Cefizox Injection: Package Insert / Prescribing Info
Package insert / product label
Generic name: ceftizoxime sodium
Dosage form: Injection
Drug class: Third generation cephalosporins
J Code (medical billing code): J0715 (Per 500 mg, injection)
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefizox and other antibacterial drugs, Cefizox should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
FOR INTRAVENOUS USE
Cefizox Injection Description
Cefizox® (ceftizoxime injection) in the Galaxy® plastic container (PL 2040) contains ceftizoxime as ceftizoxime sodium. It is a sterile, semisynthetic, broad spectrum, cephalosporin antibiotic for intravenous administration.
Chemically, it is the sodium (6R, 7R)7[2-(2imino4 thiazolin-4-yl) glyoxylamido]8oxo5thia1azabicyclo[4.2.0.]oct2ene2carboxylate 7²-(Z)-(O-methyloxime). The molecular formula is C13H12N5NaO5S2 and the molecular weight is 405.38. The structural formula of ceftizoxime sodium is as follows:
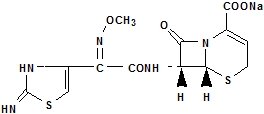
Cefizox (ceftizoxime injection) in the Galaxy® plastic container is a frozen iso-osmotic, sterile, nonpyrogenic premixed 50 mL solution containing 1 g or 2 g of ceftizoxime as ceftizoxime sodium. Dextrose, USP has been added to these dosages to adjust osmolality (approximately 1.9 g and 950 mg to the 1 g and 2 g dosages as dextrose hydrous, respectively). Thawed solutions range from very pale yellow to yellow. The pH of thawed solutions range from 5.5 to 8.0. After thawing to room temperature, the solution is intended for intravenous use only.
The Galaxy® container is fabricated from a specially designed multilayer plastic (PL 2040). Solutions are in contact with the polyethylene layer of this container and can leach out certain chemical components of the plastic in very small amounts within the expiration dating period. The suitability of the plastic has been confirmed in tests in animals according to the USP biological tests for plastic containers, as well as by tissue culture toxicity studies.
Cefizox Injection - Clinical Pharmacology
Following IV administration of 1, 2, and 3 gram doses of Cefizox to normal volunteers, the following serum levels were obtained.
| Dose | 5 min | 10 min | 30 min | 1 hr | 2 hr | 4 hr | 8 hr |
|
|||||||
| 1 gram | ND* | ND* | 60.5 | 38.9 | 21.5 | 8.4 | 1.4 |
| 2 grams | 131.8 | 110.9 | 77.5 | 53.6 | 33.1 | 12.1 | 2.0 |
| 3 grams | 221.1 | 174.0 | 112.7 | 83.9 | 47.4 | 26.2 | 4.8 |
A serum halflife of approximately 1.7 hours was observed after IV administration.
Cefizox is 30% protein bound.
Cefizox is not metabolized, and is excreted virtually unchanged by the kidneys in 24 hours. This provides a high urinary concentration. Concentrations greater than 6000 μg/mL have been achieved in the urine by 2 hours after a 1 gram dose of Cefizox intravenously. Probenecid slows tubular secretion and produces even higher serum levels, increasing the duration of measurable serum concentrations.
Cefizox achieves therapeutic levels in various body fluids, e.g., cerebrospinal fluid (in patients with inflamed meninges), bile, surgical wound fluid, pleural fluid, aqueous humor, ascitic fluid, peritoneal fluid, prostatic fluid and saliva, and in the following body tissues: heart, gallbladder, bone, biliary, peritoneal, prostatic, and uterine.
In clinical experience to date, no disulfiramlike reactions have been reported with Cefizox.
Microbiology
The bactericidal action of ceftizoxime results from inhibition of cellwall synthesis. Ceftizoxime is highly resistant to a broad spectrum of betalactamases (penicillinase and cephalosporinase), including Richmond types I, II, III, TEM, and IV, produced by both aerobic and anaerobic grampositive and gramnegative organisms. Ceftizoxime has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections described in the INDICATIONS AND USAGE section:
Aerobic Gram-Positive Microorganisms
Staphylococcus aureus (including penicillinase producing strains)
NOTE: Methicillinresistant staphylococci are resistant to cephalosporins, including ceftizoxime.
Staphylococcus epidermidis (including penicillinase producing strains)
Streptococcus agalactiae
Streptococcus pneumoniae
Streptococcus pyogenes
NOTE: A streptococcal isolate that is susceptible to penicillin can be considered susceptible to ceftizoxime.4
NOTE: Ceftizoxime is usually inactive against most strains of Enterococcus faecalis.
Aerobic Gram-Negative Microorganisms
Enterobacter spp.
Escherichia coli
Haemophilus influenzae (including ampicillinresistant strains)
Klebsiella pneumoniae
Morganella morganii
Neisseria gonorrhoeae
Proteus mirabilis
Proteus vulgaris
Providencia rettgeri
Pseudomonas aeruginosa
Serratia marcescens
Anaerobic Microorganisms
Bacteroides spp.
Peptococcus spp.
Peptostreptococcus spp.
The following in vitro data are available, but their clinical significance is unknown. At least 90% of the following microorganisms exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for ceftizoxime. However, the safety and effectiveness of ceftizoxime in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled clinical trials.
Susceptibility Testing Methods:
Dilution techniques
Quantitative methods are used to determine antimicrobial minimum inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method1 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of ceftizoxime powder. The MIC values should be interpreted according to the following criteria:
| MIC (μg/mL) | Interpretation |
| ≤8 | Susceptible (S) |
| 16-32 | Intermediate (I) |
| ≥64 | Resistant (R) |
| MIC (μg/mL) | Interpretation† |
|
|
| ≤2 | Susceptible (S) |
| MIC (μg/mL) | Interpretation† |
|
|
| ≤0.5 | Susceptible (S) |
A report of “Susceptible” indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of “Intermediate” indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone, which prevents small-uncontrolled technical factors from causing major discrepancies in interpretation. A report of “Resistant” indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable, other therapy should be selected.
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard ceftizoxime powder should provide the following MIC values:
| Microorganism | MIC (μg/mL) |
| Escherichia coli ATCC 25922 | 0.030.12 |
| Haemophilus influenzae ATCC 49247 | 0.06-0.5 |
| Neisseria gonorrhoeae ATCC 49226 | 0.008-0.03 |
| Pseudomonas aeruginosa ATCC 27853 | 16-64 |
| Staphylococcus aureus ATCC 29213 | 28 |
Diffusion Techniques
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure2 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 30-μg ceftizoxime to test the susceptibility of microorganisms to ceftizoxime.
Reports from the laboratory providing results of the standard single-disk susceptibility test with a 30-μg ceftizoxime disk should be interpreted according to the following criteria:
| Zone Diameter (mm) | Interpretation |
| ≥ 20 | Susceptible (S) |
| 15-19 | Intermediate (I) |
| ≤ 14 | Resistant (R) |
| Zone Diameter (mm) | Interpretation† |
|
|
| ≥ 26 | Susceptible (S) |
| Zone Diameter (mm) | Interpretation† |
|
|
| ≥ 38 | Susceptible (S) |
Interpretation should be as stated above for results using dilution techniques. Interpretation involves correlation of the diameter obtained in the disk test with the MIC for ceftizoxime.
As with standardized dilution techniques, diffusion methods require the use of laboratory control microorganisms that are used to control the technical aspects of the laboratory procedures. For the diffusion technique, the 30-μg ceftizoxime disk should provide the following zone diameters in these laboratory test quality control strains:
| Microorganism | Zone Diameter (mm) |
| Escherichia coli ATCC 25922 | 30-36 |
| Haemophilus influenzae ATCC 49247 | 29-39 |
| Neisseria gonorrhoeae ATCC 49226 | 42-51 |
| Pseudomonas aeruginosa ATCC 27853 | 12-17 |
| Staphylococcus aureus ATCC 25923 | 27-35 |
Anaerobic Techniques
For anaerobic bacteria, the susceptibility to ceftizoxime as MICs can be determined by standardized test methods. Agar dilution results can vary widely when using ceftizoxime. It is recommended that broth microdilution method be used when possible.3 The MIC values obtained should be interpreted according to following criteria:
| MIC(μg/mL) | |||
| Broth dilution | Agar dilution | Interpretation | |
| ≤ 16 | ≤ 32 | Susceptible (S) | |
| 32 | 64 | Intermediate (I) | |
| ≥ 64 | ≥ 128 | Resistant (R) | |
Interpretation is identical to that described in Susceptibility Testing: Dilution Techniques.
As with other susceptibility techniques, the use of laboratory control microorganisms is required to control the technical aspects of the laboratory standardized procedures. Standardized ceftizoxime powder should provide the following MIC values:
| Microorganism | MIC(μg/mL) | |
| Broth dilution | Agar dilution | |
| Eubacterium lentum ATCC 43055 | 16-64 | 16-64 |
| Bacteriodes thetaiotaomicron ATCC 29741 | --- | 4-16 |
Susceptibility Testing for Pseudomonas in Urinary Tract Infections
Most strains of Pseudomonas aeruginosa are moderately susceptible to ceftizoxime.
Ceftizoxime achieves high levels in the urine (greater than 6000 μg/mL at 2 hours with 1 gram IV) and, therefore, the following zone sizes should be used when testing ceftizoxime for treatment of urinary tract infections caused by Pseudomonas aeruginosa.
Susceptible organisms produce zones of 20 mm or greater, indicating that the
test organism is likely to respond to therapy.
Organisms that produce zones of 11 to 19 mm are expected to be susceptible
when the infection is confined to the urinary tract (in which high antibiotic levels
are attained).
Resistant organisms produce zones of 10 mm or less, indicating that other
therapy should be selected.
Indications and Usage for Cefizox Injection
Cefizox (ceftizoxime injection) is indicated in the treatment of infections due to susceptible strains of the microorganisms listed below.
Lower Respiratory Tract Infections caused by Klebsiella spp.; Proteus mirabilis; Escherichia coli; Haemophilus influenzae including ampicillinresistant strains; Staphylococcus aureus (penicillinase and nonpenicillinaseproducing); Serratia spp.; Enterobacter spp.; Bacteroides spp.; and Streptococcus spp. including S. pneumoniae, but excluding enterococci.
Urinary Tract Infections caused by Staphylococcus aureus (penicillinase and nonpenicillinaseproducing); Escherichia coli; Pseudomonas spp. including P.aeruginosa; Proteus mirabilis; P. vulgaris; Providencia rettgeri (formerly Proteus rettgeri) and Morganella morganii (formerly Proteus morganii); Klebsiella spp.; Serratia spp. including S. marcescens; and Enterobacter spp.
Gonorrhea including uncomplicated cervical and urethral gonorrhea caused by Neisseria gonorrhoeae.
Pelvic Inflammatory Disease caused by Neisseria gonorrhoeae, Escherichia coli or Streptococcus agalactiae. NOTE: Ceftizoxime, like other cephalosporins, has no activity against Chlamydia trachomatis. Therefore, when cephalosporins are used in the treatment of patients with pelvic inflammatory disease and C. trachomatis is one of the suspected pathogens, appropriate antichlamydial coverage should be added.
IntraAbdominal Infections caused by Escherichia coli; Staphylococcusepidermidis; Streptococcus spp. (excluding enterococci); Enterobacter spp.; Klebsiella spp.; Bacteroides spp. including B. fragilis; and anaerobic cocci, including Peptococcus spp. and Peptostreptococcus spp.
Septicemia caused by Streptococcus spp. including S. pneumoniae (but excluding enterococci); Staphylococcus aureus (penicillinase and nonpenicillinaseproducing); Escherichia coli; Bacteroides spp. including B. fragilis; Klebsiella spp.; and Serratia spp.
Skin and Skin Structure Infections caused by Staphylococcus aureus (penicillinase and nonpenicillinaseproducing); Staphylococcus epidermidis; Escherichia coli; Klebsiella spp.; Streptococcus spp. including Streptococcus pyogenes (but excluding enterococci); Proteus mirabilis; Serratia spp.; Enterobacter spp.; Bacteroides spp. including B. fragilis; and anaerobic cocci, including Peptococcus spp. and Peptostreptococcus spp.
Bone and Joint Infections caused by Staphylococcus aureus (penicillinase and nonpenicillinaseproducing); Streptococcus spp. (excluding enterococci); Proteusmirabilis; Bacteroides spp.; and anaerobic cocci, including Peptococcus spp. and Peptostreptococcus spp.
Meningitis caused by Haemophilus influenzae. Cefizox has also been used successfully in the treatment of a limited number of pediatric and adult cases of meningitis caused by Streptococcus pneumoniae.
Cefizox has been effective in the treatment of seriously ill, compromised patients, including those who were debilitated, immunosuppressed, or neutropenic.
Infections caused by aerobic gramnegative and by mixtures of organisms resistant to other cephalosporins, aminoglycosides, or penicillins have responded to treatment with Cefizox.
Because of the serious nature of some urinary tract infections due to P. aeruginosa and because many strains of Pseudomonas species are only moderately susceptible to Cefizox, higher dosage is recommended. Other therapy should be instituted if the response is not prompt.
Susceptibility studies on specimens obtained prior to therapy should be used to determine the response of causative organisms to Cefizox. Therapy with Cefizox may be initiated pending results of the studies; however, treatment should be adjusted according to study findings. In serious infections, Cefizox has been used concomitantly with aminoglycosides (see PRECAUTIONS). Before using Cefizox concomitantly with other antibiotics, the prescribing information for those agents should be reviewed for contraindications, warnings, precautions, and adverse reactions. Renal function should be carefully monitored.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefizox and other antibacterial drugs, Cefizox should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Contraindications
Cefizox (ceftizoxime injection) is contraindicated in patients who have known allergy to the drug.
Warnings
BEFORE THERAPY WITH CEFIZOX IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEFIZOX, OTHER CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. IF THIS PRODUCT IS TO BE GIVEN TO PENICILLINSENSITIVE PATIENTS, CAUTION SHOULD BE EXERCISED BECAUSE CROSS HYPERSENSITIVITY AMONG BETALACTAM ANTIBIOTICS HAS BEEN CLEARLY DOCUMENTED AND MAY OCCUR IN UP TO 10% OF PATIENTS WITH A HISTORY OF PENICILLIN ALLERGY. IF AN ALLERGIC REACTION TO CEFIZOX OCCURS, DISCONTINUE THE DRUG. SERIOUS ACUTE HYPERSENSITIVITY REACTIONS MAY REQUIRE TREATMENT WITH EPINEPHRINE AND OTHER EMERGENCY MEASURES, INCLUDING OXYGEN, IV FLUIDS, IV ANTIHISTAMINES, CORTICOSTEROIDS, PRESSOR AMINES, AND AIRWAY MANAGEMENT, AS CLINICALLY INDICATED.
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including ceftizoxime, and may range in severity from mild to life threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that a toxin produced by Clostridium difficile is a primary cause of “antibioticassociated” colitis.
After the diagnosis of pseudomembranous colitis has been established, appropriate therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation, and treatment with an antibacterial drug clinically effective against Clostridium difficile colitis.
Precautions
General
As with all broadspectrum antibiotics, Cefizox (ceftizoxime injection) should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Although Cefizox has not been shown to produce an alteration in renal function, renal status should be evaluated, especially in seriously ill patients receiving maximum dose therapy. As with any antibiotic, prolonged use may result in overgrowth of nonsusceptible organisms. Careful observation is essential; appropriate measures should be taken if superinfection occurs.
Cephalosporins may be associated with a fall in prothrombin activity. Those at risk include patients with renal or hepatic impairment, or poor nutritional state, as well as patients receiving a protracted course of antimicrobial therapy, and patients previously stabilized on anticoagulant therapy. Prothrombin time should be monitored in patients at risk and exogenous vitamin K administered as indicated.
Prescribing Cefizox in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Drug Interactions
Although the occurrence has not been reported with Cefizox, nephrotoxicity has been reported following concomitant administration of other cephalosporins and aminoglycosides.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Longterm studies in animals to evaluate the carcinogenic potential of ceftizoxime have not been conducted.
In an in vitro bacterial cell assay (i.e., Ames test), there was no evidence of mutagenicity at ceftizoxime concentrations of 0.0010.5 mcg/plate. Ceftizoxime did not produce increases in micronuclei in the in vivo mouse micronucleus test when given to animals at doses up to 7500 mg/kg, approximately six times greater than the maximum human daily dose on an mg/M2 basis.
Ceftizoxime had no effect on fertility when administered subcutaneously to rats at daily doses of up to 1000 mg/kg/day, approximately two times the maximum human daily dose on an mg/M2 basis. Ceftizoxime produced no histological changes in the sexual organs of male and female dogs when given intravenously for thirteen weeks at a dose of 1000 mg/kg/day, approximately five times greater than the maximum human daily dose on an mg/M2 basis.
Pregnancy: Teratogenic Effects: Pregnancy Category B.
Reproduction studies performed in rats and rabbits have revealed no evidence of impaired fertility or harm to the fetus due to Cefizox. There are, however, no adequate and wellcontrolled studies in pregnant women. Because animal reproduction studies are not always predictive of human effects, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
Cefizox is excreted in human milk in low concentrations. Caution should be exercised when Cefizox is administered to a nursing woman.
Pediatric Use
Safety and efficacy in pediatric patients from birth to six months of age have not been established. In pediatric patients six months of age and older, treatment with Cefizox has been associated with transient elevated levels of eosinophils, AST (SGOT), ALT (SGPT), and CPK (creatine phosphokinase).
The potential for the toxic effect in pediatric patients from chemicals that may leach from the singledose IV preparation in plastic has not been determined.
Geriatric Use
Clinical studies of ceftizoxime did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Information for Patients
Patients should be counseled that antibacterial drugs including Cefizox should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Cefizox is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Cefizox or other antibacterial drugs in the future.
Adverse Reactions/Side Effects
Cefizox® (ceftizoxime injection) is generally well tolerated. The most frequent adverse reactions (greater than 1% but less than 5%) are:
Hypersensitivity--Rash, pruritus, fever.
Hepatic--Transient elevation in AST (SGOT), ALT (SGPT), and alkaline phosphatase.
Hematologic--Transient eosinophilia, thrombocytosis. Some individuals have developed a positive Coombs test.
Local--Injection site--Burning, cellulitis, phlebitis with IV administration, pain, induration, tenderness, paresthesia.
The less frequent adverse reactions (less than 1%) are:
Hypersensitivity--Numbness and anaphylaxis have been reported rarely.
Hepatic--Elevation of bilirubin has been reported rarely.
Renal--Transient elevations of BUN and creatinine have been occasionally observed with Cefizox.
Hematologic--Anemia, including hemolytic anemia with occasional fatal outcome, leukopenia, neutropenia, and thrombocytopenia have been reported rarely.
Urogenital--Vaginitis has occurred rarely.
Gastrointestinal--Diarrhea; nausea and vomiting have been reported occasionally.
Symptoms of pseudomembranous colitis can appear during or after antibiotic treatment (see WARNINGS).
In addition to the adverse reactions listed above which have been observed in patients treated with ceftizoxime, the following adverse reactions and altered laboratory tests have been reported for cephalosporinclass antibiotics:
StevensJohnson syndrome, erythema multiforme, toxic epidermal necrolysis, serumsickness like reaction, toxic nephropathy, aplastic anemia, hemorrhage, prolonged prothrombin time, elevated LDH, pancytopenia, and agranulocytosis.
Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment, when the dosage was not reduced. (See DOSAGE AND ADMINISTRATION.) If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
Related/similar drugs
Cefizox Injection Dosage and Administration
The usual adult dosage is 1 or 2 grams of Cefizox (ceftizoxime injection) in Galaxy® plastic container every 8 to 12 hours. Proper dosage and route of administration should be determined by the condition of the patient, severity of the infection, and susceptibility of the causative organisms.
|
Type of Infection |
Daily Dose (Grams) |
Frequency and Route |
|
Uncomplicated Urinary Tract | 1 | 500 mg q12h IV |
| Other Sites | 2-3 | 1 gram q8-12h IV |
|
Severe or Refractory | 3-6 |
1 gram q8h IV 2 grams q8-12h IV |
| PID * | 6 | 2 grams q8h IV |
| Life-Threatening † | 9-12 | 3-4 grams q8h IV |
_______________________________________________________________
Because of the serious nature of urinary tract infections due to P. aeruginosa and because many strains of Pseudomonas species are only moderately susceptible to Cefizox, higher dosage is recommended. Other therapy should be instituted if the response is not prompt.
A single, 1 gram IM dose is the usual dose for treatment of uncomplicated gonorrhea.
The IV route may be preferable for patients with bacterial septicemia, localized parenchymal abscesses (such as intraabdominal abscess), peritonitis, or other severe or lifethreatening infections.
In those with normal renal function, the IV dosage for such infections is 2 to 12 grams of Cefizox (ceftizoxime injection) daily. In conditions such as bacterial septicemia, 6 to 12 grams/day may be given initially by the IV route for several days, and the dosage may then be gradually reduced according to clinical response and laboratory findings.
| Unit Dose | Frequency | |
|
Pediatric patients 6 months or older | 50 mg/kg | q6-8h |
Dosage may be increased to a total daily dose of 200 mg/kg (not to exceed the maximum adult dose for serious infection).
Impaired Renal Function
Modification of Cefizox dosage is necessary in patients with impaired renal function. Following an initial loading dose of 500 mg-1 gram IV, the maintenance dosing schedule shown below should be followed. Further dosing should be determined by therapeutic monitoring, severity of the infection, and susceptibility of the causative organisms.
When only the serum creatinine level is available, creatinine clearance may be calculated from the following formula. The serum creatinine level should represent current renal function at the steady state.
Males
Clcr =Weight (kg) x (140 age)
72 x serum creatinine
(mg/100 mL)
Females are 0.85 of the calculated clearance values for males.
In patients undergoing hemodialysis, no additional supplemental dosing is required following hemodialysis; however, dosing should be timed so that the patient receives the dose (according to the table below) at the end of the dialysis.
|
Creatinine Clearance mL/min |
Renal Function |
Less Severe Infections |
Life-Threatening Infections |
| 79-50 |
Mild Impairment | 500 mg q8h |
0.75-1.5 grams q8h |
| 49-5 |
Moderate to severe impairment |
250-500 mg q12h |
0.5-1 gram q12h |
| 4-0 |
Dialysis Patients |
500 mg q48h or 250 mg q24h |
0.5-1 gram q48h or 0.5 gram q24h |
DIRECTIONS FOR USE OF CEFIZOX®(ceftizoxime injection) IN Galaxy® PLASTIC CONTAINER (PL 2040)
Cefizox® (ceftizoxime injection) in Galaxy® plastic container is to be administered either as a continuous or intermittent infusion.
Thawing of Plastic Container
Thaw frozen container at room temperature (25°C/77°F) or under refrigeration (5°C/41°F). (DO NOT FORCE THAW BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION.)
Check for minute leaks by squeezing container firmly. If leaks are detected, discard solution as sterility may be impaired. The container should be visually inspected. Components of the solutions may precipitate in the frozen state and will dissolve upon reaching room temperature with little or no agitation. Potency is not affected. Agitate after solution has reached room temperature. If after visual inspection the solution remains cloudy or if insoluble precipitate is noted or if any seals or outlet ports are not intact, the container should be discarded.
DO NOT ADD SUPPLEMENTARY MEDICATION.
The thawed solution in Galaxy®plastic container (PL 2040) remains chemically stable for 28 days under refrigeration (5°C/41°F) or for 48 hours at room temperature (25°C/77°F).
DO NOT REFREEZE THAWED ANTIBIOTICS.
Preparation for Intravenous Administration (Use aseptic technique)
- Suspend container from eyelet support.
- Remove protector from outlet port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
CAUTION: Do not use plastic containers in series connections. Such use could result in an embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is complete.
How is Cefizox Injection supplied
Cefizox® (ceftizoxime injection) is supplied as a frozen, iso-osmotic, sterile, nonpyrogenic solution in 50 mL single dose Galaxy® plastic containers (PL 2040) as follows:
NDC 0469-7220-01 Product No. 722001
1 g ceftizoxime/50 mL container
NDC 0469-7221-02 Product No. 722102
2 g ceftizoxime/50 mL container
Store at or below -20°C/-4°F.
Rx only
See DIRECTIONS FOR USE OF CEFIZOX®(ceftizoxime injection) IN Galaxy® PLASTIC CONTAINER (PL 2040)
Galaxy® is registered trademark of Baxter International Inc.
Ceftizoxime sodium is a product of Japan.
Marketed by:
Astellas Pharma US, Inc.
Deerfield, IL 60015
Manufactured by:
Baxter Healthcare Corporation
Deerfield, IL 60015.
References
- National Committee for Clinical Laboratory Standards. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically – Fifth Edition. Approved Standard NCCLS Document M7-A5, Vol. 20, No. 2, NCCLS, Wayne, PA, January 2000.
- National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Disk Susceptibility Tests – Seventh Edition. Approved Standard NCCLS Document M2-A7, Vol. 20, No. 1, NCCLS, Wayne, PA, January 2000.
- National Committee for Clinical Laboratory Standards. Methods for Antimicrobial Susceptibility Testing of Anaerobic Bacteria – Fourth Edition. Approved Standard NCCLS Document M11-A4, Vol. 17, No. 22, NCCLS, Wayne, PA, December 1997.
- National Committee for Clinical Laboratory Standards. MIC Testing Supplemental Tables. NCCLS Document M100-S10 (M7), NCCLS, Wayne, PA, January 2000.
| CEFIZOX
ceftizoxime sodium solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| CEFIZOX
ceftizoxime sodium solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Astellas Pharma US, Inc. |
More about Cefizox (ceftizoxime)
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: third generation cephalosporins
- Breastfeeding
