DURA-GEST 51479 005 Pill: gray & white, capsule/oblong
Generic Name: guaifenesin/phenylephrine/phenylpropanolamine
The pill with imprint DURA-GEST 51479 005 (Gray & White, Capsule/Oblong, 0mm) has been identified as Dura-Gest 200 mg / 5 mg / 45 mg and is used for Cold Symptoms. It belongs to the drug class upper respiratory combinations and is not a controlled substance.
Images for DURA-GEST 51479 005
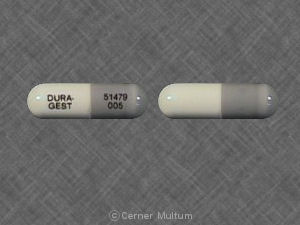
Dura-Gest
- Generic Name
- guaifenesin/phenylephrine/phenylpropanolamine
- Imprint
- DURA-GEST 51479 005
- Strength
- 200 mg / 5 mg / 45 mg
- Color
- Gray & White
- Shape
- Capsule/Oblong
- Availability
- Prescription only
- Drug Class
- Upper respiratory combinations
- Pregnancy Category
- C - Risk cannot be ruled out
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Elan Pharmaceutical/Athena Neurosciences Inc
- National Drug Code (NDC)
- 51479-0005 (Discontinued)
See also:
More about Dura-Gest (guaifenesin / phenylephrine / phenylpropanolamine)
- Check interactions
- Compare alternatives
- Drug images
- Side effects
- Drug class: upper respiratory combinations
Patient resources
Other brands
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
