Utibron Neohaler: Package Insert / Prescribing Info
Package insert / product label
Generic name: indacaterol and glycopyrrolate
Dosage form: inhalation capsule
Drug class: Bronchodilator combinations
Medically reviewed by Drugs.com. Last updated on May 19, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
UTIBRON NEOHALER safely and effectively. See full prescribing information for UTIBRON NEOHALER.
UTIBRON® NEOHALER® (indacaterol and glycopyrrolate) inhalation powder, for oral inhalation use
Initial U.S. Approval: 2015
Recent Major Changes
Indications and Usage for Utibron Neohaler
UTIBRON NEOHALER is a combination of indacaterol, a long-acting beta2-adrenergic agonist (LABA), and glycopyrrolate, an anticholinergic, indicated for the long-term, maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD). (1)
Limitations of Use: Not indicated for the relief of acute bronchospasm or for the treatment of asthma. (1, 5.1, 5.2)
Utibron Neohaler Dosage and Administration
Dosage Forms and Strengths
- Inhalation powder: UTIBRON capsules contain 27.5 mcg of indacaterol and 15.6 mcg glycopyrrolate inhalation powder for use with the NEOHALER device. (3)
Contraindications
Warnings and Precautions
- LABA as monotherapy (without an inhaled corticosteroid) for asthma increases the risk of serious asthma-related events (5.1)
- Do not initiate in acutely deteriorating COPD or to treat acute symptoms. (5.2)
- Do not use in combination with an additional medicine containing LABA because of risk of overdose. (5.3, 7.1)
- If paradoxical bronchospasm occurs, discontinue UTIBRON NEOHALER immediately and institute alternative therapy. (5.4)
- Use with caution in patients with cardiovascular or convulsive disorders, thyrotoxicosis, sensitivity to sympathomimetic drugs, diabetes mellitus, and ketoacidosis. (5.6, 5.7, 7.1)
- Worsening of narrow-angle glaucoma or urinary retention may occur. Use with caution in patients with narrow-angle glaucoma, prostatic hyperplasia, or bladder-neck obstruction and instruct patients to contact a physician immediately if symptoms occur. (5.8, 5.9)
- Be alert to hypokalemia and hyperglycemia. (5.10)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence greater than or equal to 2% and higher than placebo) are nasopharyngitis and hypertension. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sunovion Pharmaceuticals Inc. at 1-877-737-7226 or FDA at 1-800-FDA-1088 orwww.fda.gov/medwatch.
Drug Interactions
- Other adrenergic drugs may potentiate effect: Use with caution. (5.3, 7.1)
- Xanthine derivatives, steroids, diuretics or non-potassium-sparing diuretics may potentiate hypokalemia or ECG changes. Use with caution. (7.2, 7.3)
- Monoamine Oxidase inhibitors, tricyclic antidepressants, and drugs that prolong QTc interval may potentiate effect on cardiovascular system. Use with extreme caution. (7.4)
- Beta-blockers may decrease effectiveness: Use with caution and only when medically necessary. (7.5)
- Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of UTIBRON NEOHALER with other anticholinergic-containing drugs. (7.6)
Use In Specific Populations
- Use in patients with severe renal impairment should be considered if the potential benefit of the treatment outweighs the risk. (8.6)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 5/2019
Full Prescribing Information
1. Indications and Usage for Utibron Neohaler
UTIBRON® NEOHALER® is a combination of indacaterol and glycopyrrolate indicated for the long-term, maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
Important Limitations of Use: UTIBRON NEOHALER is NOT indicated for the relief of acute bronchospasm or for the treatment of asthma [see Warnings and Precautions (5.1, 5.2)]. The safety and effectiveness of UTIBRON in asthma have not been established.
2. Utibron Neohaler Dosage and Administration
For oral inhalation only. Do not swallow UTIBRON capsules, as the intended effects on the lungs will not be obtained. UTIBRON capsules should only be used with the NEOHALER device [see Overdosage (10)].
The recommended dosage of UTIBRON NEOHALER is the inhalation of the contents of one UTIBRON capsule twice-daily using the NEOHALER device.
UTIBRON NEOHALER should be administered at the same time of the day (1 capsule in the morning and 1 capsule in the evening) every day. More frequent administration or a greater number of inhalations (more than 1 capsule twice-daily) of UTIBRON NEOHALER is not recommended.
Store UTIBRON capsules in the blister, and only remove IMMEDIATELY BEFORE USE with the NEOHALER device.
No dosage adjustment is required for geriatric patients, patients with mild and moderate hepatic impairment, or patients with mild to moderate renal impairment.
3. Dosage Forms and Strengths
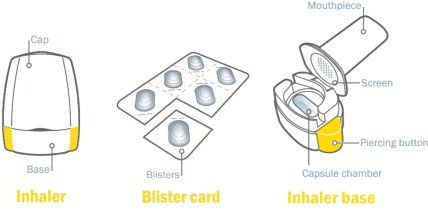
Inhalation powder: UTIBRON NEOHALER consists of UTIBRON capsules containing indacaterol/glycopyrrolate powder for oral inhalation and the NEOHALER device. UTIBRON capsules contain 27.5 mcg/15.6 mcg of indacaterol/glycopyrrolate in a hypromellose capsule with yellow transparent cap and uncolored transparent body with black “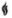 ” logo on the cap and black product code “IGP27.5_15.6” under the black bar on the body.
” logo on the cap and black product code “IGP27.5_15.6” under the black bar on the body.
4. Contraindications
Use of a LABA, including UTIBRON NEOHALER, without an inhaled corticosteroid is contraindicated in patients with asthma [see Warnings and Precautions (5.1)]. UTIBRON NEOHALER is not indicated for the treatment of asthma.
UTIBRON NEOHALER is contraindicated in patients who have demonstrated hypersensitivity to indacaterol, glycopyrrolate, or to any of the ingredients [see Warnings and Precautions (5.5)].
5. Warnings and Precautions
5.1 Serious Asthma-Related Events – Hospitalizations, Intubations, Death
- The safety and efficacy of UTIBRON NEOHALER in patients with asthma have not been established. UTIBRON NEOHALER is not indicated for the treatment of asthma [see Contraindications(4)].
- Use of long-acting beta2-adrenergic agonist (LABA) as monotherapy [without inhaled corticosteroids (ICS)] for asthma is associated with an increased risk of asthma-related death. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. These findings are considered a class effect of LABA monotherapy. When LABA are used in a fixed-dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared with ICS alone.
- A 28-week, placebo-controlled U.S. study comparing the safety of another LABA (salmeterol) with placebo, each added to usual asthma therapy, showed an increase in asthma-related deaths in patients receiving salmeterol (13/13,176 in patients treated with salmeterol versus 3/13,179 in patients treated with placebo; RR 4.37, 95% CI 1.25, 15.34). The increased risk of asthma-related death is considered a class effect of the LABAs, including indacaterol, one of the ingredients in UTIBRON NEOHALER.
- No study adequate to determine whether the rate of asthma-related death is increased in patients treated with UTIBRON NEOHALER has been conducted.
- Available data do not suggest an increased risk of death with use of LABA in patients with COPD.
5.2 Deterioration of Disease and Acute Episodes
UTIBRON NEOHALER should not be initiated in patients with acutely deteriorating or potentially life-threatening episodes of COPD. UTIBRON NEOHALER has not been studied in patients with acutely deteriorating COPD. The initiation of UTIBRON NEOHALER in this setting is not appropriate.
UTIBRON NEOHALER should not be used for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. UTIBRON NEOHALER has not been studied in the relief of acute symptoms, and extra doses should not be used for that purpose. Acute symptoms should be treated with an inhaled, short-acting beta2-agonist.
When beginning UTIBRON NEOHALER, patients who have been taking oral or inhaled, short-acting beta2-agonists on a regular basis (e.g., 4 times a day) should be instructed to discontinue the regular use of these drugs and use them only for symptomatic relief of acute respiratory symptoms. When prescribing UTIBRON NEOHALER, the healthcare provider should also prescribe an inhaled, short-acting beta2-agonist and instruct the patient on how it should be used. Increasing inhaled beta2-agonist use is a signal of deteriorating disease for which prompt medical attention is indicated.
COPD may deteriorate acutely over a period of hours or chronically over several days or longer. If UTIBRON NEOHALER no longer controls the symptoms of bronchoconstriction; the patient's inhaled, short-acting beta2-agonist becomes less effective; or the patient needs more inhalation of short-acting beta2-agonist than usual, these may be markers of deterioration of disease. In this setting, a re-evaluation of the patient and the COPD treatment regimen should be undertaken at once. Increasing the daily dose of UTIBRON NEOHALER beyond the recommended dose is not appropriate in this situation.
5.3 Excessive Use of UTIBRON NEOHALER and Use with Other Long-Acting Beta2-Adrenergic Agonists
As with other inhaled drugs containing beta2-adrenergics, UTIBRON NEOHALER should not be used more often than recommended, at higher doses than recommended, or in conjunction with other medications containing LABAs, as an overdose may result. Clinically significant cardiovascular effects and fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs. Patients using UTIBRON NEOHALER should not use another medicine containing a LABA for any reason [see Drug Interactions (7.1)].
5.4 Paradoxical Bronchospasm
As with other inhaled medicines, UTIBRON NEOHALER can produce paradoxical bronchospasm that may be life-threatening. If paradoxical bronchospasm occurs following dosing with UTIBRON NEOHALER, it should be treated immediately with an inhaled, short-acting bronchodilator; UTIBRON NEOHALER should be discontinued immediately and alternative therapy instituted.
5.5 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions have been reported after administration of indacaterol or glycopyrrolate, the components of UTIBRON NEOHALER. If signs suggesting allergic reactions occur, in particular, angioedema (including difficulties in breathing or swallowing, swelling of tongue, lips and face), urticaria, or skin rash, UTIBRON NEOHALER should be discontinued immediately and alternative therapy instituted. UTIBRON NEOHALER should be used with caution in patients with severe hypersensitivity to milk proteins.
5.6 Cardiovascular Effects
Indacaterol, like other beta2-agonists, can produce a clinically significant cardiovascular effect in some patients as measured by increases in pulse rate, systolic or diastolic blood pressure, or symptoms. If such effects occur, UTIBRON NEOHALER may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T-wave, prolongation of the QTc interval, and ST segment depression, although the clinical significance of these findings is unknown.
Therefore, UTIBRON NEOHALER should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
5.7 Coexisting Conditions
UTIBRON NEOHALER, like all medicines containing sympathomimetic amines, should be used with caution in patients with convulsive disorders or thyrotoxicosis, and in patients who are unusually responsive to sympathomimetic amines. Doses of the related beta2-agonist albuterol, when administered intravenously, have been reported to aggravate preexisting diabetes mellitus and ketoacidosis.
5.8 Worsening of Narrow-Angle Glaucoma
UTIBRON NEOHALER should be used with caution in patients with narrow-angle glaucoma. Prescribers and patients should be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.9 Worsening of Urinary Retention
UTIBRON NEOHALER should be used with caution in patients with urinary retention. Prescribers and patients should be alert for signs and symptoms of urinary retention (e.g., difficulty passing urine, painful urination), especially in patients with prostatic hyperplasia or bladder-neck obstruction. Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.10 Hypokalemia and Hyperglycemia
Beta2-adrenergic agonists may produce significant hypokalemia in some patients, which has the potential to produce adverse cardiovascular effects [see Clinical Pharmacology (12.2)]. The decrease in serum potassium is usually transient, not requiring supplementation. Inhalation of high doses of beta2-adrenergic agonists may produce increases in plasma glucose.
In patients with severe COPD, hypokalemia may be potentiated by hypoxia and concomitant treatment [see Drug Interactions (7.2)], which may increase the susceptibility for cardiac arrhythmias.
In 2 clinical trials of 12-weeks duration evaluating UTIBRON NEOHALER in subjects with COPD, there was no evidence of a treatment effect on serum glucose or potassium.
6. Adverse Reactions/Side Effects
LABAs, such as indacaterol, one of the active ingredients in UTIBRON NEOHALER, as monotherapy (without inhaled corticosteroids) for asthma increase the risk of asthma-related events. UTIBRON NEOHALER is not indicated for the treatment of asthma [see Warnings and Precautions (5.1)].
The following adverse reactions are described in greater detail in other sections:
- Paradoxical bronchospasm [see Warnings and Precautions (5.4)].
- Immediate hypersensitivity reactions [see Warnings and Precautions (5.5)].
- Cardiovascular effects [see Warnings and Precautions (5.6)].
- Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.8)].
- Worsening of urinary retention [see Warnings and Precautions (5.9)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in clinical practice.
The UTIBRON NEOHALER safety database included 2654 subjects with COPD in two 12-week lung function trials and one 52-week long-term safety study. A total of 712 subjects received treatment with UTIBRON NEOHALER 27.5 mcg/15.6 mcg twice daily (BID). The safety data described below are based on the two 12-week trials and the one 52-week trial.
12-Week Trials
The incidence of adverse reactions associated with UTIBRON NEOHALER in Table 1 is based on two 12-week, placebo-controlled trials (Trials 1 and 2; N=1,001 and N=1,042 respectively). Of the 2040 subjects, 63% were male and 91% were Caucasian. They had a mean age of 63 years and an average smoking history of 47 pack-years, with 52% identified as current smokers. At screening, the mean post-bronchodilator percent predicted forced expiratory volume in 1 second (FEV1) was 55% (range: 29% to 79%), the mean post-bronchodilator FEV1/forced vital capacity (FVC) ratio was 50% (range: 19% to 71%), and the mean percent reversibility was 23% (range: 0% to 144%).
The most common adverse reaction (incidence greater than or equal to 2% and higher than placebo) was nasopharyngitis and hypertension.
The proportion of patients who discontinued treatment due to adverse reactions was 2.95% for the UTIBRON NEOHALER treated patients and 4.13% for placebo-treated patients.
Subjects received 1 dose twice-daily of the following: UTIBRON NEOHALER 27.5 mcg/15.6 mcg, indacaterol 27.5 mcg, glycopyrrolate 15.6 mcg, or placebo.
| Adverse Reaction | UTIBRON NEOHALER 27.5/15.6 mcg BID (N=508) n (%) | Indacaterol 27.5 mcg BID (N=511) n (%) | Glycopyrrolate 15.6 mcg BID (N=513) n (%) | Placebo (N=508) n (%) |
| Nasopharyngitis | 21 (4.1) | 13 (2.5) | 12 (2.3) | 9 (1.8) |
| Hypertension | 10 (2.0) | 5 (1.0) | 3 (0.6) | 7 (1.4) |
| Back pain | 9 (1.8) | 7 (1.4) | 2 (0.4) | 3 (0.6) |
| Oropharyngeal pain | 8 (1.6) | 4 (0.8) | 8 (1.6) | 6 (1.2) |
Other adverse reactions occurring more frequently with UTIBRON NEOHALER than with placebo, but with an incidence of less than 1% include dyspepsia, gastroenteritis, chest pain, fatigue, peripheral edema, rash/pruritus, insomnia, dizziness, bladder obstruction/urinary retention, atrial fibrillation, palpitations, tachycardia.
52-Week Trial
In a long-term safety trial, 614 subjects were treated for up to 52 weeks with indacaterol/glycopyrrolate 27.5 mcg/15.6 mcg twice-daily, indacaterol/glycopyrrolate 27.5/31.2 mcg twice-daily or indacaterol 75 mcg once-daily. The demographic and baseline characteristics of the long-term safety trial were similar to those of the placebo-controlled efficacy trials described above. The adverse reactions reported in the long-term safety trial were consistent with those observed in the placebo-controlled trials of 12 weeks. Additional adverse reactions that occurred with a frequency greater than or equal to 2% in the group receiving indacaterol/glycopyrrolate 27.5 mcg/15.6 mcg twice-daily that exceeded the frequency of indacaterol 75 mcg once-daily in this trial were upper and lower respiratory tract infection, pneumonia, diarrhea, headache, gastroesophageal reflux disease, hyperglycemia, rhinitis.
6.2 Postmarketing Experience
The following additional adverse reactions of angioedema and dysphonia have been identified during worldwide post-approval use of indacaterol/glycopyrrolate at higher than the recommended dose. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Related/similar drugs
7. Drug Interactions
7.1 Adrenergic Drugs
If additional adrenergic drugs are to be administered by any route, they should be used with caution because the sympathetic effects of indacaterol, a component of UTIBRON NEOHALER, may be potentiated [see Warnings and Precautions (5.3, 5.6, 5.7, 5.10)].
7.2 Xanthine Derivatives, Steroids, or Diuretics
Concomitant treatment with xanthine derivatives, steroids, or diuretics may potentiate any hypokalemic effect of beta2-adrenergic agonists such as indacaterol, a component of UTIBRON NEOHALER [see Warnings and Precautions (5.10)].
7.3 Non-Potassium-Sparing Diuretics
The electrocardiographic (ECG) changes and/or hypokalemia that may result from the administration of non-potassium-sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, such as indacaterol, a component of UTIBRON NEOHALER, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical relevance of these effects is not known, caution is advised in the coadministration of UTIBRON NEOHALER with non-potassium-sparing diuretics.
7.4 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, QTc-Prolonging Drugs
Indacaterol, one of the components of UTIBRON NEOHALER, as with other beta2-agonists, should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors, tricyclic antidepressants, or other drugs known to prolong the QTc interval because the action of adrenergic agonists on the cardiovascular system may be potentiated by these agents. Drugs that are known to prolong the QTc interval may have an increased risk of ventricular arrhythmias.
7.5 Beta-Blockers
Beta-adrenergic receptor antagonists (beta-blockers) and UTIBRON NEOHALER may interfere with the effect of each other when administered concurrently. Beta-blockers not only block the therapeutic effects of beta-agonists, but may produce severe bronchospasm in COPD patients. Therefore, patients with COPD should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-blockers in patients with COPD. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
7.6 Anticholinergics
There is potential for an additive interaction with concomitantly used anticholinergic medicines. Therefore, avoid coadministration of UTIBRON NEOHALER with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic adverse effects [see Warnings and Precautions (5.8, 5.9), Adverse Reactions (6)].
7.7 Inhibitors of Cytochrome P450 3A4 and P-gp Efflux Transporter
Drug interaction studies with indacaterol, a component of UTIBRON NEOHALER, were carried out using potent and specific inhibitors of CYP3A4 and P-gp (i.e., ketoconazole, erythromycin, verapamil, and ritonavir). The data suggest that systemic clearance of indacaterol is influenced by modulation of both P-gp and CYP3A4 activities and that the 2-fold area under the curve (AUC) increase caused by the strong dual inhibitor ketoconazole reflects the impact of maximal combined inhibition. Indacaterol was evaluated in clinical trials for up to 1 year at doses up to 600 mcg. Inhibition of the key contributors of indacaterol clearance, CYP3A4 and P-gp, has no impact on safety of therapeutic doses of indacaterol. Therefore, no dose adjustment is warranted at the recommended 27.5/15.6 mcg twice-daily dose for UTIBRON NEOHALER when administered concomitantly with inhibitors of CYP3A4 and P-gp [see Clinical Pharmacology (12.3)].
8. Use In Specific Populations
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C
There are no adequate and well-controlled studies with UTIBRON NEOHALER or its individual components, indacaterol and glycopyrrolate, in pregnant women. Animal reproduction studies were conducted with individual components, indacaterol and glycopyrrolate. Because animal reproduction studies are not always predictive of human response, UTIBRON NEOHALER should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Women should be advised to contact their physician if they become pregnant while taking UTIBRON NEOHALER.
Indacaterol: Indacaterol was not teratogenic in Wistar rats and New Zealand rabbits at approximately 340 and 770 times, respectively, the MRHD in adults (on an AUC basis at maternal subcutaneous doses up to 1 mg/kg/day in rats and rabbits).
Glycopyrrolate: Glycopyrrolate was not teratogenic in Wistar rats or New Zealand White rabbits at approximately 1400 and 530 times, respectively, the MRHD in adults (on an AUC basis at maternal inhaled doses up to 3.83 mg/kg/day in rats and up to 4.4 mg/kg/day in rabbits).
Non-teratogenic Effects:
Indacaterol: There were no effects on perinatal and postnatal developments in rats at approximately 110 times the MRHD in adults (on an AUC basis at maternal subcutaneous doses up to 0.3 mg/kg/day).
Glycopyrrolate: There were no effects on perinatal and postnatal developments in rats at approximately 1100 times the MRHD in adults (on an AUC basis at maternal subcutaneous doses up to 1.88 mg/kg/day).
8.2 Labor and Delivery
There are no adequate and well-controlled human trials that have investigated the effects of UTIBRON NEOHALER during labor and delivery. Because beta-agonists may potentially interfere with uterine contractility, UTIBRON NEOHALER should be used during labor only if the potential benefit justifies the potential risk.
In human parturients undergoing Caesarean section, 86 minutes after a single intramuscular injection of 0.006 mg/kg glycopyrrolate, umbilical plasma concentrations were low.
8.3 Nursing Mothers
UTIBRON NEOHALER: It is not known whether UTIBRON NEOHALER is excreted in human breast milk. Because many drugs are excreted in human milk, caution should be exercised when UTIBRON NEOHALER is administered to a nursing woman. Since there are no data from well-controlled human studies on the use of UTIBRON NEOHALER by nursing mothers, based on the data for the individual components, a decision should be made whether to discontinue nursing or to discontinue UTIBRON NEOHALER, taking into account the importance of UTIBRON NEOHALER to the mother.
Indacaterol: It is not known whether indacaterol is excreted in human breast milk. Indacaterol (including its metabolites) have been detected in the milk of lactating rats.
Glycopyrrolate: It is not known whether glycopyrrolate is excreted in human breast milk. Glycopyrrolate (including its metabolites) have been detected in the milk of lactating rats and reached up to 10-fold higher concentrations in the milk than in the blood of the dam.
8.4 Pediatric Use
UTIBRON NEOHALER is not indicated for use in children. The safety and efficacy of UTIBRON NEOHALER in pediatric patients have not been established.
8.5 Geriatric Use
Based on available data, no adjustment of UTIBRON NEOHALER dosage in geriatric patients is warranted. UTIBRON NEOHALER can be used at the recommended dose in elderly patients 75 years of age and older.
Of the total number of subjects in clinical studies of UTIBRON NEOHALER, 45% were aged 65 and older, while 11% were aged 75 and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
Based on the pharmacokinetic characteristics of its monotherapy components, UTIBRON NEOHALER can be used at the recommended dose in patients with mild to moderate renal impairment. In patients with severe renal impairment (estimated GFR less than 30 mL/min/1.73 m2) or end-stage renal disease requiring dialysis, UTIBRON NEOHALER should be used if the expected benefit outweighs the potential risk since the systemic exposure to glycopyrrolate may be increased in this population [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Based on the pharmacokinetic characteristics of its monotherapy components, UTIBRON NEOHALER can be used at the recommended dose in patients with mild and moderate hepatic impairment. Studies in subjects with severe hepatic impairment have not been performed [see Clinical Pharmacology (12.3)].
10. Overdosage
In COPD patients, doses of up to 600/124.8 mcg UTIBRON NEOHALER were inhaled over 2 weeks and there were no relevant effects on heart rate, QTc interval, blood glucose or serum potassium. There was an increase in ventricular ectopies after 14 days of dosing with 300/124.8 mcg and 600/124.8 mcg UTIBRON NEOHALER, but low prevalence and small patient numbers (N=49 and N=51 for 600/124.8 mcg and 300/124.8 mcg UTIBRON NEOHALER, respectively) precluded accurate analysis. In a total of four patients, non-sustained ventricular tachycardia was recorded, with the longest episode recorded being 9 beats (4 seconds).
In healthy volunteers, single dose administration of UTIBRON NEOHALER at up to a dose of 440/499.2 mcg indacaterol/glycopyrrolate was well-tolerated without clinically significant effects on the ECG, serum potassium or blood glucose.
UTIBRON NEOHALER contains both indacaterol and glycopyrrolate; therefore, the risks associated with overdosage for the individual components described below apply to UTIBRON NEOHALER. Treatment of overdosage consists of discontinuation of UTIBRON NEOHALER together with institution of appropriate symptomatic and/or supportive therapy. The judicious use of a cardioselective beta-receptor blocker may be considered, bearing in mind that such medicine can produce bronchospasm. Cardiac monitoring is recommended in cases of overdosage.
Indacaterol
The potential signs and symptoms associated with overdosage of indacaterol are those of excessive beta-adrenergic stimulation and occurrence or exaggeration of any of the signs and symptoms, e.g., angina, hypertension or hypotension, tachycardia, with rates up to 200 bpm, arrhythmias, nervousness, headache, tremor, dry mouth, palpitation, muscle cramps, nausea, vomiting, drowsiness, dizziness, fatigue, malaise, hypokalemia, hyperglycemia, metabolic acidosis and insomnia. As with all inhaled sympathomimetic medications, cardiac arrest and even death may be associated with an overdose of indacaterol.
In COPD patients, single doses of indacaterol 3000 mcg were associated with moderate increases in pulse rate, systolic blood pressure and QTc interval.
Glycopyrrolate
An overdose of glycopyrrolate may lead to anticholinergic signs and symptoms such as nausea, vomiting, dizziness, lightheadedness, blurred vision, increased intraocular pressure (causing pain, vision disturbances or reddening of the eye), obstipation or difficulties in voiding.
In COPD patients, repeated orally inhaled administration of glycopyrrolate at total doses of 124.8 mcg and 249.6 mcg once-daily for 28 days were well tolerated.
Peak plasma levels and total systemic exposure following intravenous administration of 150 mcg glycopyrrolate (equivalent to 120 mcg active moiety) in healthy volunteers were respectively about 270-fold and 13-fold higher than the peak and total systemic exposure at steady-state achieved with the recommended daily dose of 31.2 mcg of glycopyrrolate (i.e., 15.6 mcg glycopyrrolate twice-daily) and were well tolerated.
11. Utibron Neohaler Description
UTIBRON NEOHALER consists of a dry powder formulation for delivery of a combination of indacaterol and glycopyrrolate to patients by oral inhalation only with the NEOHALER device. The inhalation powder is packaged in hypromellose (HPMC) capsules with yellow transparent cap and uncolored transparent body.
Each capsule contains a dry powder blend of 27.5 mcg/15.6 mcg of indacaterol/glycopyrrolate with approximately 24.9 mg of lactose monohydrate (which contains trace levels of milk protein) and 0.03 mg of magnesium stearate.
One active component of UTIBRON NEOHALER is indacaterol maleate, a (R) enantiomer. Indacaterol maleate is a selective beta2-adrenergic agonist. Its chemical name is (R)-5-[2-(5,6-Diethylindan-2-ylamino)-1-hydroxyethyl]-8-hydroxy-1H-quinolin-2-one maleate; its structural formula is:
Indacaterol maleate has a molecular weight of 508.56, and its empirical formula is C24H28N2O3 • C4H4O4. Indacaterol maleate is a white to very slightly grayish or very slightly yellowish powder. Indacaterol maleate is slightly soluble in ethanol and very slightly soluble in water.
The other active component of UTIBRON NEOHALER is glycopyrrolate, which is chemically described as (3RS)-3-[(2SR)-(2-cyclopentyl-2-hydroxy-2-phenylacetyl) oxy]-1,1-dimethylpyrrolidinium bromide. This synthetic quaternary ammonium compound acts as a competitive antagonist at muscarinic acetylcholine receptors, also referred to as anticholinergic. Glycopyrrolate, C19H28BrNO3, is a white powder that is freely soluble in water and sparingly soluble in absolute ethanol. It has a molecular mass of 398.33. The structural formula is:
The NEOHALER device is a plastic inhalation device used to inhale the dry powder within the UTIBRON capsule. The amount of drug delivered to the lung will depend on patient factors, such as inspiratory flow rate and inspiratory time.
Under standardized in vitro testing at a fixed flow rate of 90 L/min for 1.3 seconds, the NEOHALER inhaler delivered 20.8 mcg of indacaterol and 12.8 mcg glycopyrrolate for the 27.5 mcg/15.6 mcg dose strength (equivalent to 27.5 mcg/12.5 mcg of indacaterol/glycopyrronium) from the mouthpiece. This in vitro testing revealed that the NEOHALER device had a specific resistance of 0.059 cm H2O1/2/L/min. Peak inspiratory flow rates (PIFR) achievable through the NEOHALER device were evaluated in 26 adult patients with COPD of varying severity. Mean PIFR was 95 L/min (range 52 to 133 L/min) for adult patients. Twenty-five of 26 patients (96%) in this study generated a PIFR through the device exceeding 60 L/min.
12. Utibron Neohaler - Clinical Pharmacology
12.1 Mechanism of Action
UTIBRON NEOHALER contains both indacaterol and glycopyrrolate. The mechanisms of action described below for the individual components apply to UTIBRON NEOHALER. These drugs represent 2 different classes of medications (a LABA and an anticholinergic) that have different and additive effects on clinical and physiological indices.
Indacaterol: Indacaterol is a long-acting beta2-adrenergic agonist (LABA). When inhaled, indacaterol acts locally in the lung as a bronchodilator. Although beta2-receptors are the predominant adrenergic receptors in bronchial smooth muscle and beta1-receptors are the predominant receptors in the heart, there are also beta2-adrenergic receptors in the human heart comprising 10% to 50% of the total adrenergic receptors. The precise function of these receptors is not known, but their presence raises the possibility that even highly selective beta2-adrenergic agonists may have cardiac effects.
The pharmacological effects of beta2-adrenoceptor agonist drugs, including indacaterol, are at least in part attributable to stimulation of intracellular adenyl cyclase, the enzyme that catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3', 5'-adenosine monophosphate (cyclic monophosphate). Increased cyclic AMP levels cause relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells. In vitro studies have shown that indacaterol has more than 24-fold greater agonist activity at beta2-receptors compared to beta1-receptors and 20-fold greater agonist activity compared to beta3-receptors. The clinical significance of these findings is unknown.
Glycopyrrolate: Glycopyrrolate is a long-acting muscarinic antagonist, which is often referred to as an anticholinergic. It has similar affinity to the subtypes of muscarinic receptors M1 to M5. In the airways, it exhibits pharmacological effects through inhibition of M3 receptor at the smooth muscle leading to bronchodilation. The competitive and reversible nature of antagonism was shown with human and animal origin receptors and isolated organ preparations. In preclinical in vitro as well as in vivo studies, prevention of methacholine-induced broncho-constrictive effects was dose-dependent and lasted longer than 24 hours. The clinical relevance of these findings is unknown. The bronchodilation following inhalation of glycopyrrolate is predominantly a site-specific effect.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The QTc interval was studied in TQT studies with UTIBRON NEOHALER and with each of the monotherapy components. The TQT studies with indacaterol and glycopyrrolate demonstrated that neither of the compounds had a relevant effect on the corrected QT interval at supratherapeutic and therapeutic doses (for glycopyrrolate only a supratherapeutic dose was tested).
In a randomized, partially-blinded, placebo- and positive-controlled, crossover TQT study in 84 healthy subjects a supratherapeutic dose of UTIBRON NEOHALER (indacaterol/glycopyrrolate 440/499.2 mcg) was administered. This is a 16/32 dose multiple compared to a single dose of the recommended 27.5/15.6 mcg twice-daily dosage of UTIBRON NEOHALER, which resulted in exposure multiples for mean Cmax of 9.3 for indacaterol and 35.2 for glycopyrrolate compared to steady-state pharmacokinetics of UTIBRON NEOHALER 27.5/15.6 mcg twice-daily. The mean maximal change from baseline in QTcI compared to placebo was 8.70 msec (2-sided 90% CI 7.3, 10.1) at 30 minutes after dosing. Although a marginal QT effect of UTIBRON NEOHALER was observed at the supratherapeutic dose, it is unlikely there will be a clinically relevant effect at the therapeutic exposure.
12.3 Pharmacokinetics
Absorption
Following inhalation of UTIBRON NEOHALER, the median time to reach peak plasma concentrations of indacaterol and glycopyrrolate was achieved rapidly at approximately 15 minutes and 5 minutes, respectively.
The steady-state systemic exposure (AUC0-12h,ss; Cmax,ss) to indacaterol and glycopyrrolate is similar after the twice-daily inhalation of 2 (times 2) capsules of UTIBRON NEOHALER 27.5 mcg/15.6 mcg as compared to the twice-daily inhalation of the monotherapy products indacaterol 27.5 mcg (times 2) alone or glycopyrrolate 15.6 mcg (times 2) alone, respectively.
Indacaterol: Absolute bioavailability of indacaterol after an inhalation dose was on average 43% to 45%. Systemic exposure results from a composite of pulmonary and intestine absorption. Indacaterol serum concentrations increased with repeated once-daily administration. Steady-state was achieved within 12 to 15 days. The mean accumulation ratio of indacaterol, i.e., AUC over the 24-hour dosing interval on Day 14 or Day 15 compared to Day 1, was in the range of 2.9 to 3.8 for once-daily inhaled doses between 75 mcg and 600 mcg.
Glycopyrrolate: Following repeated once-daily inhalation in patients with COPD, pharmacokinetic steady-state of glycopyrrolate was reached within 1 week of treatment. There was no indication that the glycopyrrolate pharmacokinetics changes over time. With once-daily doses of 124.8 mcg and 249.6 mcg, steady-state exposure to glycopyrrolate (AUC over the dosing interval) was about 1.4- to 1.7-fold higher than after the first dose.
Distribution
Indacaterol: After intravenous infusion, the volume of distribution (Vz) of indacaterol was 2,361 to 2,557 L, indicating an extensive distribution. The in vitro human serum and plasma protein binding was 94.1% to 95.3% and 95.1% to 96.2%, respectively.
Glycopyrrolate: After intravenous administration, the steady-state volume of distribution (Vss) of glycopyrrolate was 83 L and the volume of distribution in the terminal phase (Vz) was 376 L. The in vitro human plasma protein binding of glycopyrrolate was 38% to 41% at concentrations of 1 to 10 ng/mL.
Metabolism
Indacaterol: In vitro investigations indicated that UGT1A1 is the only UGT isoform that metabolized indacaterol to the phenolic O-glucuronide. The oxidative metabolites were found in incubations with recombinant CYP1A1, CYP2D6, and CYP3A4. CYP3A4 is concluded to be the predominant isoenzyme responsible for hydroxylation of indacaterol. In vitro investigations further indicated that indacaterol is a low affinity substrate for the efflux pump P-gp.
After oral administration of radiolabelled indacaterol in a human ADME (absorption, distribution, metabolism, excretion) study, unchanged indacaterol was the main component in serum, accounting for about one-third of total drug-related AUC over 24 hours. A hydroxylated derivative was the most prominent metabolite in serum. Phenolic O-glucuronides of indacaterol and hydroxylated indacaterol were further prominent metabolites. A diastereomer of the hydroxylated derivative, a N-glucuronide of indacaterol, and C- and N-dealkylated products were further metabolites identified.
Glycopyrrolate: In vitro metabolism studies show glycopyrrolate hydroxylation resulting in a variety of mono- and bis-hydroxylated metabolites and direct hydrolysis resulting in the formation of a carboxylic acid derivative (M9). Further in vitro investigations showed that multiple CYP isoenzymes contribute to the oxidative biotransformation of glycopyrrolate and the hydrolysis to M9 is likely to be catalyzed by members from the cholinesterase family pre-systemically and/or via first pass metabolism from the swallowed dose fraction of orally inhaled glycopyrrolate. Glucuronide and/or sulfate conjugates of glycopyrrolate were found in urine of humans after repeated inhalation, accounting for about 3% of the dose.
Elimination
Indacaterol: In clinical studies which included urine collection, the amount of indacaterol excreted unchanged via urine was generally lower than 2% of the dose. Renal clearance of indacaterol was, on average, between 0.46 L/h and 1.20 L/h. When compared with the serum clearance of indacaterol of 18.8 L/h to 23.3 L/h, it is evident that renal clearance plays a minor role (about 2% to 6% of systemic clearance) in the elimination of systemically available indacaterol.
In a human ADME study where indacaterol was given orally, the fecal route of excretion was dominant over the urinary route. Indacaterol was excreted into human feces primarily as unchanged parent drug (54% of the dose) and, to a lesser extent, hydroxylated indacaterol metabolites (23% of the dose). Mass balance was complete with greater than or equal to 90% of the dose recovered in the excreta.
Indacaterol serum concentrations declined in a multi-phasic manner with an average terminal half-life ranging from 45.5 to 126 hours. The effective half-life, calculated from the accumulation of indacaterol after repeated dosing, ranged from 40 to 56 hours, which is consistent with the observed time to steady-state of approximately 12 to 15 days.
Glycopyrrolate: Renal elimination of parent drug accounts for about 60% to 70% of total clearance of systemically available glycopyrrolate whereas non-renal clearance processes account for about 30% to 40%. Biliary clearance contributes to the non-renal clearance, but the majority of non-renal clearance is thought to be due to metabolism.
Following inhalation of single and repeated once-daily doses between 62.4 mcg and 249.6 mcg glycopyrrolate by healthy volunteers and patients with COPD, mean renal clearance of glycopyrrolate was in the range of 17.4 L/h and 24.4 L/h. Active tubular secretion contributes to the renal elimination of glycopyrrolate.
Glycopyrrolate plasma concentrations declined in a multi-phasic manner. The mean terminal elimination half-life of glycopyrrolate was much longer after inhalation (33 to 53 hours) than after intravenous (6.2 hours) and oral (2.8 hours) administration.
Drug Interactions
There is no pharmacokinetic drug-drug interaction resulting from the concomitant administration of inhaled glycopyrrolate and inhaled indacaterol based on steady-state exposure data.
No specific drug-drug interaction studies were conducted with UTIBRON NEOHALER. Information on the potential for interactions for UTIBRON NEOHALER is based on the potential for each of its 2 monotherapy components.
Inhibitors of Cytochrome P450 3A4 and P-gp Efflux Transporter: Drug interaction studies were carried out using potent and specific inhibitors of CYP3A4 and P-gp (i.e., ketoconazole, erythromycin, verapamil and ritonavir). Coadministration of indacaterol 300 mcg (single dose) with verapamil (80 mg 3 times a day for 4 days) showed 2-fold increase in indacaterol AUC0-24h, and 1.5-fold increase in indacaterol Cmax. Coadministration of indacaterol inhalation powder 300 mcg (single dose) with erythromycin (400 mg 4 times a day for 7 days) showed a 1.4-fold increase in indacaterol AUC0-24h, and 1.2-fold increase in indacaterol Cmax. Coadministration of indacaterol inhalation powder 300 mcg (single dose) with ketoconazole (200 mg twice-daily for 7 days) caused a 1.9-fold increase in indacaterol AUC0-24h, and 1.3-fold increase in indacaterol Cmax. Coadministration of indacaterol 300 mcg (single dose) with ritonavir (300 mg twice-daily for 7.5 days) resulted in a 1.7-fold increase in indacaterol AUC0-24h whereas indacaterol Cmax was unaffected [see Drug Interactions (7.7)].
Cimetidine or Other Inhibitors of Organic Cationic Transport: In a clinical study in healthy volunteers, cimetidine, an inhibitor of organic cation transport that is thought to contribute to the renal excretion of glycopyrrolate, increased total exposure (AUC) to glycopyrrolate by 22% and decreased renal clearance by 23%.
Effect of UTIBRON on co-administered drugs exposure
In vitro studies show that UTIBRON NEOHALER is not likely to inhibit or induce the metabolism of other drugs, nor processes involving drug transporters.
Indacaterol: In vitro investigations indicated that indacaterol has negligible potential to cause metabolic interactions with medications (by inhibition or induction of cytochrome P450 enzymes, or induction of UGT1A1) at the systemic exposure levels achieved in clinical practice. In vitro investigation furthermore indicated that, in vivo, indacaterol is unlikely to significantly inhibit transporter proteins such as P-gp, MRP2, BCRP, the cationic substrate transporters hOCT1 and hOCT2, and the human multidrug and toxin extrusion transporters hMATE1 and hMATE2K, and that indacaterol has negligible potential to induce P-gp or MRP2.
Glycopyrrolate: In vitro inhibition studies demonstrated that glycopyrrolate has no relevant capacity to inhibit CYP1A2, CYP2A6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1 or CYP3A4/5, the efflux transporters MDR1, MRP2 or MXR, and the uptake transporters OATP1B1, OATP1B3, OAT1, OAT3, OCT1 or OCT2. In vitro enzyme induction studies did not indicate a clinically relevant induction by glycopyrrolate for any of the cytochrome P450 isoenzymes tested as well as for UGT1A1 and the transporters MDR1 and MRP2.
Specific Populations
Population pharmacokinetic analysis showed no evidence of a clinically significant effect of age (40 to 85 years), body weight (45 to 120 kg), gender, smoking status, and baseline FEV1 on systemic exposure of either indacaterol or glycopyrrolate following inhalation of UTIBRON NEOHALER.
Similarly no relevant covariate effect (of age, body weight, gender, smoking status, and baseline FEV1) was observed following the inhalation of the 2 components indacaterol and glycopyrrolate separately.
Patients with Renal Impairment
Indacaterol: Due to the very low contribution of the urinary pathway to total body elimination of indacaterol, a study in renally impaired subjects was not performed.
Glycopyrrolate: Renal impairment has an impact on the systemic exposure to glycopyrrolate. A moderate mean increase in total systemic exposure (AUC last) of up to 1.4-fold was seen in subjects with mild and moderate renal impairment [estimated GFR greater than or equal to 30 mL/min/1.73m2] and up to 2.2-fold in subjects with severe renal impairment and end stage renal disease [estimated GFR less than 30 mL/min/1.73m2] [see Use in Specific Populations (8.6)].
Patients with Hepatic Impairment
Based on the clinical pharmacokinetic characteristics of its monotherapy components, UTIBRON NEOHALER can be used at the recommended dose in patients with mild and moderate hepatic impairment. UTIBRON NEOHALER has not been evaluated in subjects with severe hepatic impairment.
Indacaterol: Patients with mild and moderate hepatic impairment showed no relevant changes in Cmax or AUC of indacaterol, nor did protein binding differ between mild and moderate hepatic impaired subjects and their healthy controls. Studies in subjects with severe hepatic impairment were not performed [see Use in Specific Populations (8.7)].
Glycopyrrolate: Clinical studies in patients with hepatic impairment have not been conducted. Glycopyrrolate is cleared predominantly from the systemic circulation by renal excretion.
Ethnicity
There was no evidence of a clinically significant ethnic/race effect (across Caucasian, Chinese, and Japanese subjects) on the systemic exposure to indacaterol and glycopyrrolate following inhalation of UTIBRON NEOHALER.
Similarly, no relevant ethnic effect was observed following the inhalation of the 2 components indacaterol and glycopyrrolate separately.
12.5 Pharmacogenomics
Indacaterol: The pharmacokinetics of indacaterol were prospectively investigated in subjects with the UGT1A1 (TA)7/(TA)7 genotype (low UGT1A1 expression; also referred to as *28) and the (TA)6, (TA)6 genotype. Steady-state AUC and Cmax of indacaterol were 1.2-fold higher in the [(TA)7, (TA)7] genotype, suggesting no relevant effect of UGT1A1 genotype of indacaterol exposure.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies of carcinogenicity, mutagenicity, or impairment of fertility were conducted with UTIBRON NEOHALER; however, studies are available for the individual components, indacaterol and glycopyrrolate, as described below.
Indacaterol: Indacaterol produced no treatment-related increases in the incidence of tumors in a 2-year inhalation study in Wistar rats at inhaled doses up to 2.09 mg/kg/day (approximately 110 times the MRHD in adults on an AUC basis, respectively). A 26-week oral (gavage) study in CB6F1/TgrasH2 hemizygous mice with indacaterol at doses up to 600 mg/kg/day did not show any evidence of tumorigenicity.
Indacaterol tested negative in the following genotoxicity assays: the in vitro Ames test, in vitro chromosome aberration test in V79 Chinese hamster cells, and in vivo rat bone marrow micronucleus test.
Indacaterol had no effects on fertility and reproductive performance in male and female rats at subcutaneous doses up to 2 mg/kg/day (approximately 890 and 670 times in males and females, respectively, the MRHD in adults on an AUC basis).
Glycopyrrolate: Glycopyrrolate produced no treatment-related increases in the incidence of tumors in a 2-year inhalation study in Wistar rats at inhaled doses up to 0.56 mg/kg/day (approximately 170 times the MRHD in adults on an AUC basis). A 26-week oral (gavage) study in male and female TgrasH2 mice that received glycopyrrolate at doses up to 93.8 and 125.1 mg/kg/day, respectively, did not show any evidence of tumorigenicity.
Glycopyrrolate tested negative in the following genotoxicity assays: the in vitro Ames assay, in vitro human lymphocyte chromosomal aberration assay, and in vivo rat bone marrow micronucleus assay.
Impairment of fertility was observed in male and female rats at a subcutaneous dose of 1.88 mg/kg/day (approximately 1900 and 1100 times the MRHD in adults on an AUC basis, respectively) based upon findings of decreased corpora lutea, implantation sites and live fetuses. No effects on fertility and reproductive performance in male and female rats were observed at a subcutaneous dose of 0.63 mg/kg/day (approximately 350 times the MRHD in adults on an AUC basis).
13.2 Animal Toxicology
Eye findings were seen for glycopyrrolate and were observed in Wistar rats at inhaled doses of 0.67 mg/kg/day and higher (approximately 280 times the MRHD in adults on an AUC basis) based upon findings of anterior capsule opacity, prominent anterior suture line, and anterior cataract. No eye findings in Wistar rats were observed at an inhaled dose of 0.09 mg/kg/day (approximately 60 times the MRHD in adults on an AUC basis). Eye findings were observed in beagle dogs at an inhaled dose of 0.33 mg/kg/day (approximately 150 times the MRHD in adults on an AUC basis) based upon findings of conjunctival hyperemia and corneal opacity. No eye findings in beagle dogs were observed at an inhaled dose of 0.12 mg/kg/day (approximately 100 times the MRHD in adults on an AUC basis).
14. Clinical Studies
The safety and efficacy of UTIBRON NEOHALER were evaluated in a clinical development program that included 3 dose-ranging trials, 2 lung function trials of 12 weeks duration (placebo-controlled and active-controlled) and a 12-month long-term safety trial. The efficacy of UTIBRON NEOHALER is based primarily on the dose-ranging trials in 562 subjects with COPD or asthma and the 2 placebo- and active-controlled confirmatory trials in 2043 subjects with additional support from one active-controlled 12-month trial in 615 subjects with COPD.
14.1 Dose-Ranging Trials
Dose selection for UTIBRON NEOHALER for COPD was based on data for the individual components, indacaterol and glycopyrrolate.
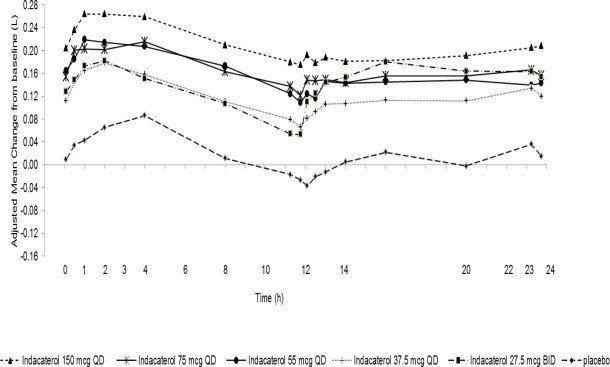
Indacaterol: Indacaterol dose selection in UTIBRON NEOHALER is based on the registered dose of 75 mcg once-daily and is also supported by a single-dose, multicenter, randomized, double-blind, placebo-controlled, crossover study in asthma evaluating 5 doses of indacaterol in 91 subjects, (37.5 mcg, 55 mcg, 75 mcg, and 150 mcg once-daily (QD), and 27.5 mcg twice-daily (BID)). A dose-related increase in FEV1 AUC0-24h compared with placebo was observed. The differences in change from baseline in FEV1 AUC0-24h Rafter single dosing for the indacaterol 37.5 mcg, 55 mcg, 75 mcg, and 150 mcg once-daily and the 27.5 mcg twice-daily doses compared to placebo were 0.099 L (95% CI: 0.069, 0.128), 0.132 L (0.103, 0.162), 0.143 L (0.114, 0.171), 0.187 L (0.157, 0.216), and 0.121 L (0.092, 0.151), respectively. These results supported the evaluation of indacaterol 27.5 mcg twice-daily in the confirmatory COPD trials. UTIBRON NEOHALER is not indicated for asthma.
Figure 1. Adjusted mean change from baseline in FEV1(L) over 24 hours on Day 1
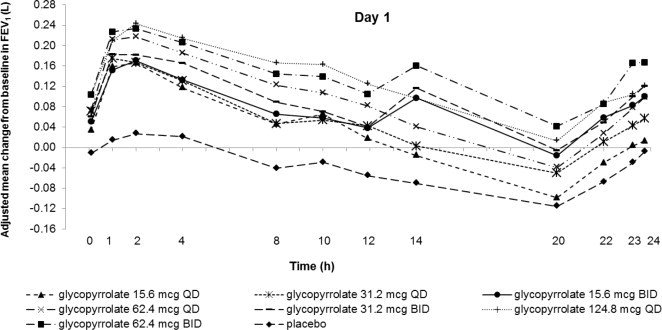
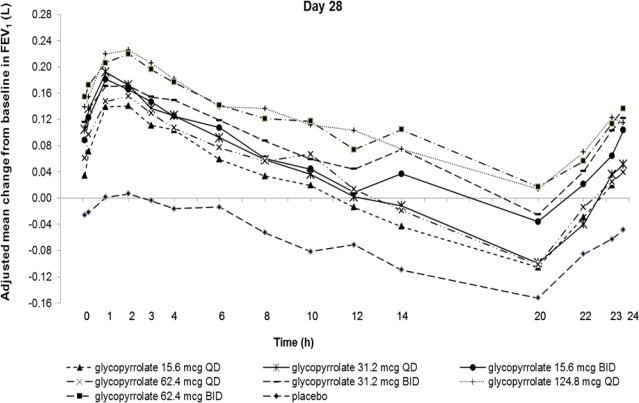
Glycopyrrolate: Dose selection for glycopyrrolate in UTIBRON NEOHALER in COPD was supported by a 28-day, randomized, double-blind, placebo-controlled, 2-period, crossover study evaluating 7 doses of glycopyrrolate (15.6 mcg, 31.2 mcg, 62.4 mcg, and 124.8 mcg once-daily and 15.6 mcg, 31.2 mcg, and 62.4 mcg twice-daily) or placebo in 388 subjects with COPD. The differences in trough FEV1 from baseline after 28 days compared to placebo for the 15.6 mcg, 31.2 mcg, 62.4 mcg, and 124.8 mcg once-daily and for 15.6 mcg, 31.2 mcg, and 62.4 mcg twice-daily doses were 0.083 L (95% CI: 0.030, 0.136), 0.098 L (0.048, 0.148), 0.090 L (0.038, 0.142), 0.176 L (0.132, 0.220), 0.139 L (0.089, 0.189), 0.167 L (0.115, 0.219), and 0.177 L (0.132, 0.222), respectively. The dose-ranging results supported the evaluation of glycopyrrolate 15.6 mcg twice-daily in the confirmatory COPD trials.
Figure 2. Adjusted mean change from baseline in FEV1(L) over 24 hours on Days 1 and 28
Based on the findings from dose-ranging studies of the individual components, a twice-daily dose of
indacaterol/glycopyrrolate 27.5 mcg/15.6 mcg was evaluated in the confirmatory COPD trials.
14.2 Confirmatory Trials
The clinical development program for UTIBRON NEOHALER included two (Trial 1 and Trial 2) 12-week, randomized, double-blinded, placebo- and active-controlled, parallel-group trials in subjects with COPD designed to evaluate the efficacy and safety of UTIBRON NEOHALER; and one 12-month, randomized, double-blind, active-controlled trial (Trial 3) that evaluated bronchodilation and effects on long-term safety.
The 12-week trials evaluated the efficacy of 2038 subjects that had a clinical diagnosis of COPD, were 40 years of age or older, had a history of smoking greater than 10 pack-years, had a post-albuterol FEV1 greater than or equal to 30% and less than 80% of predicted normal values, had a ratio of FEV1/FVC of less than 0.7, and were symptomatic as determined by a Modified Medical Research Council (mMRC) score greater than or equal to 2. Of the 2038 subjects included in the efficacy analysis, 63% were male and 91% were Caucasian. They had a mean age of 63 years and an average smoking history of 47 pack-years, with 52% identified as current smokers, and 46% used concomitant inhaled corticosteroids. At screening, the mean post-bronchodilator percent predicted FEV1 was 55% (range: 29% to 79%), the mean post-bronchodilator FEV1/FVC ratio was 50% (range: 19% to 71%), and the mean percent reversibility was 23% (range: 0% to 144%).
Trial 1 and Trial 2 evaluated UTIBRON NEOHALER (indacaterol/glycopyrrolate) 27.5 mcg/15.6 mcg twice-daily (BID), indacaterol 27.5 mcg BID, glycopyrrolate 15.6 mcg BID, and placebo BID. The primary endpoint was the change from baseline in FEV1 AUC0-12h Rfollowing the morning dose at Day 85 (defined as the mean FEV1 change from baseline over 0 to 12 hours divided by 12 hours) compared with placebo, glycopyrrolate 15.6 mcg BID, and indacaterol 27.5 mcg BID. The comparison of UTIBRON NEOHALER with indacaterol 27.5 mcg and glycopyrrolate 15.6 mcg was assessed to evaluate the contribution of the individual comparators to UTIBRON NEOHALER. In both trials, UTIBRON NEOHALER demonstrated a larger increase in mean change from baseline in FEV1 AUC0-12h Rcompared to placebo, indacaterol 27.5 mcg BID, and glycopyrrolate 15.6 mcg BID (see Table 2).
|
N = Number in intent-to-treat population |
||||
|
* The indacaterol and glycopyrrolate comparators used the same inhaler and excipients as UTIBRON NEOHALER |
||||
|
With the limited data available, there was no suggestion of a difference in FEV1 AUC0-12h Rwith respect to age, sex, degree of airflow limitation, GOLD stage, smoking status, or inhaled corticosteroid use. |
||||
| Treatment | N | FEV1(L) AUC0-12hat Week 12 | ||
| Difference from | ||||
| Placebo
LS Mean (95% CI) | Indacaterol
27.5 mcg BID* LS Mean (95% CI) | Glycopyrrolate
15.6 mcg BID* LS Mean (95% CI) |
||
| Trial 1 (N=996) | ||||
| UTIBRON NEOHALER 27.5 mcg/15.6 mcg BID | 249 | N=246 0.262 L (0.224, 0.300) | N=251 0.112 L (0.075, 0.149) | N=250 0.079 L (0.042, 0.116) |
| Trial 2 (N=1039) | ||||
| UTIBRON NEOHALER 27.5 mcg/15.6 mcg BID | 258 | N=260 0.231 L (0.192, 0.271) | N=260 0.094 L (0.055, 0.133) | N=261 0.098 L (0.059, 0.137) |
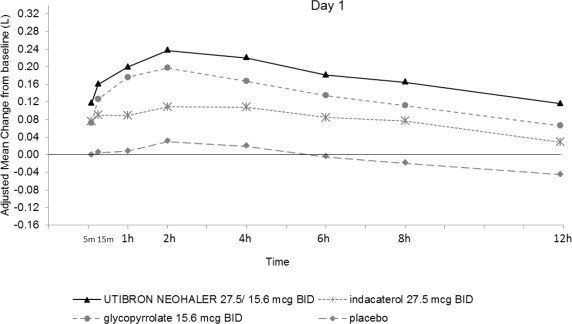
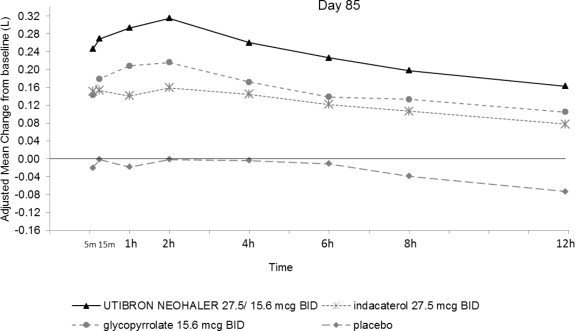
In Trial 1 and Trial 2, serial spirometric evaluations throughout the 12-hour dosing interval were performed in all subjects at Days 1 and 85. The spirometric curves from Trial 1 at Days 1 and 85 are displayed in Figure 3. In Trial 2, the results for UTIBRON NEOHALER in FEV1 AUC0-12h were similar to those observed in Trial 1.
Figure 3. Adjusted mean change from baseline in FEV1(L) over 12 hours on Days 1 and 85 in Trial 1 (All patient Population)
The peak FEV1 was defined as the maximum FEV1 recorded within 4 hours after the morning dose on Days 1 and 85. The mean peak FEV1 improvement from baseline for UTIBRON NEOHALER compared with placebo at Day 1 and at Day 85 was 0.185 L and 0.290 L (Trial 1) and 0.151 L and 0.260 L (Trial 2), respectively. The median time to onset on Day 1, defined as a 100 mL increase from baseline in FEV1, was 12 minutes and 16 minutes in Trials 1 and 2, respectively, in subjects receiving UTIBRON NEOHALER.
In Trials 1 and 2, patients treated with UTIBRON NEOHALER used less daily rescue albuterol compared to patients treated with placebo.
The St. George's Respiratory Questionnaire (SGRQ) was assessed in Trials 1 and 2. In Trial 2, the SGRQ responder rate
(defined as an improvement in score of 4 or more as threshold) was 57%, 46%, 48%, and 39%, for UTIBRON NEOHALER, glycopyrrolate, indacaterol, and placebo, respectively, with odds ratios of 1.6 (95% CI 1.1, 2.3), 1.5 (95% CI 1.1, 2.2), and 2.2 (95% CI 1.5, 3.2), for UTIBRON NEOHALER vs. glycopyrrolate, UTIBRON NEOHALER vs. indacaterol, and UTIBRON NEOHALER vs. placebo, respectively. In Trial 1, the trends were similar, with odds ratios of 1.4 (95% CI 1.0, 2.0), 1.1 (95% CI 0.8, 1.7), and 2.9 (95% CI 1.9, 4.2), for UTIBRON NEOHALER vs. glycopyrrolate, UTIBRON NEOHALER vs. indacaterol, and UTIBRON NEOHALER vs placebo, respectively.
16. How is Utibron Neohaler supplied
How Supplied
UTIBRON NEOHALER contains UTIBRON (indacaterol/glycopyrrolate, (27.5 mcg, 15.6 mcg) inhalation powder) in hypromellose (HPMC) capsule with yellow transparent cap and uncolored transparent body, with capsules packaged in aluminum blister cards, one NEOHALER device, and an FDA-approved Patient Information.
Unit Dose (blister pack), Box of 60 (10 blister cards with 6 yellow transparent capsules each) NDC 63402-681-60
Unit Dose (blister pack), Box of 6 (1 blister card with 6 yellow transparent capsules each) NDC 63402-681-06
The NEOHALER device consists of a white protective cap and a base with mouthpiece, capsule chamber and 2 yellow push buttons.
Storage and Handling
Store in a dry place at 77°F (25°C); excursions permitted to 59°F to 86°F (15°C to 30°C) [see USP Controlled Room Temperature].
- UTIBRON capsules should be used with the NEOHALER device only. Do not use the NEOHALER device with any other capsules.
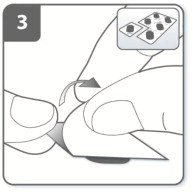
- Store UTIBRON capsules in the blister protected from light and moisture. Remove the UTIBRON capsules from the blister immediately before use.
- Always use the new NEOHALER inhaler provided with each new prescription.
Keep out of the reach of children.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Serious Asthma-Related Events
Inform patients that LABAs, such as indacaterol, one of the active ingredients in UTIBRON NEOHALER, when used as monotherapy [without an inhaled corticosteroid] increase the risk of serious asthma-related events, including asthma-related death. UTIBRON NEOHALER is not indicated for the treatment of asthma.
Not for Acute Symptoms
Inform patients that UTIBRON NEOHALER is not meant to relieve acute symptoms of COPD and extra doses should not be used for that purpose. Advise them to treat acute symptoms with a rescue inhaler such as albuterol. Provide patients with such medicine and instruct them on how it should be used [see Warnings and Precautions (5.2)].
Instruct patients to seek medical attention immediately if they experience any of the following:
- Symptoms get worse
- Need for more inhalations than usual of their rescue inhaler
Patients should not stop therapy with UTIBRON NEOHALER without physician/healthcare provider guidance since symptoms may recur after discontinuation.
Do Not Use Additional Long-Acting Beta2-Agonists
Instruct patients to not use other medicines containing a LABA. Patients should not use more than the recommended twice-daily dose of UTIBRON NEOHALER [see Warnings and Precautions (5.3)].
Instruct patients who have been taking inhaled, short-acting beta2-agonists on a regular basis to discontinue the regular use of these products and use them only for the symptomatic relief of acute symptoms.
Paradoxical Bronchospasm
Inform patients that UTIBRON NEOHALER can produce paradoxical bronchospasm. Advise patients that if paradoxical bronchospasm occurs, patients should discontinue UTIBRON NEOHALER.
Risks Associated with Beta2-Agonist Therapy
Inform patients of adverse effects associated with beta2-agonists, such as palpitations, chest pain, rapid heart rate, tremor, or nervousness. Instruct patients to consult a physician immediately should any of these signs or symptoms develop [see Warnings and Precautions (5.6)].
Worsening of Narrow-Angle Glaucoma
Instruct patients to be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop [see Warnings and Precautions (5.8)].
Worsening of Urinary Retention
Instruct patients to be alert for signs and symptoms of urinary retention (e.g., difficulty passing urine, painful urination). Instruct patients to consult a physician immediately should any of these signs or symptoms develop [see Warnings and Precautions (5.9)].
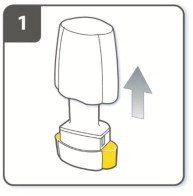
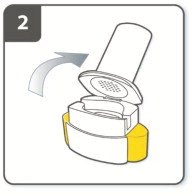
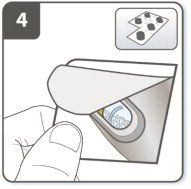
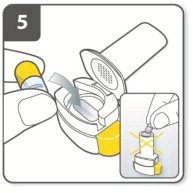
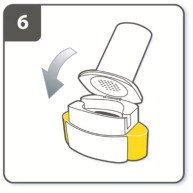
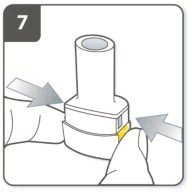
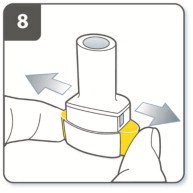
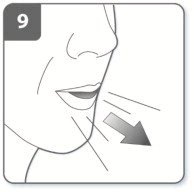
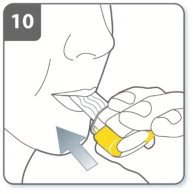
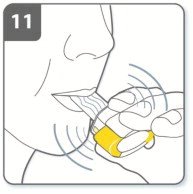
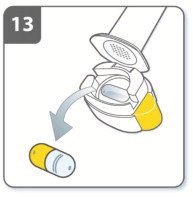
Instructions for Administering UTIBRON NEOHALER
It is important for patients to understand how to correctly administer UTIBRON capsules using the NEOHALER device [see Instructions for Use]. Instruct patients that UTIBRON capsules should only be administered via the NEOHALER device and the NEOHALER device should not be used for administering other medications. Remind patients that the contents of UTIBRON capsules are for oral inhalation only and must not be swallowed.
Instruct patients always to store UTIBRON capsules in sealed blisters and to only remove 1 UTIBRON capsule immediately before use or it may not be as effective. Instruct patients to discard unused additional UTIBRON capsules that are exposed to air (i.e., not intended for immediate use).
Inform patients to use 1 inhalation of UTIBRON NEOHALER orally twice-daily (1 capsule in the morning and 1 capsule in the evening) at the same time every day.
Inform patients that if they miss a dose of UTIBRON NEOHALER, they should use their next capsule at the usual time. Instruct patients to not use 2 capsules at one time and to not use more than 2 capsules in a day.
Manufactured for
Sunovion Pharmaceuticals Inc.
Marlborough, MA 01752 USA
Made in Switzerland
© 2019 Sunovion Pharmaceuticals Inc.
For customer service, call 1-888-394-7377
For medical information, call 1-800-739-0565
To report suspected adverse events, call 1-877-737-7226
Revised: 05/2019
10184-02
|
This Patient Information has been approved by the U.S. Food and Drug Administration. 05/2019 |
|
| PATIENT INFORMATION
UTIBRON®(yoo-TEE-bron) NEOHALER® (indacaterol and glycopyrrolate) inhalation powder, for oral inhalation use |
|
| Important: Do not swallow UTIBRON capsules. UTIBRON capsules are used only with the NEOHALER inhaler that comes with UTIBRON NEOHALER. Do not place a capsule in the mouthpiece of the NEOHALER inhaler. | |
| What is UTIBRON NEOHALER?
UTIBRON NEOHALER combines a long-acting beta2 adrenergic agonist (LABA) medicine (indacaterol) and an anticholinergic medicine (glycopyrrolate).
|
|
Do not use UTIBRON NEOHALER if you:
|
|
Before using UTIBRON NEOHALER, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| UTIBRON NEOHALER contains lactose (milk sugar) and a small amount of milk proteins. It is possible that allergic reactions may happen in people who have a severe milk protein allergy. Tell your healthcare provider about all the medicines you take, including prescription medicines, over-the-counter medicines, vitamins, and herbal supplements. UTIBRON NEOHALER may affect the way other medicines work, and other medicines can affect how UTIBRON NEOHALER works. Using UTIBRON NEOHALER with other medicines may cause serious side effects. Know the medicines you take. Keep a list of your medicines with you and show it to your healthcare provider and pharmacist when you get a new medicine. |
|
| How should I use UTIBRON NEOHALER?
Read the step-by-step instructions for using UTIBRON NEOHALER at the end of this Patient Information leaflet.
|
|
| What are the possible side effects of UTIBRON NEOHALER?
UTIBRON NEOHALER can cause serious side effects, including:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
If you have these symptoms, stop taking UTIBRON NEOHALER and call your healthcare provider right away before taking another dose.
|
|
|
|
| If you have these symptoms, stop taking UTIBRON NEOHALER and call your healthcare provider right away before using another dose. |
|
|
|
| Common side effects of UTIBRON NEOHALER include sore throat and runny nose, high blood pressure, and back pain. Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all of the possible side effects of UTIBRON NEOHALER. For more information, ask your healthcare provider or pharmacist. Call your doctor or pharmacist for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store UTIBRON NEOHALER?
|
|
| Keep UTIBRON NEOHALER and all medicines out of the reach of children. | |
| General information about the safe and effective use of UTIBRON NEOHALER.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use UTIBRON NEOHALER for a condition for which it was not prescribed. Do not give UTIBRON NEOHALER to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information that is written for healthcare professionals. For more information about UTIBRON NEOHALER or to report side effects, go to www.Utibron.com or call 1-888-394-7377. |
|
|
What are the ingredients in UTIBRON NEOHALER? Active ingredients: indacaterol maleate, glycopyrrolate Inactive ingredients: lactose monohydrate (contains milk proteins) and magnesium stearate. Revised: 05/2019 |
|
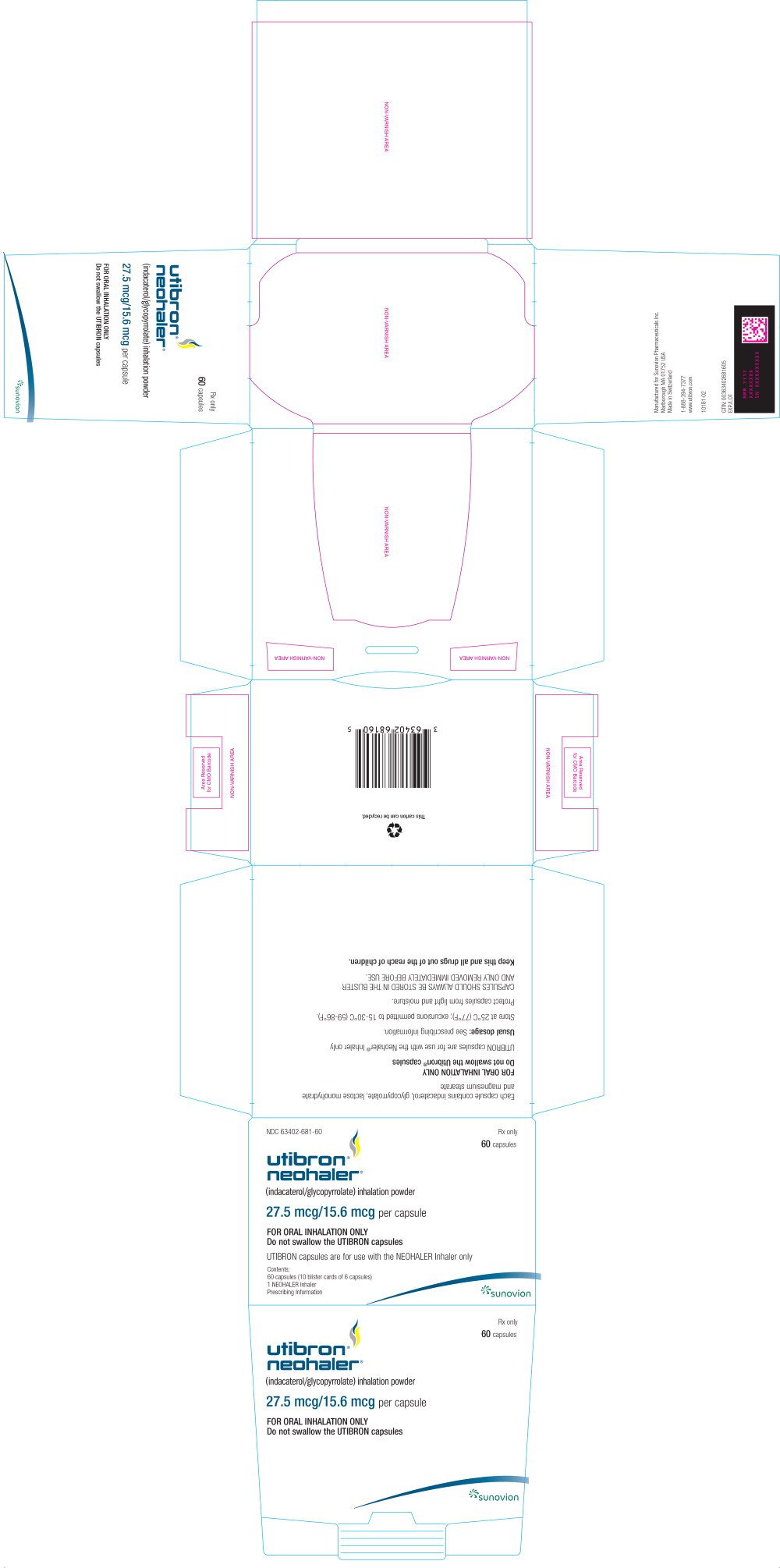
CARTON LABEL - PRINCIPAL DISPLAY PANEL – 27.5 mcg/15.6 mcg – 60 Capsules Carton
Rx only
NDC 63402-681-60
Utibron® Neohaler®
(indacaterol/glycopyrrolate) inhalation powder
27.5/15.6 mcg per capsule
FOR ORAL INHALATION ONLY
Do not swallow the UTIBRON capsules
UTIBRON capsules are for use with the NEOHALER Inhaler only
Contents:
60 Capsules (10 blister cards of 6 capsules)
1 NEOHALER Inhaler
Prescribing Information
Manufactured for Sunovion Pharmaceuticals Inc.
GTIN: 00363402681605
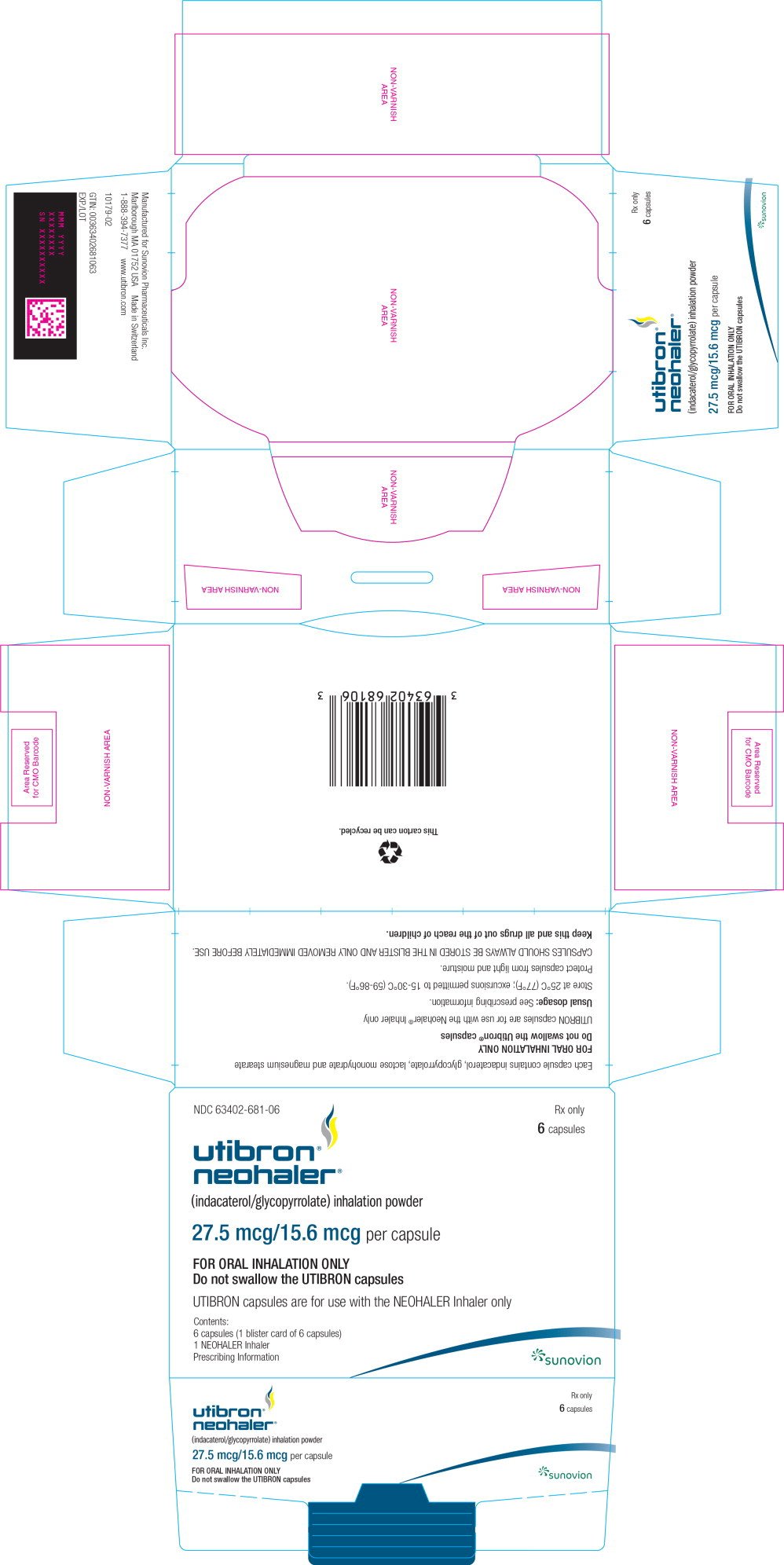
CARTON LABEL - PRINCIPAL DISPLAY PANEL – 27.5 mcg/15.6 mcg – 6 Capsules Carton
NDC 63402-681-06
Rx only
Utibron® Neohaler®
(indacaterol/glycopyrrolate) inhalation powder
27.5 mcg/15.6 mcg per capsule
FOR ORAL INHALATION ONLY
Do not swallow UTIBRON capsules
UTIBRON capsules are for use with the NEOHALER Inhaler only
Contents:
6 Capsules (1 blister card of 6 capsules)
1 NEOHALER Inhaler
Prescribing Information
Manufactured for Sunovion Pharmaceuticals Inc.
GTIN: 00363402681063
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL – Blisters
NDC NDC 63402-681-01
1 blister card of 6 capsules
Utibron® Neohaler®
(indacaterol/glycopyrrolate) inhalation powder
27.5 mcg indacaterol/15.6 mcg glycopyrrolate per capsule
Do not push the capsule through foil
For use with NEOHALER only
Do not swallow capsule
EXP/Lot
Mfd. for Sunovion Pharmaceuticals Inc.
| UTIBRON NEOHALER
indacaterol maleate and glycopyrrolate capsule |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Sunovion Pharmaceuticals Inc. (131661746) |
Frequently asked questions
More about Utibron Neohaler (glycopyrrolate / indacaterol)
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: bronchodilator combinations


 Follow the instructions below for using UTIBRON NEOHALER. You will breathe-in (inhale) the medicine in the UTIBRON capsules from the NEOHALER inhaler. If you have any questions,
Follow the instructions below for using UTIBRON NEOHALER. You will breathe-in (inhale) the medicine in the UTIBRON capsules from the NEOHALER inhaler. If you have any questions,