23 57 V Pill: white, capsule/oblong, 20mm
The pill with imprint 23 57 V (White, Capsule/Oblong, 20mm) has been identified as Acetaminophen, Butalbital and Caffeine 500 mg / 50 mg / 40 mg and is used for Headache. It belongs to the drug class analgesic combinations and is not a controlled substance.
Images for 23 57 V
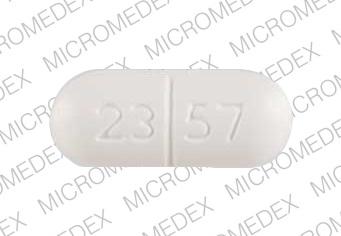
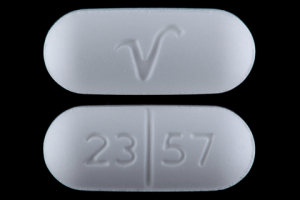
Acetaminophen, Butalbital and Caffeine
- Imprint
- 23 57 V
- Strength
- 500 mg / 50 mg / 40 mg
- Color
- White
- Size
- 20.00 mm
- Shape
- Capsule/Oblong
- Availability
- Prescription only
- Drug Class
- Analgesic combinations
- Pregnancy Category
- C - Risk cannot be ruled out
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Qualitest Pharmaceuticals Inc.
- National Drug Code (NDC)
- 00603-2545 (Discontinued)
- Inactive Ingredients
-
silicon dioxide,
croscarmellose sodium,
magnesium stearate,
microcrystalline cellulose,
stearic acid,
crospovidone,
povidone,
corn starch
Note: Inactive ingredients may vary.
Related images for "23 57 V"
More about acetaminophen / butalbital / caffeine
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (265)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Drug class: analgesic combinations
- En español
Patient resources
- Acetaminophen, butalbital, and caffeine drug information
- Butalbital, acetaminophen, and caffeine (Advanced Reading)
Other brands
Fioricet, Esgic, Esgic-Plus, Ezol, ... +10 more
Professional resources
- Butalbital, Acetaminophen and Caffeine prescribing information
- Butalbital, Acetaminophen and Caffeine Capsule (FDA)
- Butalbital,Acetaminophen and Caffeine Solution (FDA)
Other brands
Fioricet, Esgic, Esgic-Plus, Phrenilin Forte, ... +4 more
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.

