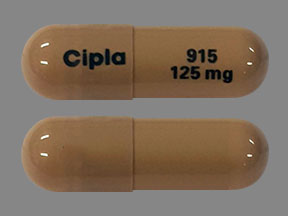
Cipla 915 125 mg Pill: brown, capsule/oblong, 22mm
The pill with imprint Cipla 915 125 mg (Brown, Capsule/Oblong, 22mm) has been identified as Flutamide 125 mg and is used for Prostate Cancer, and Hirsutism. It belongs to the drug classes antiandrogens, hormones/antineoplastics and is not a controlled substance.
Images for Cipla 915 125 mg
Flutamide
- Imprint
- Cipla 915 125 mg
- Strength
- 125 mg
- Color
- Brown
- Size
- 22.00 mm
- Shape
- Capsule/Oblong
- Availability
- Prescription only
- Drug Class
- Antiandrogens, Hormones/antineoplastics
- Pregnancy Category
- D - Positive evidence of risk
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Cipla USA Inc.
- National Drug Code (NDC)
- 69097-0915
- Inactive Ingredients
-
ferrosoferric oxide,
corn starch,
D&C Yellow No. 10,
FD&C Blue No. 1,
FD&C Blue No. 2,
FD&C Red No. 40,
aluminum oxide,
gelatin,
lactose monohydrate,
magnesium stearate,
ferric oxide red,
sodium lauryl sulfate,
titanium dioxide,
ferric oxide yellow
Note: Inactive ingredients may vary.
See also:
More about flutamide
- Check interactions
- Compare alternatives
- Pricing & coupons
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: antiandrogens
- En español
Patient resources
Other brands
Professional resources
Other brands
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
