Solu-Cortef Dosage
Generic name: HYDROCORTISONE SODIUM SUCCINATE 100mg in 2mL
Dosage form: injection, powder, for solution
Drug class: Glucocorticoids
Medically reviewed by Drugs.com. Last updated on Sep 5, 2025.
Because of possible physical incompatibilities, SOLU-CORTEF should not be diluted or mixed with other solutions.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
This preparation may be administered by intravenous injection, by intravenous infusion, or by intramuscular injection, the preferred method for initial emergency use being intravenous injection. Following the initial emergency period, consideration should be given to employing a longer acting injectable preparation or an oral preparation.
Therapy is initiated by administering SOLU-CORTEF Sterile Powder intravenously over a period of 30 seconds (e.g., 100 mg) to 10 minutes (e.g., 500 mg or more). In general, high dose corticosteroid therapy should be continued only until the patient's condition has stabilized, usually not beyond 48 hours to 72 hours. When high dose hydrocortisone therapy must be continued beyond 48 –72 hours, hypernatremia may occur. Under such circumstances, it may be desirable to replace SOLU-CORTEF with a corticoid such as methylprednisolone sodium succinate which causes little or no sodium retention.
The initial dose of SOLU-CORTEF Sterile Powder is 100 mg to 500 mg, depending on the specific disease entity being treated. However, in certain overwhelming, acute, life-threatening situations, administration in dosages exceeding the usual dosages may be justified and may be in multiples of the oral dosages.
This dose may be repeated at intervals of 2, 4, or 6 hours as indicated by the patient's response and clinical condition.
It Should Be Emphasized that Dosage Requirements Are Variable and Must Be Individualized on the Basis of the Disease Under Treatment and the Response of the Patient. After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage that maintains an adequate clinical response is reached. Situations that may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient's individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this latter situation, it may be necessary to increase the dosage of the corticosteroid for a period of time consistent with the patient's condition. If after long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
In pediatric patients, the initial dose of hydrocortisone may vary depending on the specific disease entity being treated. The range of initial doses is 0.56 mg/kg/day to 8 mg/kg/day in three or four divided doses (20 mg/m2bsa/day to 240 mg/m2bsa/day). For the purpose of comparison, the following is the equivalent milligram dosage of the various glucocorticoids:
|
Cortisone, 25 |
Triamcinolone, 4 |
|
Hydrocortisone, 20 |
Paramethasone, 2 |
|
Prednisolone, 5 |
Betamethasone, 0.75 |
|
Prednisone, 5 |
Dexamethasone, 0.75 |
|
Methylprednisolone, 4 |
These dose relationships apply only to oral or intravenous administration of these compounds. When these substances or their derivatives are injected intramuscularly or into joint spaces, their relative properties may be greatly altered.
Preparation of Solutions:
100 mg Plain
For intravenous or intramuscular injection, prepare solution by aseptically adding not more than 2 mL of Bacteriostatic Water for Injection or Bacteriostatic Sodium Chloride Injection to the contents of one vial. For intravenous infusion, first prepare solution by adding not more than 2 mL of Bacteriostatic Water for Injection to the vial; this solution may then be added to 100 mL to 1,000 mL of the following: 5% dextrose in water (or isotonic saline solution or 5% dextrose in isotonic saline solution if patient is not on sodium restriction). From a microbiological point of view, unless the method of opening/reconstitution/dilution precludes the risk of microbial contamination, the product should be used immediately. If not used immediately, in use storage times and conditions are the responsibility of the user.
This product, like many other steroid formulations, is sensitive to heat. Therefore, it should not be autoclaved when it is desirable to sterilize the exterior of the vial.
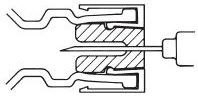
DIRECTIONS FOR USING THE ACT-O-VIAL SYSTEM
- 1.
- Press down on plastic activator to force diluent into the lower compartment.
- 2.
- Gently agitate to effect solution.
- 3.
- Remove plastic tab covering center of stopper.
- 4.
- Sterilize top of stopper with a suitable germicide.
- 5.
- Insert needle squarely through center of stopper until tip is just visible. Invert vial and withdraw dose.
Further dilution is not necessary for intravenous or intramuscular injection. For intravenous infusion, first prepare solution as just described. The 100 mg solution may then be added to 100 mL to 1,000 mL of 5% dextrose in water (or isotonic saline solution or 5% dextrose in isotonic saline solution if patient is not on sodium restriction). The 250 mg solution may be added to 250 mL to 1,000 mL, the 500 mg solution may be added to 500 mL to 1,000 mL, and the 1,000 mg solution to 1,000 mL of the same diluents. In cases where administration of a small volume of fluid is desirable, 100 mg to 3,000 mg of SOLU-CORTEF may be added to 50 mL of the above diluents. The resulting solutions are stable for at least 4 hours and may be administered either directly or by IV piggyback.
When reconstituted as directed, pH's of the solutions range from 7 to 8.
Frequently asked questions
- Can you use hydrocortisone cream for vulvar itching?
- Can I use hydrocortisone cream on shingles rash?
- What are steroid injections (cortisone shots)?
- Can you put hydrocortisone cream on hemorrhoids?
- What is the difference between hydrocortisone and cortisone?
- Can hydrocortisone cream be used on sunburn?
- How long does it take for neomycin, polymyxin b and hydrocortisone ear drops to work?
- Can you use hydrocortisone cream on babies?
- Does hydrocortisone cream stop itching?
More about Solu-Cortef (hydrocortisone)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- During pregnancy
- Generic availability
- Drug class: glucocorticoids
- Breastfeeding
- En español
Patient resources
Other brands
Cortenema, Alkindi Sprinkle, Cortifoam, A-Hydrocort
Professional resources
- Solu-Cortef prescribing information
- Hydrocortisone, Hydrocortisone Sodium Succinate (AHFS Monograph)
Other brands
Khindivi, Cortenema, Alkindi Sprinkle, A-Hydrocort, Colocort
Other formulations
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.