Primsol Oral Solution: Package Insert / Prescribing Info
Package insert / product label
Generic name: trimethoprim hydrochloride
Dosage form: oral solution
Drug class: Urinary anti-infectives
Medically reviewed by Drugs.com. Last updated on Dec 12, 2024.
On This Page
Primsol Oral Solution Description
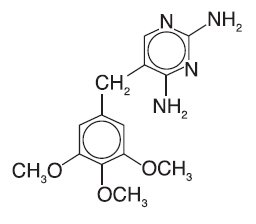
PRIMSOL (trimethoprim hydrochloride oral solution) is a solution of the synthetic antibacterial trimethoprim in water prepared with the aid of hydrochloric acid. Each 5 mL for oral administration contains trimethoprim hydrochloride equivalent to 50 mg trimethoprim and the inactive ingredients bubble gum flavor, fructose, glycerin, methylparaben, monoammonium glycyrrhizinate, povidone, propylparaben, propylene glycol, saccharin sodium, sodium benzoate, sorbitol, water and hydrochloric acid and/or sodium hydroxide to adjust pH to a range of 3.0 - 5.0. Trimethoprim is 2,4-diamino-5- (3,4,5-trimethoxybenzyl) pyrimidine. Trimethoprim is a white to cream-colored, odorless, bitter compound with a molecular formula of C 14H 18N 4O 3 and a molecular weight of 290.32 and the following structural formula:
Primsol Oral Solution - Clinical Pharmacology
Trimethoprim is rapidly absorbed following oral administration.
It exists in the blood as unbound, protein-bound and metabolized forms. Ten to twenty percent of trimethoprim is metabolized, primarily in the liver; the remainder is excreted unchanged in the urine. The principal metabolites of trimethoprim are the 1- and 3-oxides and the 3′- and 4′- hydroxy derivatives. The free form is considered to be the therapeutically active form. Approximately 44% of trimethoprim is bound to plasma proteins.
Mean peak plasma concentrations of approximately 1 mcg/mL occur 1 to 4 hours after oral administration of a single 100 mg dose. A single 200 mg dose will result in plasma concentrations approximately twice as high. The mean half-life of trimethoprim is approximately 9 hours. However, patients with severely impaired renal function exhibit an increase in the half-life of trimethoprim, which requires either dosage regimen adjustment or not using the drug in such patients (see DOSAGE AND ADMINISTRATION section). During a 13-week study of trimethoprim tablets administered at a dosage of 50 mg q.i.d., the mean minimum steady-state concentration of the drug was 1.1 mcg/mL. Steady-state concentrations were achieved within two to three days of chronic administration and were maintained throughout the experimental period.
Excretion of trimethoprim is primarily by the kidneys through glomerular filtration and tubular secretion. Urine concentrations of trimethoprim are considerably higher than are the concentrations in the blood. After a single oral dose of 100 mg, urine concentrations of trimethoprim ranged from 30 to 160 mcg/mL during the 0- to 4-hour period and declined to approximately 18 to 91 mcg/mL during the 8- to 24-hour period. A 200 mg single oral dose will result in trimethoprim urine concentrations approximately twice as high. After oral administration, 50% to 60% of trimethoprim is excreted in the urine within 24 hours, approximately 80% of this being unmetabolized trimethoprim.
Trimethoprim half-life, clearance, and volume of distribution vary with age. Excluding newborns, an apparent trend of increasing half-life, volume of distribution, and decreasing clearance is observed with increasing age until adulthood.
Since normal vaginal and fecal flora are the source of most pathogens causing urinary tract infections, it is relevant to consider the distribution of trimethoprim into these sites.
Concentrations of trimethoprim in vaginal secretions are consistently greater than those found simultaneously in the serum, being typically 1.6 times the concentrations of simultaneously obtained serum samples. Sufficient trimethoprim is excreted in the feces to markedly reduce or eliminate trimethoprim-susceptible organisms from the fecal flora. The dominant non- Enterobacteriaceae fecal organisms, Bacteroides spp. and Lactobacillus spp., are not susceptible to trimethoprim concentrations obtained with the recommended dosage. Trimethoprim also concentrates into middle ear fluid (MEF) very efficiently. In a study in children aged 1 to 12 years, administration of a single 4 mg/kg dose resulted in a mean peak MEF concentration of 2.0 mcg/mL.
Trimethoprim also passes the placental barrier and is excreted in breast milk.
Microbiology
Mechanism of Action
Trimethoprim blocks the production of tetrahydrofolic acid from dihydrofolic acid by binding to and reversibly inhibiting the required enzyme, dihydrofolate reductase. This binding is very much stronger for the bacterial enzyme than for the corresponding mammalian enzyme. Thus, trimethoprim selectively interferes with bacterial biosynthesis of nucleic acids and proteins.
Resistance
Resistance to trimethorprim may be conferred by a variety of mechanisms including cell wall impermeability, overproduction of the chromosomal dihydrofolate reductase (DHFR) enzyme, production of a resistant chromosomal DHFR enzyme or production of a plasmid-mediated thrimethoprim-resistant DHFR enzyme, Acinetobacter baumannii/Acinetobacter calcoaceticus complex, Burkholderia cepacia complex, Pseudomonas aeruginosa, Stenotrophomonas maltophilia are intrinsically resistant to trimethoprim. Non- Enterobacteriaceae fecal organisms, Bacteroides spp. and Lactobacillus spp. are not susceptible to trimethoprim at the concetrations obtained with the recommended dosage. Moraxella catarrhalis isolates were found consistently resistant to trimethoprim. Entercoccus spp. ( E. faecalis, E. faecium, E. gallinarum/E. casseliflavus) may appear active in vitro to trimethoprim but are not effective clinically and should not be reported as susceptible.
Antimicrobial Activity
Trimethoprim has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section
Susceptibility Test Methods
When available, the clinical microbiology laboratory should provide cumulative reports of in vitro susceptibility test results for antimicrobial drugs used in local hospitals and practice areas to the physician as periodic reports that describe the susceptibility profile of nosocomial and community-acquired pathogens. These reports should aid the physician in selecting an antibacterial drug for treatment.
Dilution techniques
Quantitative methods are used to determine antimicrobial minimun inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure (broth and/or agar). 1,3 The MIC values should be interpreted according to the criteria provided in Table 1.
Diffusion techniques
Quanitative methods that require measurement of zone diameters can also provide reproducible estimates of the suseptibility of bacteria to antimicrobial compounds. The zone size should be determined using a standardized test method. 2,3 This procedure uses paper disks impregnated with 5mcg trimethoprim to test the susceptibility of bacteria to trimethoprim. The disc diffusion breakpoints are provided in Table 1.
| Pathogen | Minimum Inhibitory Concentrations
(mcg/mL) | Zone Diameters
(mm) |
||||
|---|---|---|---|---|---|---|
| S | I | R | S | I | R | |
| Note: S. pneumoniae are not susceptible to trimethoprim at the concentrations obtainced with the recommended dosage. | ||||||
| Enterobacteriaceae | ≤8 | ≥16 | ≥16 | 11-15 | ≤10 | |
| Coagulase negative staphylococci (including S. saprophyticus | ≤8 | ≥16 | ≥16 | 11-15 | ≤10 | |
| Haemophilus influenzae | ≤0.5 | 1-2 | ≥4 | ≥16 | 11-15 | ≤10 |
A report of Susceptible (S) indicates that the antimicrobial drug is likely to inhibit growth of the pathogen if the antimicrobial drug reaches the concentration usually achievable at the site of infection. A report of Intermediate (I) indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where a high dose of the drug can be used. This category also provides a buffer zone that prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of Resistant (R) indicates that the antimicrobial drug is not likely to inhibit growth of the pathogen if the antimicrobial drug reaches the concentration usually achievable at the infection site; other therapy should be selected.
Quality Control
Standardized susceptibility test procedures require the use of laboratory controls to monitor and ensure the accuracy and precision of supplies and reagents used in the assay, and the techniques of the individuals performing the test 1,2,3. Standard trimethoprim powder should provide the following range of MIC values noted in Table 2. For the diffusion technique using the 5 mcg disk, the criteria in Table 2 should be achieved.
| QC Strain | Minimum Inhibitory Concentrations
(mcg/mL) | Zone Diameters
(mm) |
|---|---|---|
| Enterococcus faecalis ATCC 29212 | 0.12-0.50 | -- |
| Escherichia coli ATCC 25922 | 0.5-2.0 | 21-28 |
| Haemophilus influenzae ATCC 49247 | 0.06-0.50 | 27-33 |
| Staphylococcus aureus ATCC 29213 | 1-4 | -- |
| Staphylococcus aureus ATCC 25923 | -- | 19-26 |
| Streptococcus pneumoniae ATCC 49619 | 1-4 | -- |
| Pseudomonas aeruginosa ATCC 27853 | >64 | -- |
Indications and Usage for Primsol Oral Solution
PRIMSOL Solution is indicated for the treatment of infections caused by susceptible strains of the designated microorganisms in the conditions listed below.
Pediatric Patients
Acute Otitis Media
For the treatment of acute otitis media due to susceptible strains of Streptococcus pneumoniae and Haemophilus influenzae.
NOTE: Moraxella catarrhalis isolates were found consistently resistant to trimethoprim in vitro. Therefore, when infection with Moraxella catarrhalis is suspected, the use of alternative antimicrobial agents should be considered.
PRIMSOL is not indicated for prophylactic or prolonged administration in otitis media at any age.
Adults
Urinary Tract Infections
For the treatment of initial episodes of uncomplicated urinary tract infections due to susceptible strains of the following organisms:
Escherichia coli, Proteus mirabilis, Klebsiella pneumoniae, Enterobacter species and coagulase-negative Staphylococcus species, including S. saprophyticus.
Cultures and susceptibility tests should be performed to determine the susceptibility of the bacteria to trimethoprim. Therapy may be initiated prior to obtaining the results of these tests.
Clinical Studies
The results of one multicenter, 30-day, comparative, randomized clinical trial without tympanocentesis in 262 pediatric patients with acute otitis media (AOM) are shown below. In this clinical trial, strict evaluability criteria were used to determine clinical response.
| PRIMSOL | SMX + TMP * | |
|---|---|---|
|
||
| Enrolled | 133 | 129 |
| Evaluable | 130 | 129 |
| Clinical Cure | 64/130 (49%) | 63/129 (49%) |
| Clinical Improvement | 30/130 (23%) | 31/129 (24%) |
| Relapse/Recurrence | 19/130 (15%) | 18/129 (14%) |
| Outcome (based on 95% confidence interval) | PRIMSOL equivalent to TMP + SMX | |
The results of an uncontrolled 30-day trial with tympanocentesis in 120 pediatric patients with AOM are shown below:
| Number of patients | ||
|---|---|---|
| Enrolled | 120 | |
| Clinically Evaluable | 102 | |
| Microbiologically Evaluable | 58 | |
| Clinical Cure | 50/102 (49%) | |
| Clinical Improvement | 22/102 (22%) | |
| Clinical Relapse/Recurrence | 20/102 (20%) | |
| Microbiologic Eradication Rates n=58 | Day 5
post-therapy | Day 20
post-therapy |
| Streptococcus pneumoniae | 16/20 (80%) | 14/20 (70%) |
| Haemophilus influenzae | 14/17 (82%) | 13/17 (77%) |
Moraxella catarrhalis, isolated from five patients, was found consistently resistant to trimethoprim in vitro.
Contraindications
PRIMSOL is contraindicated in individuals hypersensitive to trimethoprim and in those with documented megaloblastic anemia due to folate deficiency.
Warnings
Experience with trimethoprim alone is limited, but it has been reported rarely to interfere with hematopoiesis, especially when administered in large doses and/or for prolonged periods.
The presence of clinical signs such as sore throat, fever, pallor or purpura may be early indications of serious blood disorders.
Precautions
General
Trimethoprim should be given with caution to patients with possible folate deficiency. Folates may be administered concomitantly without interfering with the antibacterial action of trimethoprim. Trimethoprim should also be given with caution to patients with impaired renal or hepatic function. If any clinical signs of a blood disorder are noted in a patient receiving trimethoprim, a complete blood count should be obtained and the drug discontinued if a significant reduction in the count of any formed blood element is found.
Drug Interactions
PRIMSOL may inhibit the hepatic metabolism of phenytoin. Trimethoprim, given at a common clinical dosage, increased the phenytoin half-life by 51% and decreased the phenytoin metabolic clearance rate by 30%. When administering these drugs concurrently, one should be alert for possible excessive phenytoin effect.
Drug/Laboratory Test Interactions
Trimethoprim can interfere with a serum methotrexate assay as determined by the competitive binding protein technique (CBPA) when a bacterial dihydrofolate reductase is used as the binding protein. No interference occurs, however, if methotrexate is measured by a radioimmunoassay (RIA).
The presence of trimethoprim may also interfere with the Jaffé alkaline picrate reaction assay for creatinine resulting in overestimations of about 10% in the range of normal values.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic potential have not been conducted with trimethoprim. Trimethoprim was demonstrated to be non-mutagenic in the Ames assay. No chromosomal damage was observed in human leukocytes cultured in vitro with trimethoprim; the concentration used exceeded blood levels following therapy with PRIMSOL. No adverse effects on fertility or general reproductive performance were observed in rats given trimethoprim in oral dosages as high as 70 mg/kg/day for males and 14 mg/kg/day for females.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Trimethoprim has been shown to be teratogenic in the rat when given in doses 40 times the human dose. In some rabbit studies, the overall increase in fetal loss (dead and resorbed and malformed conceptuses) was associated with doses 6 times the human therapeutic dose.
While there are no large well-controlled studies on the use of trimethoprim in pregnant women, Brumfitt and Pursell, 4 in a retrospective study, reported the outcome of 186 pregnancies during which the mother received either placebo or trimethoprim in combination with sulfamethoxazole. The incidence of congenital abnormalities was 4.5% (3 of 66) in those who received placebo and 3.3% (4 of 120) in those receiving trimethoprim plus sulfamethoxazole. There were no abnormalities in the 10 children whose mothers received the drug during the first trimester. In a separate survey, Brumfitt and Pursell also found no congenital abnormalities in 35 children whose mothers had received trimethoprim plus sulfamethoxazole at the time of conception or shortly thereafter.
Because trimethoprim may interfere with folic acid metabolism, PRIMSOL should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Adverse Reactions/Side Effects
To report SUSPECTED ADVERSE REACTIONS, contact Allegis Holdings, LLC at 1-866-633-9033, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Adverse Events Reported During Pediatric Clinical Trials With PRIMSOL
The following table lists those drug-related adverse events reported most frequently during the clinical trials in pediatric patients aged 6 months to 12 years. Most of these events were determined to be mild. The incidence of drug related adverse events was significantly lower for PRIMSOL, which was most apparent for those events related to skin/appendages as a body system.
| Drug-related Adverse Event | Percent of Pediatric Patients | |
|---|---|---|
| PRIMSOL
(N=310) | SMX + TMP
*
(N=197) |
|
|
||
| Body as a whole | ||
| abdominal pain | <1 | 2.5 |
| Digestive system | ||
| diarrhea | 4.2 | 4.6 |
| vomiting | 1.6 | 1.5 |
| Skin/Appendages | ||
| rash | 1.3 | 6.1 |
An increase in lymphocytes and eosinophils was noted in some pediatric patients following treatment with PRIMSOL or sulfamethoxazole + trimethoprim oral suspension.
Adverse Reactions Reported For Trimethoprim
In addition to the adverse events listed above which have been observed in pediatric patients receiving PRIMSOL, the following adverse reactions and altered laboratory tests have been previously reported for trimethoprim and therefore, may occur with PRIMSOL therapy:
Dermatologic reactions: pruritus and exfoliative dermatitis. At the recommended adult dosage regimens of 100 mg b.i.d., or 200 mg q.d., each for 10 days, the incidence of rash is 2.9% to 6.7%. In clinical studies which employed high doses of trimethoprim in adults, an elevated incidence of rash was noted. These rashes were maculopapular, morbilliform, pruritic and generally mild to moderate, appearing 7 to 14 days after the initiation of therapy.
Gastrointestinal reactions: Epigastric distress, nausea, and glossitis.
Hematologic reactions: Thrombocytopenia, leukopenia, neutropenia, megaloblastic anemia and methemoglobinemia.
Metabolic reactions: Hyperkalemia, hyponatremia.
Miscellaneous reactions: Fever, elevation of serum transaminase and bilirubin, and increases in BUN and serum creatinine levels.
Related/similar drugs
Overdosage
Acute
Signs of acute overdosage with trimethoprim may appear following ingestion of 1 gram or more of the drug and include nausea, vomiting, dizziness, headaches, mental depression, confusion and bone marrow depression (see OVERDOSAGE-Chronic).
Treatment consists of gastric lavage and general supportive measures. Acidification of the urine will increase renal elimination of trimethoprim. Peritoneal dialysis is not effective and hemodialysis only moderately effective in eliminating the drug.
Chronic
Use of trimethoprim at high doses and/or for extended periods of time may cause bone marrow depression manifested as thrombocytopenia, leukopenia and/or megaloblastic anemia. If signs of bone marrow depression occur, trimethoprim should be discontinued and the patient should be given leucovorin, 3 to 6 mg intramuscularly daily for three days, or as required to restore normal hematopoiesis.
Primsol Oral Solution Dosage and Administration
Acute Otitis Media in Pediatric Patients
The recommended dose for pediatric patients with acute otitis media is 10 mg/kg trimethoprim per 24 hours, given in divided doses every 12 hours for 10 days. The following table is a guideline for the attainment of this dosage:
| Weight | Dose (every 12 hours) | ||
|---|---|---|---|
| lb | kg | tsp | mL |
| 11 | 5 | ½ | 2.5 |
| 22 | 10 | 1 | 5 |
| 33 | 15 | 1½ | 7.5 |
| 44 | 20 | 2 | 10 |
| 55 | 25 | 2½ | 12.5 |
| 66 | 30 | 3 | 15 |
| 77 | 35 | 3½ | 17.5 |
| ≥88 | ≥40 | 4 | 20 |
How is Primsol Oral Solution supplied
PRIMSOL (trimethoprim hydrochloride oral solution) is a dye-free, alcohol-free, bubble gum flavored, oral solution containing trimethoprim hydrochloride equivalent to 50 mg of trimethoprim in each 5 mL.
NDC 71297-500-20: 20 mL (2/3 ounce)
NDC 71297-500-16: 473 mL (1 Pint)
References
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility for Bacteria that Grow Aerobically; Approved Standard-Tenth Edition. CLSI document M07-A10 [2015], Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests; Approved Standard-Twelfth Edition. CLSI document M02-A12 [2015], Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Tests; Twenty-sixth Informational Supplement, CLSI document M100-S26 [2016], Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA.
- Brumfitt W, Prusell R: Trimethoprim/Sulfamethoxazole in the treatment of Bacteriuria in Women, J Infect Dis128 (suppl): S657-S663, 1973.
| PRIMSOL
trimethoprim hydrochloride solution |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Allegis Holdings, LLC (080556861) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Halo Pharmaceutical | 829609168 | manufacture(71297-500) | |
Frequently asked questions
More about Primsol (trimethoprim)
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: urinary anti-infectives
- Breastfeeding

