Provenge Dosage
Generic name: SIPULEUCEL-T 500000001
Dosage form: injection
Drug class: Therapeutic vaccines
Medically reviewed by Drugs.com. Last updated on Oct 10, 2024.
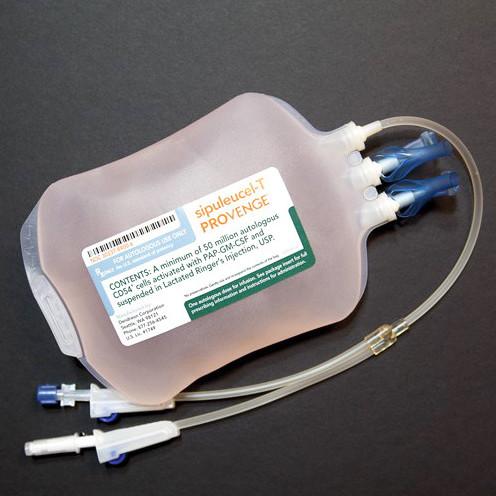
For autologous use only.
For intravenous use only.
Dose
Each dose of PROVENGE contains a minimum of 50 million autologous CD54 + cells activated with PAP-GM-CSF [ see Description ( 11) ].
The recommended course of therapy for PROVENGE is 3 complete doses, given at approximately 2-week intervals. In controlled clinical trials, the median dosing interval between infusions was 2 weeks (range 1 to 15 weeks); the maximum dosing interval has not been established.
If, for any reason, the patient is unable to receive a scheduled infusion of PROVENGE, the patient will need to undergo an additional leukapheresis procedure prior to continuing a course of treatment. Advise patients of this possibility prior to initiating treatment.
Administration
- Do not use PROVENGE until confirmation of product release is received from Dendreon.
- Dendreon will send the Final Product Disposition Notification form containing the patient identifiers, expiration date and time, and the disposition status (approved for infusion or rejected), to the infusion site. Infusion must begin prior to the expiration date and time indicated on the Final Product Disposition Notification form and Product Label. Do not use expired PROVENGE. Keep the sealed, patient-specific PROVENGE infusion bag within the insulated polyurethane container inside the outer cardboard shipping box until the time of administration.
- To minimize potential acute infusion reactions, premedicate the patients orally with acetaminophen and an antihistamine, such as diphenhydramine, approximately 30 minutes prior to administration of PROVENGE.
Administration steps:
- Remove the infusion bag from the insulated polyurethane container and inspect the bag for signs of leakage or external damage. Contents of the bag will be clear to opaque, with a white to red color, including shades of off-white, cream, light yellow and orange. Remove the infusion bag from the insulated polyurethane container and inspect the bag for signs of leakage or external damage. Contents of the bag will be clear to opaque, with a white to red color, including shades of off-white, cream, light yellow and orange.
- Gently mix and resuspend the contents of the bag, inspecting for clumps and clots. Small clumps of cellular material should disperse with gentle manual mixing. Do not administer if the bag leaks during handling, is damaged, or if clumps remain in the bag.
- Match the patient's identity with the patient identifiers on the Final Product Disposition Notification form and the PROVENGE infusion bag.
- Infuse the entire volume of the PROVENGE infusion bag intravenously over approximately 60 minutes. Do not use a cell filter.
- Observe the patient for acute infusion reactions for at least 30 minutes following each infusion.
- If acute infusion reactions occur, such as chills, fatigue, fever, nausea, and joint ache, interrupt or slow the infusion and administer appropriate medical treatment as needed. In controlled clinical trials, symptoms of acute infusion reactions were treated with acetaminophen, intravenous H1 and/or H2 blockers, and low-dose intravenous meperidine.
- If the infusion is interrupted, keep the PROVENGE infusion bag at room temperature.
- Do not resume infusion if the PROVENGE infusion bag has been at room temperature for more than 3 hours.
More about Provenge (sipuleucel-T)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (6)
- Drug images
- Side effects
- During pregnancy
- FDA approval history
- Drug class: therapeutic vaccines
- En español
Patient resources
Professional resources
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.