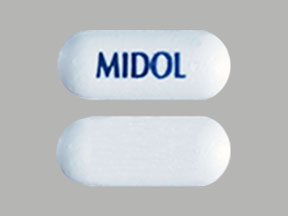
MIDOL Pill: blue, capsule/oblong
Generic Name: ibuprofen
The pill with imprint MIDOL (Blue, Capsule/Oblong, 0mm) has been identified as Midol IB ibuprofen 200 mg and is used for Back Pain, Aseptic Necrosis, Pain, Headache, and Fever. It belongs to the drug class Nonsteroidal anti-inflammatory drugs and is not a controlled substance.
Images for MIDOL
Midol IB
- Generic Name
- ibuprofen
- Imprint
- MIDOL
- Strength
- ibuprofen 200 mg
- Color
- Blue
- Shape
- Capsule/Oblong
- Availability
- Rx and/or OTC
- Drug Class
- Nonsteroidal anti-inflammatory drugs
- Pregnancy Category
- C - Risk cannot be ruled out - prior to 30 weeks gestation, D - Positive evidence of risk - starting at 30 weeks gestation
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Bayer HealthCare LLC - Consumer Care
- National Drug Code (NDC)
- 00280-8080
- Inactive Ingredients
-
FD&C Blue No. 1,
gelatin,
hypromelloses,
mannitol,
polyethylene glycol,
povidone k30,
water,
sorbitan,
sorbitol,
titanium dioxide,
alpha-tocopherol
Note: Inactive ingredients may vary.
Related images for "MIDOL"
More about Midol IB (ibuprofen)
- Check interactions
- Compare alternatives
- Imprints, shape & color data
- Latest FDA alerts (14)
- Side effects
- Dosage information
- During pregnancy
- Drug class: Nonsteroidal anti-inflammatory drugs
- Breastfeeding
Patient resources
Other brands
Advil, IBU, A-G Profen, Addaprin, ... +19 more
Professional resources
Other brands
Advil, Motrin, IBU, Advil Liqui-Gels, ... +8 more
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.