Witness FeLV-FIV
This treatment applies to the following species:FELINE LEUKEMIA VIRUS ANTIGEN - FELINE IMMUNODEFICIENCY VIRUS ANTIBODY TEST KIT
I. General Information
WITNESS™ FeLV-FIV test is indicated for use when history and/or clinical signs may suggest an infection by feline retroviruses. It will also be particularly recommended prior to an FeLV vaccination, especially in cats belonging to an at risk population.
Ii. Test Principles
The WITNESS™ FeLV-FIV is a simple test based on Rapid Immuno Migration (RIM™) technology. FeLV antigen is detected using antibodies directed against the circulating p27 capside protein.
The detection of FIV is based on the presence of antibodies against the transmembrane region of the virus, using a synthetic peptide from the gp40 protein.
In both cases, sensitized colloidal gold particles will form a complex with either the p27 antigen (FeLV) or antibodies (FIV) present in the sample.
The formed complexes migrate along the strips. The complexes are then captured on a sensitized reaction line where their accumulation causes the formation of a clearly visible pink/red band.
Control bands located at the end of the reading windows (3) ensure that the test was performed correctly.
III. SAMPLE COLLECTION
● The test can be performed on unclotted whole blood anticoagulated with EDTA or heparin, serum or plasma.
● Samples should always be collected with a sterile needle and syringe.
● Haemolysis does not significantly interfere with the test, but strongly haemolysed samples may partly obscure a weak positive band.
IV. SAMPLE STORAGE
Anticoagulated whole blood samples should preferably be tested immediately after collection but not longer than 4 hours after collection, if stored at room temperature.
If testing is further delayed, samples should be kept refrigerated (+2°C and 8°C) for up to 4 days.
For prolonged storage, samples (serum and plasma only) should be kept frozen (-20°C).
V. KIT CONTENTS
● 10 or 25 Pouches, each containing 1 test device and desiccant.
● 10 or 25 Pipettes.
● 1 or 2 Saline Buffer dropper bottles (5.4 mL).
● Instructions for use.
VI. PRECAUTIONS
● Do not use this kit or any of its components after expiration date.
● Kit should be stored at +2 °C - 25 °C (35 °F - 77 °F). Kit should not be frozen.
● Use the test immediately after opening the sealed pouch (within 10 minutes).
● Avoid touching or damaging the membrane in the sample wells or the results windows.
● The WITNESS™ device should be placed on a flat, horizontal surface while performing the test.
● Use a separate pipette for each sample.
● Hold pipette and buffer bottle vertically when dispensing sample and buffer respectively.
● Handle all reagents and samples as biohazardous material.
● Saline Buffer contains sodium azide as a preservative.
● For veterinary use only.
VII. TEST PROCEDURE
Important: Allow samples and buffer drops to fall onto the membrane in each sample window. Do not touch pipette tip, sample or buffer drops, or buffer bottle tip directly to the membrane.
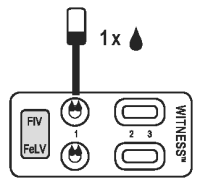
1. SAMPLE APPLICATION
● Tear open a pouch provided and place the test device on a flat horizontal surface.
● Holding the provided pipette vertically, add one drop of sample to each test well (1).
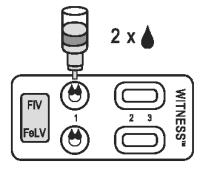
2. BUFFER DISPENSING
● Check that the sample has fully absorbed into the membrane.
● Remove the cap from the buffer bottle, hold it vertically and add two drops of buffer to each sample well (1).
● Leave the test device flat during migration of sample/reagent complex through the reading windows.
3. READING TEST
After 10 minutes, observe the presence or absence of pink/red bands in the reading windows (2) and (3).
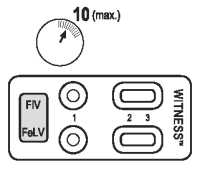
Note:
● It is possible to read the test before 10 minutes if two pink/red bands are clearly visible in the results windows (2) and (3).
● The presence of only one band in reading window (3), prior to the end of the development time (10 minutes), does not mean that the test is complete, as a test band may appear more slowly than the control band.
VIII. RESULTS
Valid Results
● Test is valid if a pink/red band is present in each reading window (3).
Interpretation of Results
● Positive. One band in reading window (2), with one band in window (3): sample is positive for FeLV antigen and/or FIV antibodies.
● Negative. No band in reading window (2), with one band in window (3): sample is negative for FeLV antigen and/or FIV antibodies.
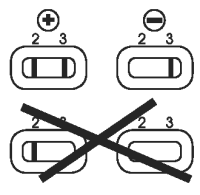
Note:
● No band in control window (3): invalid test.
● A test result should always be interpreted in the context of all available clinical information and history for the cat being tested.
SYMBOL DESCRIPTIONS

Zoetis Inc., Kalamazoo, MI 49007, USA
VLN/PCN 190/502A.01
EC REP
ZOETIS FRANCE, 23 Rue Pierre Gilles de Gennes, 69007, Lyon, FRANCE
For product information call:
US VMIPS 1-888-963-8471
Canada 1-800-461-0917
40023082
CPN: 3690618.0
333 PORTAGE STREET, KALAMAZOO, MI, 49007
| Telephone: | 269-359-4414 | |
| Customer Service: | 888-963-8471 | |
| Website: | www.zoetis.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
