Dipivefrin: Package Insert / Prescribing Info
Package insert / product label
Generic name: dipivefrin hydrochloride
Dosage form: ophthalmic solution
Drug class: Ophthalmic glaucoma agents
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
Dipivefrin Description
Dipivefrin hydrochloride ophthalmic solution is a sterile, isotonic solution. Dipivefrin hydrochloride is a white, crystalline powder, freely soluble in water. It is classified as a sympathomimetic agent and has the following structural formula:
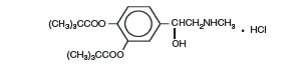
Molecular Formula: C19H29O5N • HCI Molecular weight: 387.90
Chemical Name: [±] -3,4-Dihydroxy-α-[(methylamino)methyl]benzyl alcohol 3,4-dipivalate hydrochloride.
Each mL Contains:
ACTIVE: Dipivefrin hydrochloride 1 mg (0.1%)
INACTIVES: Edetate Disodium, Sodium Chloride, Purified Water, Hydrochloric Acid and/or Sodium Hydroxide may be added to adjust pH (2.5 - 3.5).
PRESERVATIVE ADDED: Benzalkonium Chloride 0.005%.
Dipivefrin - Clinical Pharmacology
Dipivefrin hydrochloride, is a member of a class of drugs known as prodrugs. Prodrugs are usually not active in themselves and require biotransformation to the parent compound before therapeutic activity is seen. These modifications are undertaken to enhance absorption, decrease side effects and enhance stability and comfort, thus making the parent compound a more useful drug. Enhanced absorption makes the prodrug a more efficient delivery system for the parent drug because less drug will be needed to produce the desired therapeutic response.
Dipivefrin hydrochloride is a prodrug of epinephrine formed by the diesterification of epinephrine and pivalic acid. The addition of pivaloyl groups to the epinephrine molecule enhances its lipophilic character and, as a consequence, its penetration into the anterior chamber.
Dipivefrin hydrochloride is converted to epinephrine inside the human eye by enzyme hydrolysis. The liberated epinephrine, an adrenergic agonist, appears to exert its action by decreasing aqueous production and by enhancing outflow facility. The dipivefrin hydrochloride prodrug delivery system is a more efficient way of delivering the therapeutic effects of epinephrine, with fewer side effects than are associated with conventional epinephrine therapy.
The onset of action with one drop of dipivefrin hydrochloride ophthalmic solution occurs about 30 minutes after treatment, with maximum effect seen at about one hour.
Using a prodrug means that less drug is needed for therapeutic effect since absorption is enhanced with the prodrug. Dipivefrin hydrochloride 0.1% was judged less irritating than a 1% solution of epinephrine hydrochloride or bitartrate. In addition, only 8 of 455 patients (1.8%) treated with dipivefrin reported discomfort due to photophobia, glare or light sensitivity.
Indications and Usage for Dipivefrin
Dipivefrin Hydrochloride Ophthalmic Solution is indicated as initial therapy for the control of intraocular pressure in chronic open-angle glaucoma. Patients responding inadequately to other antiglaucoma therapy may respond to addition of dipivefrin.
In controlled and open-label studies of glaucoma, dipivefrin demonstrated a statistically significant intraocular pressure-lowering effect. Patients using dipivefrin twice daily in studies with mean durations of 76 to 146 days experienced mean pressure reductions ranging from 20 to 24%.
Therapeutic response to 0.1% dipivefrin twice-daily is somewhat less than 2% epinephrine twice daily. Controlled studies showed statistically significant differences in lowering of intraocular pressure between 0.1% dipivefrin and 2% epinephrine. In controlled studies in patients with a history of epinephrine intolerance, only 3% of patients treated with dipivefrin exhibited intolerance, while 55% of those treated with epinephrine again developed an intolerance.
Therapeutic response to 0.1% dipivefrin twice-daily is comparable to 2% pilocarpine 4 times-daily. In controlled clinical studies comparing 0.1% dipivefrin and 2% pilocarpine, there were no statistically significant differences in the maintenance of IOP levels for the two medications.
Dipivefrin hydrochloride does not produce miosis or accommodative spasm which cholinergic agents are known to produce. The blurred vision and night blindness often associated with miotic agents are not present with dipivefrin therapy. Patients with cataracts avoid the inability to see around lenticular opacities caused by constricted pupil.
Contraindications
Dipivefrin hydrochloride should not be used in patients with narrow angles since any dilation of the pupil may predispose the patient to an attack of angle-closure glaucoma. This product is contraindicated in patients who are hypersensitive to any of its components.
Precautions
General
APHAKIC PATIENTS: Macular edema has been shown to occur in up to 30% of aphakic patients treated with epinephrine. Discontinuation of epinephrine generally results in reversal of the maculopathy.
Information for patients
To avoid contamination, do not touch tip of container to the eye, eyelid or any surface.
Pregnancy
Pregnancy Category B: Reproduction studies have been performed in rats and rabbits at daily oral doses up to 10 mg/kg body weight (5 mg/kg in teratogenicity studies), and have revealed no evidence of impaired fertility or harm to the fetus due to dipivefrin. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Adverse Reactions/Side Effects
Cardiovascular Effects
Tachycardia, arrhythmias and hypertension have been reported with ocular administration of epinephrine.
Local Effects
The most frequent side effects reported with dipivefrin hydrochloride alone were injection in 6.5% of patients and burning and stinging in 6%. Follicular conjunctivitis, mydriasis and allergic reactions to dipivefrin have been reported infrequently. Epinephrine therapy can lead to adrenochrome deposits in the conjunctiva and cornea.
Related/similar drugs
Dipivefrin Dosage and Administration
Initial Glaucoma Therapy
The usual dosage of Dipivefrin Hydrochloride Ophthalmic Solution 0.1% is one drop in the eye(s) every 12 hours.
Replacement with Dipivefrin Hydrochloride Ophthalmic Solution
When patients are being transferred to dipivefrin from antiglaucoma agents other than epinephrine, on the first day continue the previous medication and add one drop of dipivefrin to each eye every 12 hours. On the following day, discontinue the previously used antiglaucoma agent and continue with dipivefrin.
Transferring Patients from Conventional Epinephrine Therapy to Dipivefrin Hydrochloride Ophthalmic Solution
In transferring patients from conventional epinephrine therapy to dipivefrin, simply discontinue the epinephrine medication and institute the Dipivefrin Hydrochloride Ophthalmic Solution regimen
How is Dipivefrin supplied
Dipivefrin Hydrochloride Ophthalmic Solution USP, 0.1%, is supplied sterile in plastic dropper bottles as follows:
5 mL - Prod. No. 27807
10 mL - Prod. No. 27809
15 mL - Prod. No. 27811
FOR OPHTHALMIC USE ONLY
DO NOT USE IF IMPRINTED NECKBAND IS NOT INTACT.
| DIPIVEFRIN HYDROCHLORIDE
dipivefrin hydrochloride solution/ drops |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Bausch & Lomb Incorporated |
More about dipivefrin ophthalmic
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: ophthalmic glaucoma agents
