Sulfacetamide Eye Drops: Package Insert / Prescribing Info
Package insert / product label
Generic name: sulfacetamide sodium
Dosage form: ophthalmic solution
Drug class: Ophthalmic anti-infectives
Medically reviewed by Drugs.com. Last updated on Oct 14, 2024.
On This Page
Sulfacetamide Eye Drops Description
Sulfacetamide sodium ophthalmic solution, USP 10% is a sterile, topical anti-bacterial agent for ophthalmic use. The active ingredient is represented by the following structural formula:
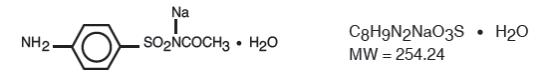
Chemical Name: N-Sulfanilylacetamide monosodium salt monohydrate.
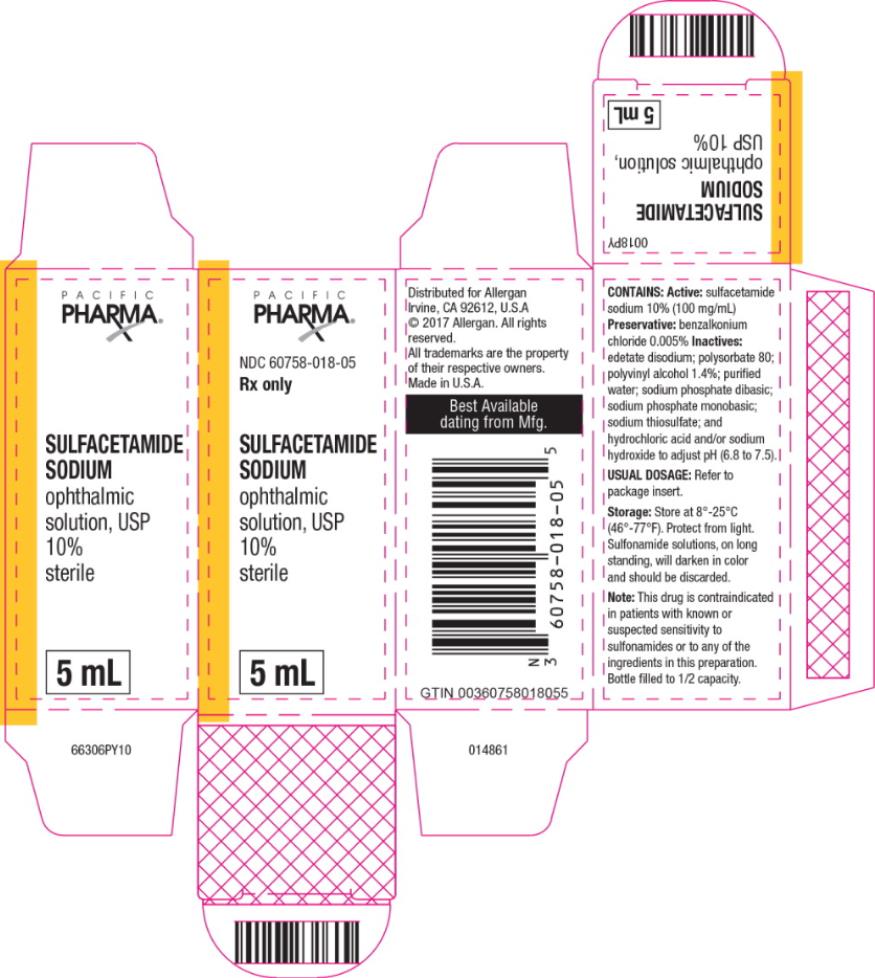
Contains: Active: sulfacetamide sodium 10% (100 mg/mL)
Preservative: benzalkonium chloride 0.005%
lnactives: edetate disodium; polysorbate 80; polyvinyl alcohol 1.4%; purified water; sodium phosphate dibasic; sodium phosphate monobasic; sodium thiosulfate; hydrochloric acid and/or sodium hydroxide to adjust pH (6.8 to 7.5).
Sulfacetamide Eye Drops - Clinical Pharmacology
Microbiology: The sulfonamides are bacteriostatic agents and the spectrum of activity is similar for all. Sulfonamides inhibit bacterial synthesis of dihydrofolic acid by preventing the condensation of the pteridine with aminobenzoic acid through competitive inhibition of the enzyme dihydropteroate synthetase. Resistant strains have altered dihydropteroate synthetase with reduced affinity for sulfonamides or produce increased quantities of aminobenzoic acid.
Topically applied sulfonamides are considered active against susceptible strains of the following common bacterial eye pathogens: Escherichia coli, Staphylococcus aureus, Streptococcus pneumoniae, Streptococcus (viridans group), Haemophilus influenzae, Klebsiella species, and Enterobacter species.
Topically applied sulfonamides do not provide adequate coverage against Neisseria species, Serratia marcescens and Pseudomonas aeruginosa. A significant percentage of staphylococcal isolates are completely resistant to sulfa drugs.
Indications and Usage for Sulfacetamide Eye Drops
Sulfacetamide sodium solution is indicated for the treatment of conjunctivitis and other superficial ocular infections due to susceptible microorganisms, and as an adjunctive in systemic sulfonamide therapy of trachoma: Escherichia coli, Staphylococcus aureus, Streptococcus pneumoniae, Streptococcus (viridans group), Haemophilus influenzae, Klebsiella species, and Enterobacter species.
Topically applied sulfonamides do not provide adequate coverage against Neisseria species, Serratia marcescens and Pseudomonas aeruginosa. A significant percentage of staphylococcal isolates are completely resistant to sulfa drugs.
Contraindications
Sulfacetamide sodium solution is contraindicated in individuals who have a hypersensitivity to sulfonamides or to any ingredient of the preparation.
Warnings
FOR TOPICAL EYE USE ONLY - NOT FOR INJECTION.
FATALITIES HAVE OCCURRED, ALTHOUGH RARELY, DUE TO SEVERE REACTIONS TO SULFONAMIDES INCLUDING STEVENS-JOHNSON SYNDROME, TOXIC EPIDERMAL NECROLYSIS, FULMINANT HEPATIC NECROSIS, AGRANULOCYTOSIS, APLASTIC ANEMIA AND OTHER BLOOD DYSCRASIAS. Sensitizations may recur when a sulfonamide is readministered, irrespective of the route of administration. Sensitivity reactions have been reported in individuals with no prior history of sulfonamide hypersensitivity. At the first sign of hypersensitivity, skin rash or other serious reaction, discontinue use of this preparation.
Precautions
General
Prolonged use of topical anti-bacterial agents may give rise to overgrowth of nonsusceptible organisms including fungi. Bacterial resistance to sulfonamides may also develop.
The effectiveness of sulfonamides may be reduced by the para-aminobenzoic acid present in purulent exudates.
Sensitization may recur when a sulfonamide is readministered irrespective of the route of administration, and cross-sensitivity between different sulfonamides may occur.
At the first sign of hypersensitivity, increase in purulent discharge, or aggravation of inflammation or pain, the patient should discontinue use of the medication and consult a physician (see WARNINGS).
Information for Patients
To avoid contamination, do not touch tip of container to the eye, eyelid or any surface.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted in animals or in humans to evaluate the possibility of these effects with ocularly administered sulfacetamide. Rats appear to be especially susceptible to the goitrogenic effects of sulfonamides, and long-term oral administration of sulfonamides has resulted in thyroid malignancies in these animals.
Pregnancy
Animal reproduction studies have not been conducted with sulfonamide ophthalmic preparations. Kernicterus may occur in the newborn as a result of treatment of a pregnant woman at term with orally administered sulfonamides. There are no adequate and well controlled studies of sulfonamide ophthalmic preparations in pregnant women and it is not known whether topically applied sulfonamides can cause fetal harm when administered to a pregnant woman. This product should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
Systemically administered sulfonamides are capable of producing kernicterus in infants of lactating women. Because of the potential for the development of kernicterus in neonates, a decision should be made whether to discontinue nursing or discontinue the drug taking into account the importance of the drug to the mother.
Adverse Reactions/Side Effects
Bacterial and fungal corneal ulcers have developed during treatment with sulfonamide ophthalmic preparations.
The most frequently reported reactions are local irritation, stinging and burning. Less commonly reported reactions include non-specific conjunctivitis, conjunctival hyperemia, secondary infections and allergic reactions.
Fatalities have occurred, although rarely, due to severe reactions to sulfonamides including Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anemia, and other blood dyscrasias (see WARNINGS).
Related/similar drugs
Sulfacetamide Eye Drops Dosage and Administration
For conjunctivitis and other superficial ocular infections: Instill one or two drops into the conjunctival sac(s) of the affected eye(s) every two to three hours initially. Dosages may be tapered by increasing the time interval between doses as the condition responds. The usual duration of treatment is seven to ten days.
For trachoma: Instill two drops into the conjunctival sac(s) of the affected eye(s) every two hours. Topical administration must be accompanied by systemic administration.
How is Sulfacetamide Eye Drops supplied
Sulfacetamide sodium ophthalmic solution, USP 10% is supplied sterile in opaque white LDPE plastic bottles and white dropper tips with white high impact polystyrene (HIPS) caps as follows:
5 mL in 10 mL bottle - NDC 60758-018-05
Storage:
Store at 8°-25°C (46°-77°F). Protect from light. Sulfonamide solutions, on long standing, will darken in color and should be discarded.
Revised: 9/2018
Distributed for: Allergan USA, Inc.
Madison, NJ 07940
© 2018 Allergan. All rights reserved.
All trademarks are the property of their respective owners.
v1.0USPI018
| SULFACETAMIDE
sulfacetamide sodium solution/ drops |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Pacific Pharma, Inc. (877645267) |
More about sulfacetamide sodium ophthalmic
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (4)
- Side effects
- Dosage information
- During pregnancy
- Drug class: ophthalmic anti-infectives
- En español