Byvalson: Package Insert / Prescribing Info
Package insert / product label
Generic name: nebivolol hydrochloride and valsartan
Dosage form: tablet, film coated
Drug class: Miscellaneous antihypertensive combinations
Medically reviewed by Drugs.com. Last updated on Jan 20, 2025.
The Byvalson brand name has been discontinued in the U.S. If generic versions of this product have been approved by the FDA, there may be generic equivalents available.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
BYVALSON (nebivolol and valsartan) tablets, for oral use
Initial U.S. Approval: 2016
WARNING: FETAL TOXICITY
See full prescribing information for complete boxed warning.
Indications and Usage for Byvalson
BYVALSON is a beta adrenergic blocker and an angiotensin II receptor blocker (ARB) indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. (1)
Byvalson Dosage and Administration
- As initial therapy and in patients not adequately controlled on valsartan 80 mg or nebivolol up to and including 10 mg, the recommended dose is 5 mg/ 80 mg taken orally once daily. (2)
- Maximum antihypertensive effects are attained within 2 to 4 weeks. (2)
- BYVALSON may be substituted for its components in patients already receiving 5 mg nebivolol and 80 mg valsartan. (2)
Dosage Forms and Strengths
- Tablets: 5 mg/ 80 mg (3)
Contraindications
- Severe bradycardia (4)
- Heart block greater than first degree (4)
- Patients with cardiogenic shock (4)
- Decompensated cardiac failure (4)
- Sick sinus syndrome (unless a permanent pacemaker is in place) (4)
- Patients with severe hepatic impairment (Child-Pugh >B) (4)
- Hypersensitivity to any component of this product (4)
- Do not co-administer aliskiren with BYVALSON in patients with diabetes. (4)
Warnings and Precautions
Adverse Reactions/Side Effects
No adverse reactions were observed more frequently on BYVALSON than on placebo. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Allergan at 1-800-433-8871 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- CYP2D6 enzyme inhibitors increase nebivolol levels. (7)
- Reserpine or clonidine may produce excessive reduction of sympathetic activity. (7)
- Digitalis glycosides increase the risk of bradycardia. (7)
- Verapamil- or diltiazem-type calcium channel blockers may cause excessive reductions in heart rate, blood pressure, and cardiac contractility. (7)
- Potassium sparing diuretics, potassium supplements or salt substitutes may lead to increases in serum potassium. (7)
- NSAID use may lead to increased risk of renal impairment and loss of antihypertensive effect. (7)
- Dual inhibition of the renin-angiotensin system: Increased risk of renal impairment, hypotension, and hyperkalemia. (7)
- Lithium: Increases in serum lithium concentrations and lithium toxicity (7)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 1/2019
Full Prescribing Information
1. Indications and Usage for Byvalson
BYVALSON is indicated for the treatment of hypertension, to lower blood pressure.
Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes, including the β-blocker class to which nebivolol principally belongs and the ARB class to which valsartan principally belongs. There are no controlled trials demonstrating risk reduction with BYVALSON.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mm Hg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
BYVALSON may be used alone or in combination with other antihypertensive agents.
2. Byvalson Dosage and Administration
As initial therapy and in patients not adequately controlled on valsartan 80 mg or nebivolol up to and including 10 mg, the recommended dose of BYVALSON is one tablet, 5 mg/ 80 mg (nebivolol/valsartan) taken orally once daily. Maximum antihypertensive effects are attained within 2 to 4 weeks. Increasing the dose of BYVALSON does not result in any meaningful further blood pressure reduction [see Clinical Studies (14)].
BYVALSON may be substituted for its components in patients already receiving 5 mg nebivolol and 80 mg valsartan.
3. Dosage Forms and Strengths
BYVALSON is available as a purple, capsule shaped, film-coated tablet with FL1 debossed on one side containing 5 mg of nebivolol and 80 mg of valsartan.
4. Contraindications
BYVALSON is contraindicated in the following conditions:
- Severe bradycardia
- Heart block greater than first degree
- Patients with cardiogenic shock
- Decompensated cardiac failure
- Sick sinus syndrome (unless a permanent pacemaker is in place)
- Patients with severe hepatic impairment (Child-Pugh >B)
- Patients who are hypersensitive to any component of this product.
- Do not co-administer aliskiren with BYVALSON in patients with diabetes [see Drug Interactions (7)].
5. Warnings and Precautions
5.1 Fetal Toxicity
Valsartan
Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue BYVALSON as soon as possible [see Use in Specific Populations (8.1)].
5.2 Hypotension
In patients with an activated renin-angiotensin-aldosterone system, such as volume- and/or salt-depleted patients (e.g., those receiving high doses of diuretics), symptomatic hypotension may occur in patients receiving BYVALSON. Correct these conditions prior to administration of BYVALSON, or start the treatment under close medical supervision.
If excessive hypotension occurs, the patient should be placed in the supine position and, if necessary, given an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further treatment, which usually can be continued without difficulty once the blood pressure has stabilized.
5.3 Abrupt Cessation of Therapy
Nebivolol
Do not abruptly discontinue BYVALSON in patients with coronary artery disease. Severe exacerbation of angina, myocardial infarction and ventricular arrhythmias have been reported in patients with coronary artery disease following the abrupt discontinuation of therapy with β-blockers. Myocardial infarction and ventricular arrhythmias may occur with or without preceding exacerbation of the angina pectoris. As with other β-blocker therapies, when discontinuation of BYVALSON is planned, carefully observe and advise patients to minimize physical activity. Taper nebivolol using monotherapy over 1 to 2 weeks when possible. If the angina worsens re-start nebivolol promptly, at least temporarily.
5.6 Anesthesia and Major Surgery
Nebivolol
Chronically administered beta-blocking therapy should not be routinely withdrawn prior to major surgery, however the impaired ability of the heart to respond to reflex adrenergic stimuli may augment the risks of general anesthesia and surgical procedures. Monitor patients closely when anesthetic agents which depress myocardial function, such as ether, cyclopropane, and trichloroethylene, are used.
5.7 Diabetes and Hypoglycemia
Nebivolol
β-blockers may mask some of the manifestations of hypoglycemia, particularly tachycardia. Nonselective β-blockers may potentiate insulin-induced hypoglycemia and delay recovery of serum glucose levels. It is not known whether nebivolol has these effects. Advise patients subject to spontaneous hypoglycemia and diabetic patients receiving insulin or oral hypoglycemic agents about these possibilities.
5.11 Impaired Renal Function
Valsartan
Changes in renal function including acute renal failure can be caused by drugs that inhibit the renin-angiotensin system and by diuretics. Patients whose renal function may depend in part on the activity of the renin-angiotensin system (e.g. patients with renal artery stenosis, chronic kidney disease, severe congestive heart failure, or volume depletion) may be at particular risk of developing acute renal failure on valsartan. Monitor renal function periodically in these patients. Consider withholding or discontinuing therapy in patients who develop a clinically significant decrease in renal function on valsartan [see Drug Interactions (7)].
5.12 Risk of Anaphylactic Reactions
Nebivolol
While taking β-blockers, patients with a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated accidental, diagnostic, or therapeutic challenge. Such patients may be unresponsive to the usual doses of epinephrine used to treat allergic reactions.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are included in more detail in the Warnings and Precautions section of the label:
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
BYVALSON
BYVALSON has been evaluated for safety in patients with hypertension. A total of 1,664 patients received at least 1 dose of a fixed-dose combination of nebivolol/valsartan in an 8-week trial. A total of 807 patients received at least 1 dose of nebivolol and valsartan in an open-label safety study; of these, 621 patients completed 180 days and 476 patients completed 360 days of open-label treatment with nebivolol and valsartan.
The safety of the 5 mg/ 80 mg dose of nebivolol/valsartan was evaluated during the first 4 weeks of an 8-week placebo-controlled trial. During the 4 week period, the overall incidence of adverse events on therapy with BYVALSON 5 mg/ 80 mg was similar to placebo and the individual components (nebivolol 5 mg and valsartan 80 mg). Discontinuation of therapy due to a clinical adverse event occurred in 2.0% of patients treated with BYVALSON 5 mg/ 80 mg versus 3.2% of patients given placebo and approximately 1% of patients given nebivolol 5 mg or valsartan 80 mg as monotherapy.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of either nebivolol or valsartan. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Nebivolol
The following adverse reactions have been reported in post-marketing experience:
Cardiac: atrioventricular block (both second and third degree), myocardial infarction
Central Nervous System: somnolence, syncope, vertigo
Circulatory: Raynaud's phenomenon, peripheral ischemia/claudication, thrombocytopenia
Dermatologic: pruritus, psoriasis, various rashes and skin disorders
Digestive: vomiting
Hepatic: Abnormal hepatic function (including increased AST, ALT and bilirubin)
Hypersensitivity: hypersensitivity (including urticaria, allergic vasculitis and rare reports of angioedema)
Renal: acute renal failure
Respiratory: acute pulmonary edema, bronchospasm
Sexual Dysfunction: erectile dysfunction
Valsartan
The following additional adverse reactions have been reported in post-marketing experience:
Hypersensitivity: Angioedema.
Digestive: Elevated liver enzymes, hepatitis
Renal: Impaired renal function, renal failure
Clinical Laboratory Tests: Hyperkalemia
Dermatologic: Alopecia, bullous dermatitis
Blood and Lymphatic: thrombocytopenia
Vascular: Vasculitis
Related/similar drugs
7. Drug Interactions
No drug interaction studies have been conducted with BYVALSON and other drugs. Studies with the individual nebivolol and valsartan components are described below.
CYP2D6 Inhibitors: Avoid concomitant use of nebivolol with CYP2D6 inhibitors (quinidine, propafenone, fluoxetine, paroxetine, etc.) [see Clinical Pharmacology (12.3)].
Hypotensive Agents: Do not use nebivolol with other β-blockers. Closely monitor patients receiving catecholamine-depleting drugs, such as reserpine or guanethidine, because the added β-blocking action of nebivolol may produce excessive reduction of sympathetic activity. In patients who are receiving nebivolol and clonidine, discontinue nebivolol for several days before the gradual tapering of clonidine.
Digitalis Glycosides: Concomitant use can increase the risk of bradycardia. Both digitalis glycosides and β-blockers slow atrioventricular conduction and decrease heart rate. Monitor for bradycardia.
Calcium Channel Blockers: Nebivolol can exacerbate the effects of myocardial depressants or inhibitors of AV conduction, such as certain calcium antagonists (particularly of the phenylalkylamine [verapamil] and benzothiazepine [diltiazem] classes), or antiarrhythmic agents, such as disopyramide. Monitor for effects on heart rate and cardiac conduction.
Agents Increasing Serum Potassium: Concomitant use of valsartan with other agents that block the renin-angiotensin system, potassium-sparing diuretics (e.g., spironolactone, triamterene, amiloride), potassium supplements, salt substitutes containing potassium or other agents that may increase potassium levels (e.g., heparin) may result in hyperkalemia. Monitor serum potassium in such patients.
Non-Steroidal Anti-Inflammatory Agents including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors): In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, co-administration of NSAIDs, including selective COX-2 inhibitors, with angiotensin II receptor antagonists, including valsartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving valsartan and NSAID therapy.
The antihypertensive effect of angiotensin II receptor antagonists, including valsartan may be attenuated by NSAIDs including selective COX-2 inhibitors.
Use with Other Renin Angiotensin System inhibitors: Use of angiotensin receptor blockers with ACE inhibitors or with aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Most patients receiving the combination of two RAS inhibitors do not obtain any additional benefit compared to monotherapy. Closely monitor blood pressure, renal function and electrolytes in patients on valsartan and ACE inhibitors or aliskiren.
Do not co-administer aliskiren with BYVALSON in patients with diabetes. Avoid use of aliskiren with BYVALSON in patients with renal impairment (GFR <60 mL/min).
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
BYVALSON can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Most epidemiologic studies examining fetal abnormalities after exposure to angiotensin receptor blockers use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. Studies in rats and rabbits with valsartan showed fetotoxicity only at maternally toxic doses [see Data]. Embryo-fetal and perinatal lethality have been observed when nebivolol was given to pregnant rats during organogenesis at doses approximately equivalent to the MRHD. Published reports include cases of anhydramnios and oligohydramnios in pregnant women treated with valsartan. When pregnancy is detected, discontinue BYVALSON as soon as possible.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Disease-associated maternal and/or embryo/fetal risk
Hypertension in pregnancy increases the maternal risk for pre-eclampsia, gestational diabetes, premature delivery, and delivery complications (e.g., need for cesarean section, and post-partum hemorrhage). Hypertension increases the fetal risk for intrauterine growth restriction and intrauterine death. Pregnant women with hypertension should be carefully monitored and managed accordingly.
Nebivolol
Neonates of women with hypertension, who are treated with beta-blockers during pregnancy, may be at increased risk for hypotension, bradycardia, hypoglycemia, and respiratory depression. Observe newborns for symptoms of hypotension, bradycardia, hypoglycemia and respiratory depression and manage accordingly.
Valsartan
Oligohydramnios in pregnant women who use drugs affecting the renin-angiotensin system in the second and third trimesters of pregnancy can result in the following: reduced fetal renal function leading to anuria and renal failure, fetal lung hypoplasia and skeletal deformations, including skull hypoplasia, hypotension, and death. In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus.
BYVALSON
In patients taking BYVALSON during pregnancy, perform serial ultrasound examinations to assess the intra-amniotic environment. Fetal testing may be appropriate, based on the week of gestation. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to BYVALSON for hypotension, oliguria, and hyperkalemia. If oliguria or hypotension occur in neonates with a history of in utero exposure to BYVALSON, support blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and substituting for disordered renal function.
Animal Data
No reproductive animal toxicity studies have been conducted with the combination of nebivolol and valsartan. Reproductive animal toxicity studies have been conducted for nebivolol and valsartan alone.
Nebivolol
Nebivolol was shown to increase embryo-fetal and perinatal lethality in rats at approximately 1.2 times the maximum recommended human dose (MRHD) or 40 mg/day on a mg/m2 basis. Decreased pup body weights occurred at 1.25 and 2.5 mg/kg in rats, when exposed during the perinatal period (late gestation, parturition and lactation). At 5 mg/kg and higher doses (1.2 times the MRHD), prolonged gestation, dystocia and reduced maternal care were produced with corresponding increases in late fetal deaths and stillbirths and decreased birth weight, live litter size and pup survival. These events occurred only when nebivolol was given during the perinatal period (late gestation, parturition and lactation). Insufficient numbers of pups survived at 5 mg/kg to evaluate the offspring for reproductive performance.
In studies in which pregnant rats were given nebivolol during organogenesis, reduced fetal body weights were observed at maternally toxic doses of 20 and 40 mg/kg/day (5 and 10 times the MRHD), and small reversible delays in sternal and thoracic ossification associated with the reduced fetal body weights and a small increase in resorption occurred at 40 mg/kg/day (10 times the MRHD).
No adverse effects on embryo-fetal viability, sex, weight or morphology were observed in studies in which nebivolol was given to pregnant rabbits at doses as high as 20 mg/kg/day (10 times the MRHD).
Valsartan
No teratogenic effects were observed when valsartan was administered to pregnant mice and rats at oral doses up to 600 mg/kg/day and to pregnant rabbits at oral doses up to 10 mg/kg/day. However, significant decreases in fetal weight, pup birth weight, pup survival rate, and slight delays in developmental milestones were observed in studies in which parental rats were treated with valsartan at oral, maternally toxic (reduction in body weight gain and food consumption) doses of 600 mg/kg/day during organogenesis or late gestation and lactation. In rabbits, fetotoxicity (i.e., resorptions, litter loss, abortions, and low body weight) associated with maternal toxicity (mortality) was observed at doses of 5 and 10 mg/kg/day. The no observed adverse effect doses of 600, 200 and 2 mg/kg/day in mice, rats and rabbits represent 9, 6, and 0.1 times, respectively, the maximum recommended human dose on a mg/m2 basis. Calculations assume an oral dose of 320 mg/day and a 60-kg patient.
8.2 Lactation
Risk Summary
There is no information regarding the presence of BYVALSON or its individual components in human milk, the effects on the breastfed infant, or the effects on milk production. Nebivolol and valsartan are present in rat milk [see Data]. Because of the potential for β-blockers to produce serious adverse reactions in nursing infants, especially bradycardia, and the potential for valsartan to affect postnatal renal development in nursing infants, advise a nursing woman not to breastfeed during treatment with BYVALSON.
Data
In lactating rats, maximum milk levels of unchanged nebivolol were observed at 4 hours after single and repeat doses of 2.5 mg/kg/day. The daily dose (mg/kg body weight) ingested by a rat pup is 0.3% of the dam dose for unchanged nebivolol. For valsartan, drug was detected in the milk of lactating rats 15 minutes after administration of a 3 mg/kg dose.
8.5 Geriatric Use
BYVALSON
In clinical trials of BYVALSON, 204 (8.3%) patients who were treated with BYVALSON were ≥ 65 years and 16 (0.6%) were ≥ 75 years.
No overall differences in safety or effectiveness were observed between elderly patients and younger patients, and other reported clinical experience have not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
Safety and effectiveness of BYVALSON in patients with moderate and severe renal impairment (creatinine clearance [CrCl] ≤ 60 mL/min) have not been studied. No dosage adjustment is required for patients with mild or moderate renal impairment.
BYVALSON is not recommended as initial treatment in patients with severe renal impairment, because the recommended starting dose of nebivolol in this population, 2.5 mg once daily, is lower than the dose of nebivolol contained in BYVALSON.
8.7 Hepatic Impairment
There are no studies of BYVALSON in patients with hepatic insufficiency. No initial dosage adjustment is required for patients with mild hepatic impairment.
BYVALSON is not recommended as initial treatment in patients with moderate hepatic impairment, because the recommended starting dose of nebivolol, 2.5 mg once daily, is not available.
BYVALSON is not recommended for use in patients with severe hepatic impairment [see Contraindications (4)].
10. Overdosage
Nebivolol
In clinical trials and worldwide postmarketing experience there were reports of nebivolol overdose. The most common signs and symptoms associated with nebivolol overdosage are bradycardia and hypotension. Other important adverse reactions reported with nebivolol overdose include cardiac failure, dizziness, hypoglycemia, fatigue and vomiting. Other adverse reactions associated with β-blocker overdose include bronchospasm and heart block.
The largest known ingestion of nebivolol worldwide involved a patient who ingested up to 500 mg of nebivolol along with several 100 mg tablets of acetylsalicylic acid in a suicide attempt. The patient experienced hyperhydrosis, pallor, depressed level of consciousness, hypokinesia, hypotension, sinus bradycardia, hypoglycemia, hypokalemia, respiratory failure and vomiting. The patient recovered.
Because of extensive drug binding to plasma proteins, hemodialysis is not expected to enhance nebivolol clearance.
If overdose occurs, provide general supportive and specific symptomatic treatment. Based on expected pharmacologic actions and recommendations for other β-blockers, consider the following general measures, including stopping nebivolol, when clinically warranted:
Bradycardia: Administer IV atropine. If the response is inadequate, isoproterenol or another agent with positive chronotropic properties may be given cautiously. Under some circumstances, transthoracic or transvenous pacemaker placement may be necessary.
Hypotension: Administer IV fluids and vasopressors. Intravenous glucagon may be useful.
Heart Block (second or third degree): Monitor and treat with isoproterenol infusion. Under some circumstances, transthoracic or transvenous pacemaker placement may be necessary.
Congestive Heart Failure: Initiate therapy with digitalis glycoside and diuretics. In certain cases, consider the use of inotropic and vasodilating agents.
Bronchospasm: Administer bronchodilator therapy such as a short acting inhaled β2-agonist and/or aminophylline.
Hypoglycemia: Administer IV glucose. Repeated doses of IV glucose or possibly glucagon may be required.
Supportive measures should continue until clinical stability is achieved. The half-life of low doses of nebivolol is 12-19 hours.
Valsartan
Limited data are available related to overdosage in humans. The most likely manifestations of overdosage would be hypotension and tachycardia; bradycardia could occur from parasympathetic (vagal) stimulation. Depressed level of consciousness, circulatory collapse and shock have been reported. If symptomatic hypotension should occur, supportive treatment should be instituted.
Valsartan is not removed from the plasma by hemodialysis.
Valsartan was without grossly observable adverse effects at single oral doses up to 2000 mg/kg in rats and up to 1,000 mg/kg in marmosets, except for salivation and diarrhea in the rat and vomiting in the marmoset at the highest dose (60 and 31 times, respectively, the maximum recommended human dose on a mg/m2 basis). (Calculations assume an oral dose of 320 mg/day and a 60-kg patient.)
11. Byvalson Description
BYVALSON
BYVALSONTM is available as tablets for oral administration. Each tablet contains 5.45 mg of nebivolol hydrochloride, which is equivalent to 5 mg of nebivolol free base, and 80 mg of valsartan. The inactive ingredients in the tablets are lactose monohydrate, microcrystalline cellulose, copovidone, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, talc, ferric oxide, hypromellose, polysorbate 80, and Opadry® II Purple film-coat. The Opadry® II is made of polyvinyl alcohol-part hydrolyzed, titanium dioxide, talc, polyethylene glycol, iron oxide red, and ferrosoferric oxide/black iron oxide.
Nebivolol
BYVALSONTM tablets contain nebivolol hydrochloride as one of two active ingredients. Nebivolol hydrochloride is chemically described as (1RS,1'RS)-1,1'-[(2RS, 2'SR)-bis(6-fluoro-3,4-dihydro-2H-1-benzopyran-2-yl)]-2,2'-iminodiethanol hydrochloride. Nebivolol hydrochloride is present as a racemic mixture of d-Nebivolol hydrochloride and l-Nebivolol hydrochloride with the stereochemical designations of [SRRR]-nebivolol hydrochloride and [RSSS]-nebivolol hydrochloride, respectively. The molecular formula of both the forms is C22H25F2NO4•HCl and their structural formulas are provided below:
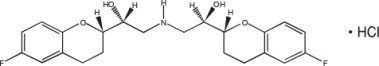
(SRRR) - or d-nebivolol hydrochloride
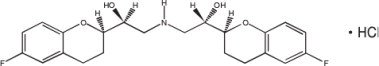
(RSSS) - or l-nebivolol hydrochloride
MW: 441.90 g/mol
Nebivolol hydrochloride is a white to almost white powder that is soluble in methanol, dimethylsulfoxide, and N,N-dimethylformamide, sparingly soluble in ethanol, propylene glycol, and polyethylene glycol, and very slightly soluble in hexane, dichloromethane, and methylbenzene.
Valsartan
Valsartan, the other active ingredient in BYVALSON, is chemically described as N-(1-Oxopentyl)-N-[[2´-(1H-tetrazol-5-yl) [1,1´-biphenyl]-4-yl]methyl]-L-valine. Its molecular formula is C24H29N5O3, its molecular weight is 435.52, and its structural formula is:
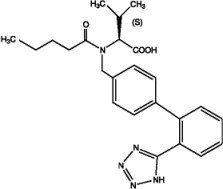
Valsartan is a white or almost white fine powder. It is freely soluble in anhydrous ethanol, sparingly soluble in methylene chloride, and practically insoluble in water.
12. Byvalson - Clinical Pharmacology
12.1 Mechanism of Action
Nebivolol
Nebivolol is a β-adrenergic receptor blocking agent. In extensive metabolizers (most of the population) and at doses less than or equal to 10 mg, nebivolol is preferentially β1 selective. In poor metabolizers and at higher doses, nebivolol inhibits both β1- and β2- adrenergic receptors. Nebivolol lacks intrinsic sympathomimetic and membrane stabilizing activity at therapeutically relevant concentrations. At clinically relevant doses, nebivolol does not demonstrate α1-adrenergic receptor blockade activity. Various metabolites, including glucuronides, contribute to β-blocking activity.
The mechanism of action of the antihypertensive response of nebivolol has not been definitively established. Possible factors that may be involved include: (1) decreased heart rate, (2) decreased myocardial contractility, (3) decreased sympathetic activity, (4) suppression of renin activity, and (5) vasodilation and decreased peripheral vascular resistance.
Valsartan
Angiotensin II is formed from angiotensin I in a reaction catalyzed by angiotensin-converting enzyme (ACE, kininase II). Angiotensin II is the principal pressor agent of the renin-angiotensin system, with effects that include vasoconstriction, stimulation of synthesis and release of aldosterone, cardiac stimulation, and renal reabsorption of sodium. Valsartan blocks the vasoconstrictor and aldosterone-secreting effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor in many tissues, such as vascular smooth muscle and the adrenal gland. Its action is therefore independent of the pathways for angiotensin II synthesis.
There is also an AT2 receptor found in many tissues, but AT2 is not known to be associated with cardiovascular homeostasis. Valsartan has much greater affinity (about 20,000-fold) for the AT1 receptor than for the AT2 receptor. The increased plasma levels of angiotensin II following AT1 receptor blockade with valsartan may stimulate the unblocked AT2 receptor. The primary metabolite of valsartan is essentially inactive with an affinity for the AT1 receptor about one- 200th that of valsartan itself.
Blockade of the renin-angiotensin system with ACE inhibitors, which inhibit the biosynthesis of angiotensin II from angiotensin I, is widely used in the treatment of hypertension. ACE inhibitors also inhibit the degradation of bradykinin, a reaction also catalyzed by ACE. Because valsartan does not inhibit ACE (kininase II), it does not affect the response to bradykinin. Whether this difference has clinical relevance is not yet known. Valsartan does not bind to or block other hormone receptors or ion channels known to be important in cardiovascular regulation.
Blockade of the angiotensin II receptor inhibits the negative regulatory feedback of angiotensin II on renin secretion, but the resulting increased plasma renin activity and angiotensin II circulating levels do not overcome the effect of valsartan on blood pressure.
12.2 Pharmacodynamics
BYVALSON
In a placebo-controlled hypertensive patient study, valsartan was associated with an increase in PRA (60-73% increase) whereas nebivolol was associated with a 51-65% reduction in PRA. Nebivolol in combination with valsartan reduced PRA (3-39% reduction). Nebivolol, valsartan, and BYVALSON decreased plasma aldosterone levels. Administration of BYVALSON to patients with essential hypertension results in a significant reduction of sitting, and standing diastolic and systolic blood pressure. Decreases in pulse rate from baseline were also observed in the BYVALSON and nebivolol treatment groups.
12.3 Pharmacokinetics
BYVALSON
Following oral administration of BYVALSON, peak plasma nebivolol concentrations are reached approximately 1 to 6 hours post-dosing. Peak plasma valsartan concentrations are reached in approximately 2 to 4 hours post-dosing. The rate and extent of absorption of nebivolol and valsartan from BYVALSON are the same as when administered separately. Food had a minor impact on the pharmacokinetics of nebivolol, nebivolol glucuronides and valsartan. BYVALSON may be administered without regard to meals. BYVALSON demonstrates dose-proportional increases in exposure over the clinical dosing range.
Nebivolol
Exposure to l-nebivolol is higher than to d-nebivolol but l-nebivolol contributes little to the drug's β-blocking activity as d-nebivolol's β-receptor affinity is > 1,000-fold higher than l-nebivolol.
For the same dose, CYP2D6 poor metabolizers (PMs) attain a 5-fold higher Cmax and 10-fold higher AUC of nebivolol than do extensive metabolizers (EMs). With repeated once-daily dosing of BYVALSON, nebivolol accumulates about 1.2-fold in EMs and 3-fold in PMs, respectively; valsartan accumulates about 1.5-fold.
The in vitro human plasma protein binding of nebivolol is approximately 98%, mostly to albumin, and is independent of nebivolol concentrations.
Valsartan
The steady state volume of distribution of valsartan after intravenous administration is 17 L indicating that valsartan does not distribute into tissues extensively. Valsartan is highly bound to serum proteins (95%), mainly serum albumin.
Nebivolol
Nebivolol is predominantly metabolized via direct glucuronidation of parent and to a lesser extent via N-dealkylation and oxidation via cytochrome P450 2D6. d-Nebivolol, has an effective half-life of about 12 hours in CYP2D6 EMs, and 19 hours in PMs and exposure to d-nebivolol is substantially increased in PMs. The difference in d-nebivolol exposure between EMs and PMs is not considered important because nebivolol metabolites, including the hydroxyl metabolite and glucuronides (the prominent circulating metabolites) contribute to the pharmacologic activity.
After a single oral administration of 14C-nebivolol, 38% of the dose was recovered in urine and 44% in feces for EMs and 67% in urine and 13% in feces for PMs. Essentially all nebivolol was excreted as multiple oxidative metabolites or their corresponding glucuronide conjugates.
Valsartan
Valsartan, when administered as an oral solution, is primarily recovered in feces (about 83% of dose) and urine (about 13% of dose). Valsartan shows bi-exponential decay kinetics following intravenous administration with an average elimination half-life of about 6 hours. The recovery is mainly as unchanged drug, with only about 20% of dose recovered as metabolites. The primary metabolite, accounting for about 9% of dose, is valeryl-4-hydroxy valsartan. In vitro metabolism studies involving recombinant CYP450 enzymes indicated that the CYP2C9 isozyme is responsible for the formation of valeryl-4-hydroxy valsartan. Valsartan does not inhibit CYP450 isozymes at clinically relevant concentrations. CYP450 mediated drug interactions between valsartan and co-administered drugs are unlikely because of the low extent of metabolism.
Following intravenous administration, plasma clearance of valsartan is about 2 L/h and its renal clearance is 0.62 L/h (about 30% of total clearance).
Nebivolol
d-Nebivolol peak plasma concentration increased 3-fold, exposure (AUC) increased 10-fold, and the apparent clearance decreased by 86% in patients with moderate hepatic impairment (Child-Pugh Class B). No formal studies have been performed in patients with severe hepatic impairment; nebivolol is contraindicated for these patients [see Use in Specific Population (8.7)].
Valsartan
On average, patients with mild-to-moderate chronic liver disease have twice the exposure (measured by AUC values) to valsartan of healthy volunteers (matched by age, sex and weight). In general, no dosage adjustment is needed in patients with mild-to-moderate liver disease.
Renal Impairment
Nebivolol
The apparent clearance of nebivolol was unchanged following a single 5 mg dose of nebivolol in patients with mild renal impairment (CrCl 50 to 80 mL/min, n=7), and it was reduced negligibly in patients with moderate renal impairment (CrCl 30 to 50 mL/min, n=9), but clearance was reduced by 53% in patients with severe renal impairment (CrCl <30 mL/min, n=5). No studies have been conducted in patients on dialysis [see Use in Specific Population (8.6)].
Valsartan
There is no apparent correlation between renal function (measured by CrCl) and exposure (measured by AUC) to valsartan in patients with different degrees of renal impairment. Consequently, dose adjustment is not required in patients with mild-to-moderate renal dysfunction. No studies have been performed in patients with severe impairment of renal function (CrCl <10 mL/min). Valsartan is not removed from the plasma by hemodialysis [see Use in Specific Population (8.6)].
Nebivolol
Drugs that inhibit CYP2D6 can be expected to increase plasma levels of nebivolol. When nebivolol is co-administered with an inhibitor or an inducer of this enzyme, monitor patients closely and adjust the nebivolol dose according to blood pressure response. In vitro studies have demonstrated that at therapeutically relevant concentrations, d- and l-nebivolol do not inhibit any cytochrome P450 pathways.
Fluoxetine: Fluoxetine, a CYP2D6 inhibitor, administered at 20 mg per day for 21 days prior to a single 10 mg dose of nebivolol to 10 healthy adults, led to an 8-fold increase in the AUC and 3-fold increase in Cmax for d-nebivolol [see Drug Interactions (7)].
Digoxin: Concomitant administration of nebivolol (10 mg once daily) and digoxin (0.25 mg once daily) for 10 days in 14 healthy adult individuals resulted in no significant changes in the pharmacokinetics of digoxin or nebivolol [see Drug Interactions (7)].
Warfarin: Administration of nebivolol (10 mg once daily for 10 days) led to no significant changes in the pharmacokinetics of nebivolol or R- or S-warfarin following a single 10 mg dose of warfarin. Similarly, nebivolol has no significant effects on the anticoagulant activity of warfarin, as assessed by Prothrombin time and INR profiles from 0 to 144 hours after a single 10 mg warfarin dose in 12 healthy adult volunteers.
Diuretics: No pharmacokinetic interactions were observed in healthy adults between nebivolol (10 mg daily for 10 days) and furosemide (40 mg single dose), hydrochlorothiazide (25 mg once daily for 10 days), or spironolactone (25 mg once daily for 10 days).
Ramipril: Concomitant administration of nebivolol (10 mg once daily) and ramipril (5 mg once daily) for 10 days in 15 healthy adult volunteers produced no pharmacokinetic interactions.
Losartan: Concomitant administration of nebivolol (10 mg single dose) and losartan (50 mg single dose) in 20 healthy adult volunteers did not result in pharmacokinetic interactions.
Valsartan: Concomitant administration of nebivolol (20 mg once daily) and valsartan (320 mg once daily) in 30 healthy adult volunteers did not result in clinically significant pharmacokinetic interactions.
Histamine-2 Receptor Antagonists: The pharmacokinetics of nebivolol (5 mg single dose) were not affected by the co-administration of ranitidine (150 mg twice daily). Cimetidine (400 mg twice daily) causes a 23% increase in the plasma levels of d-nebivolol.
Charcoal: The pharmacokinetics of nebivolol (10 mg single dose) were not affected by repeated co-administration (4, 8, 12, 16, 22, 28, 36, and 48 hours after nebivolol administration) of activated charcoal (Actidose-Aqua®).
Sildenafil: The co-administration of nebivolol and sildenafil decreased AUC and Cmax of sildenafil by 21 and 23% respectively. The effect on the Cmax and AUC for d-nebivolol was also small (< 20%). The effect on vital signs (e.g., pulse and blood pressure) was approximately the sum of the effects of sildenafil and nebivolol.
Other Concomitant Medications: Utilizing population pharmacokinetic analyses using data derived from hypertensive patients, the following drugs are not expected to have an effect on the pharmacokinetics of nebivolol: acetaminophen, acetylsalicylic acid, atorvastatin, esomeprazole, ibuprofen, levothyroxine sodium, metformin, sildenafil, simvastatin, or tocopherol.
Protein Binding: No meaningful changes in the extent of in vitro binding of nebivolol to human plasma proteins were noted in the presence of high concentrations of diazepam, digoxin, diphenylhydantoin, enalapril, hydrochlorothiazide, imipramine, indomethacin, propranolol, sulfamethazine, tolbutamide, or warfarin. Additionally, nebivolol did not significantly alter the protein binding of the following drugs: diazepam, digoxin, diphenylhydantoin, hydrochlorothiazide, imipramine, or warfarin at their therapeutic concentrations.
Valsartan
No clinically significant pharmacokinetic interactions were observed when valsartan was co-administered with nebivolol, amlodipine, atenolol, cimetidine, digoxin, furosemide, glyburide, hydrochlorothiazide, or indomethacin.
Co-administration of valsartan and warfarin did not change the pharmacokinetics of valsartan or the time-course of the anticoagulant properties of warfarin.
Transporters: The results from an in vitro study with human liver tissue indicate that valsartan is a substrate of the hepatic uptake transporter OATP1B1 and the hepatic efflux transporter MRP2. Co-administration of inhibitors of the uptake transporter (rifampin, cyclosporine) or efflux transporter (ritonavir) may increase the systemic exposure to valsartan.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Nebivolol
In a two-year study of nebivolol in mice, a statistically significant increase in the incidence of testicular Leydig cell hyperplasia and adenomas was observed at 40 mg/kg/day (5 times the maximally recommended human dose of 40 mg on a mg/m2 basis). Similar findings were not reported in mice administered doses equal to approximately 0.3 or 1.2 times the maximum recommended human dose. No evidence of a tumorigenic effect was observed in a 24-month study in Wistar rats receiving doses of nebivolol 2.5 mg/kg/day, 10 mg/kg/day and 40 mg/kg/day (equivalent to 0.6, 2.4, and 10 times the maximally recommended human dose). Co-administration of dihydrotestosterone reduced blood LH levels and prevented the Leydig cell hyperplasia, consistent with an indirect LH-mediated effect of nebivolol in mice and not thought to be clinically relevant in man.
A randomized, double-blind, placebo- and active-controlled, parallel-group study in healthy male volunteers was conducted to determine the effects of nebivolol on adrenal function, luteinizing hormone, and testosterone levels. This study demonstrated that 6 weeks of daily dosing with 10 mg of nebivolol had no significant effect on ACTH-stimulated mean serum cortisol AUC0-120 min, serum LH, or serum total testosterone.
Effects on spermatogenesis were seen in male rats and mice at ≥ 40 mg/kg/day (10 and 5 times the MRHD, respectively). For rats the effects on spermatogenesis were not reversed and may have worsened during a four week recovery period. The effects of nebivolol on sperm in mice, however, were partially reversible.
Mutagenesis: Nebivolol was not genotoxic when tested in a battery of assays (Ames, in vitro mouse lymphoma TK+/-, in vitro human peripheral lymphocyte chromosome aberration, in vivo Drosophila melanogaster sex-linked recessive lethal, and in vivo mouse bone marrow micronucleus tests).
Valsartan
There was no evidence of carcinogenicity when valsartan was administered in the diet to mice and rats for up to 2 years at doses up to 160 and 200 mg/kg/day, respectively. These doses in mice and rats are about 2.6 and 6 times, respectively, the maximum recommended human dose on a mg/m2 basis. (Calculations assume an oral dose of 320 mg/day and a 60-kg patient.)
Mutagenicity assays did not reveal any valsartan-related effects at either the gene or chromosome level. These assays included bacterial mutagenicity tests with Salmonella (Ames) and E coli; a gene mutation test with Chinese hamster V79 cells; a cytogenetic test with Chinese hamster ovary cells; and a rat micronucleus test.
Valsartan had no adverse effects on the reproductive performance of male or female rats at oral doses up to 200 mg/kg/day. This dose is 6 times the maximum recommended human dose on a mg/m2 basis. (Calculations assume an oral dose of 320 mg/day and a 60-kg patient.)
14. Clinical Studies
BYVALSON was studied in a Phase 3, double-blind, placebo-controlled, dose-escalating, 8-week study in 4,161 patients with Stage 1 or 2 hypertension. Patients were initially randomized to 1 of 8 treatment groups including: 3 fixed-dose combinations (FDC) of nebivolol and valsartan (5 mg/ 80 mg, 5 mg/ 160 mg, 10 mg/ 160 mg), nebivolol monotherapy (5 mg, 20 mg), valsartan monotherapy (80 mg, 160 mg), or placebo. After 4 weeks of treatment, all doses were doubled in the FDC groups (to 10 mg/ 160 mg, 10 mg/ 320 mg, and 20 mg/ 320 mg), nebivolol monotherapy groups (to 10 mg, 40 mg), and valsartan monotherapy groups (to 160 mg, 320 mg).
Mean baseline systolic blood pressure was 155 mm Hg and mean baseline diastolic blood pressure was 100 mm Hg. Approximately 56% of patients were male, 85% were Caucasian, 10% were Black, 41% were Hispanic, 15% were diabetic, and 9% were 65 years of age or older.
Treatment with BYVALSON 5 mg/ 80 mg for 4 weeks resulted in placebo-adjusted reductions from baseline in systolic (SBP) and diastolic (DBP) blood pressure of -8.3 and -7.2 mmHg, respectively. Treatment with BYVALSON 5 mg/ 80 mg resulted in greater reductions in SBP and DBP than did treatment with nebivolol 5 mg alone (p<0.0001 for both SBP and DBP) or valsartan 80 mg, alone (p=0.0007 for SBP and p<0.0001 for DBP).
| Change From Baseline | Byvalson 5mg/ 80mg
mmHg (95% CI) | Valsartan 80 mg
mmHg (95% CI) | Valsartan 160 mg
mmHg (95% CI) | Nebivolol
5 mg mmHg (95% CI) | Nebivolol
20 mg mmHg (95% CI) | Byvalson 5mg/160mg
mmHg (95% CI) | Byvalson 10mg/160mg
mmHg (95% CI) |
| DBP | -7.2 (–8.4, –5.9) | -3.9 (–5.1, –2.6) | -4.6 (–5.8, –3.3) | -4.4 (–5.7, –3.2) | -7.5 (–8.7, –6.2) | -7.4 (–8.7, –6.1) | -7.9 (–9.2, –6.6) |
| SBP | -8.3 (–10.3, –6.3) | -5.4 (–7.4, –3.4) | -7.5 (–9.5, –5.4) | -4.7 (–6.7, –2.7) | -7.4 (–9.4, –5.4) | -8.8 (–10.9, –6.8) | -9.0 (–11.0, –7.0) |
The antihypertensive effect of valsartan and nebivolol was independent of age and gender in clinical trials evaluating the antihypertensive effect of nebivolol and valsartan administered as monotherapies. Antihypertensive drugs that affect the renin-angiotensin system such as angiotensin converting enzyme inhibitors and angiotensin-II blockers have generally been found to be less effective in low-renin hypertensive patients (frequently Blacks) as compared to high-renin hypertensive patients (frequently whites). Although the effectiveness of nebivolol as a monotherapy has been established in Blacks, as a monotherapy, the magnitude of nebivolol's effect in Blacks was somewhat less than in Caucasians.
The blood pressure lowering effect of BYVALSON was maintained over the 24-hour dosing interval based on Ambulatory Blood Pressure Monitoring (ABPM) assessments. Mean heart rate (assessed as a part of 24-hour measurements or at sitting-trough in the 8-week or the 52-week studies) was reduced in patients groups treated with BYVALSON.
16. How is Byvalson supplied
BYVALSON tablets are supplied in the following strengths and package configurations:
| BYVALSON | ||||
| Tablet Strength | Package Configuration | NDC # | Tablet Color/Shape | Tablet Marking |
| 5 mg nebivolol/ 80 mg valsartan per tablet | Bottle of 30 | 0023-5874-30 | Purple / capsule shaped | FL1 on one side |
| Bottle of 90 | 0023-5874-90 | |||
Store at 20° to 25°C (68° to 77°F) [see USP for Controlled Room Temperature].
Dispense in a tightly closed container.
17. Patient Counseling Information
Fetal Toxicity
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to notify their healthcare provider with a known or suspected pregnancy [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed during treatment with BYVALSON [see Use in Specific Populations (8.2)].
Symptomatic Hypotension
Advise patients that lightheadedness can occur, especially during the first days of therapy, and that it should be reported to the prescribing physician. Tell patients that if syncope occurs to discontinue BYVALSON until the physician has been consulted.
Caution all patients that inadequate fluid intake, excessive perspiration, diarrhea, or vomiting can lead to an excessive fall in blood pressure, with the same consequences of lightheadedness and possible syncope.
Hyperkalemia
Advise patents not to use salt substitutes containing potassium without consulting their physician.
Distributed by:
Allergan USA, Inc.
Irvine, CA 92612
Actidose-Aqua® is a registered trademark of Paddock Laboratories, Inc. All other trademarks herein are the property of respective owners.
© 2016 Allergan. All rights reserved.
|
This Patient Information has been approved by the U.S. Food and Drug Administration |
Issued: 06/2016 |
| PATIENT INFORMATION
BYVALSON (bye-VAL-son) (nebivolol/valsartan) tablets |
|
|
What is the most important information I should know about BYVALSON?
|
|
| What is BYVALSON?
BYVALSON is a prescription medicine that contains nebivolol, a beta-blocker and valsartan, an angiotensin receptor blocker (ARB). BYVALSON is used to treat high blood pressure (hypertension) in adults. It is not known if BYVALSON is safe and effective in children. |
|
| Who should not take BYVALSON?
Do not take BYVALSON if you:
|
|
| What should I tell my healthcare provider before taking BYVALSON?
Before taking BYVALSON, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking BYVALSON with certain other medicines can affect each other and cause serious side effects. Especially tell your healthcare provider if you take:
|
|
| Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. | |
|
How should I take BYVALSON?
|
|
| What are the possible side effects of BYVALSON?
BYVALSON may cause serious side effects, including:
|
|
|
|
Stop taking BYVALSON, lie down and call your healthcare provider right away if you feel faint or dizzy.
|
|
|
|
|
|
| The most common side effect of BYVALSON is: slow heartbeat Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects with BYVALSON. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store BYVALSON?
|
|
| Keep BYVALSON and all medicines out of the reach of children. |
|
| General information about the safe and effective use of BYVALSON.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use BYVALSON for a condition for which it was not prescribed. Do not give BYVALSON to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about BYVALSON. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about BYVALSON that is written for healthcare professionals. |
|
| What is high blood pressure (hypertension)?
Blood pressure is the force in your blood vessels when your heart beats and when your heart rests. You have high blood pressure when the force is too great. High blood pressure makes the heart work harder to pump blood through the body and causes damage to the blood vessels. BYVALSON can help your blood vessels relax so your blood pressure is lower. Medicines that lower your blood pressure lower your chance of having a stroke or heart attack. |
|
| What are the ingredients in BYVALSON?
Active Ingredients: Nebivolol as hydrochloride salt and Valsartan Inactive Ingredients: Lactose monohydrate, microcrystalline cellulose, copovidone, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, talc, ferric oxide, hypromellose, polysorbate 80, and Opadry® II Purple film-coat. The Opadry® II is made of polyvinyl alcohol-part hydrolyzed, titanium dioxide, talc, polyethylene glycol, iron oxide red, and ferrosoferric oxide/black iron oxide. Distributed by: Allergan USA, Inc., Irvine, CA 92612. Actidose-Aqua® is a registered trademark of Paddock Laboratories, Inc. All other trademarks herein are the property of respective owners, © 2016 Allergan. All rights reserved. For more information, go to www.BYVALSON.com or call 1-800-433-8871. |
|
| BYVALSON
nebivolol hydrochloride and valsartan tablet, film coated |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Allergan, Inc. (144796497) |
Frequently asked questions
More about Byvalson (nebivolol / valsartan)
- Check interactions
- Compare alternatives
- Imprints, shape & color data
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: miscellaneous antihypertensive combinations
- En español

