 Doses | |||
| Lbs | Kg | Approximate Single Dose mg/q8h | Vol. (mL) needed with dilution of 125 mg/mL | Approximate Single Dose mg/q6h | Vol. (mL) needed with dilution of 125 mg/mL |
| 10 | 4.5 | 40 mg | 0.35 mL | 30 mg | 0.25 mL |
| 20 | 9 | 75 mg | 0.6 mL | 55 mg | 0.45 mL |
| 30 | 13.6 | 115 mg | 0.9 mL | 85 mg | 0.7 mL |
| 40 | 18.1 | 150 mg | 1.2 mL | 115 mg | 0.9 mL |
| 50 | 22.7 | 190 mg | 1.5 mL | 140 mg | 1.1 mL |
| Weight | 50 mg/kg/day Divided into 3 Doses | 50 mg/kg/day Divided into 4 Doses | |||
| Lbs | Kg | Approximate Single Dose mg/q8h | Vol. (mL) needed with dilution of 225 mg/mL | Approximate Single Dose mg/q6h | Vol. (mL) needed with dilution of 225 mg/mL |
| 10 | 4.5 | 75 mg | 0.35 mL | 55 mg | 0.25 mL |
| 20 | 9 | 150 mg | 0.7 mL | 110 mg | 0.5 mL |
| 30 | 13.6 | 225 mg | 1 mL | 170 mg | 0.75 mL |
| 40 | 18.1 | 300 mg | 1.35 mL | 225 mg | 1 mL |
| 50 | 22.7 | 375 mg | 1.7 mL | 285 mg | 1.25 mL |
In pediatric patients with mild to moderate renal impairment (creatinine clearance of 70 to 40 mL/min.), 60 percent of the normal daily dose given in equally divided doses every 12 hours should be sufficient. In patients with moderate impairment (creatinine clearance of 40 to 20 mL/min.), 25 percent of the normal daily dose given in equally divided doses every 12 hours should be adequate. Pediatric patients with severe renal impairment (creatinine clearance of 20 to 5 mL/min.) may be given 10 percent of the normal daily dose every 24 hours. All dosage recommendations apply after an initial loading dose.
RECONSTITUTION
Preparation of Parenteral Solution
Parenteral drug products should be SHAKEN WELL when reconstituted, and inspected visually for particulate matter prior to administration. If particulate matter is evident in reconstituted fluids, the drug solutions should be discarded.
When reconstituted or diluted according to the instructions below, Cefazolin for Injection is stable for 24 hours at room temperature or for 10 days if stored under refrigeration (5°C or 41°F). Reconstituted solutions may range in color from pale yellow to yellow without a change in potency.
Note on Refrigeration and Crystallization
Refrigeration of reconstituted solutions may result in crystal formation. This crystallization phenomenon is expected after refrigeration and does not affect the quality or potency of the
product. Before administration, reconstituted solutions with a crystallized appearance should be redissolved by hand-warming the vials until the solution is clear.
ADMINISTRATION
Intramuscular Administration
Reconstitute vials with Sterile Water for Injection according to the dilution table above. Shake well until dissolved. Cefazolin for Injection should be injected into a large muscle mass. Pain on injection is infrequent with Cefazolin for Injection.
Intravenous Administration
Direct (bolus) injection: Following reconstitution according to the above table, further dilute vials with approximately 5 mL Sterile Water for Injection. Inject the solution slowly over 3 to 5 minutes, directly or through tubing for patients receiving parenteral fluids (see list below).
Intermittent or continuous infusion: Dilute reconstituted Cefazolin for Injection in 50 to 100 mL of 1 of the following solutions:
- Sodium Chloride Injection, USP
- 5% or 10% Dextrose Injection, USP
- 5% Dextrose in Lactated Ringer's Injection, USP
- 5% Dextrose and 0.9% Sodium Chloride Injection, USP
- 5% Dextrose and 0.45% Sodium Chloride Injection, USP
- 5% Dextrose and 0.2% Sodium Chloride Injection, USP
- Lactated Ringer's Injection, USP
- Invert Sugar 5% or 10% in Sterile Water for Injection
- Ringer's Injection, USP
- 5% Sodium Bicarbonate Injection, USP
How is Cefazolin Injection supplied
Cefazolin for Injection, USP is supplied in vials containing cefazolin sodium equivalent to 1 gram cefazolin.
| Cefazolin for Injection, USP | Vial Size | Packaged | NDC No. |
| 1 gram | 10 mL | Carton of 25 vials | 72572-055-25 |
Also available as Pharmacy Bulk Package, as follows:
| Cefazolin for Injection, USP | Vial Size | Packaged | NDC No. |
| 10 grams | 100 mL | Carton of 10 vials | 72572-056-10 |
As with other cephalosporins, Cefazolin for Injection, USP tends to darken depending on storage conditions; within the stated recommendations, however, product potency is not adversely affected.
Before reconstitution protect from light and store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
CLINITEST is a registered trademark of Miles, Inc.
CLINISTIX is a registered trademark of Bayer Corporation.
Distributed by:
Civica, Inc.
Lehi, Utah 84043
Manufactured by:
Hikma Farmacêutica (Portugal), S.A.
Estrada do Rio da Mó, 8, 8A e 8B – Fervença – 2705-906 Terrugem SNT, Portugal
Revised: January 2025
PIN603-CIV/2
| CEFAZOLIN
cefazolin injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Civica, Inc. (081373942) |
| Registrant - HIKMA FARMACEUTICA (PORTUGAL), S.A. (452742943) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| HIKMA FARMACEUTICA (PORTUGAL), S.A | 452742943 | analysis(72572-055) , label(72572-055) , manufacture(72572-055) , pack(72572-055) | |
More about cefazolin
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (5)
- Latest FDA alerts (3)
- Side effects
- Dosage information
- During pregnancy
- Drug class: first generation cephalosporins
- Breastfeeding
- En español


