The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
VetScan Feline Leukemia Virus Antigen-Feline Immunodeficiency Virus Antibody Test Kit (FeLV/FIV Combo)
This treatment applies to the following species:Feline Leukemia Virus Antigen-Feline Immunodeficiency Virus Antibody Test Kit (FeLV-FIV Combo) for the qualitative detection of feline leukemia virus Antigen and feline immunodeficiency virus antibodies in feline whole blood, serum, or plasma
Intended Use
The VetScan Feline Leukemia Virus Antigen-Feline Immunodeficiency Virus Antibody (FeLV-FIV Combo) Rapid Test is a visual rapid test for the qualitative detection of Feline Leukemia Virus antigen and Feline Immunodeficiency Virus antibody in feline whole blood, serum, or plasma. This test is for veterinary use only.
FIV and FeLV are two of the most commonly diagnosed causes of disease and death in domestic cats (2,3). Feline leukemia virus (FeLV) is a highly contagious retrovirus that can severely compromise a cat’s immune system and predisposes cats to various diseases (1,2).
Transmission occurs from cat to cat via body fluids during casual contact or fighting. Identification of infected cats helps prevent the spread of FeLV. Rapid diagnosis of FeLV infection is generally performed by detecting viral antigen p27 from the core protein, which is found in high level in viremic cats. (1,2) Feline immunodeficiency virus (FIV) is a lentivirus with world-wide distribution (4). Similarly to FeLV, FIV is transmitted mainly by saliva. FIV infection has a long latent phase and therefore infected cats remain free of clinical signs for several years. Most clinical signs and symptoms are not caused by FIV, but are the consequences of the immunodeficiency that FIV induces (5). Typical manifestations are lymphadenopathy, weight loss, renal failure and chronic rhinitis (5).
The VetScan Feline Leukemia Virus Antigen-Feline Immunodeficiency Virus Antibody (FeLV-FIV Combo) Rapid Test is based on the use of FeLV-specific antibodies and an FIV antigen in its respective immunochromatographic sandwich assay. For the FeLV strip, antibody-coated colloidal gold particles bind to FeLV antigen in the sample. The bound antigen flows through the strip and is then captured by antibodies on the test strip. For the FIV strip, antigen-coated colloidal gold particles bind to FIV antibody in the sample. The bound antibodies flow through the strip and are then captured by antigen on the test strip. The accumulation of the captured gold particle/antigen or antibody complex causes a color to become visible in the Test (T) area. To serve as a procedural control, a colored line in the Control line (C) area will always appear regardless if the sample is positive or negative.
INSTRUCTIONS FOR USE
● Samples must be at room temperature (15-27 °C/59-80 °F), before running the assay - DO NOT HEAT.
● Previously frozen or older samples must be centrifuged before use.
● Serum or Plasma, either fresh, previously frozen or stored at 2-7 °C (36-45 °F), may be used in this test. Serum or plasma may be stored for up to 7 days at 2-7 °C. For longer storage, sample should be frozen at -20 °C or colder (-4 °F).
● Whole Blood may be used. Whole blood must be anticoagulated (e.g. EDTA, heparin) and may be used either fresh or after refrigeration at 2-7 °C (36-45 °F) for up to 5 days.
● Hemolyzed samples will not affect the results.
● EDTA or heparin in plasma will not affect the results.
PRECAUTIONS AND WARNINGS
● Important: Do not remove Test Device from the pouch until ready for use.
● For veterinary use only.
● Do not use components after expiration date.
● The Test Device must be used as soon as possible after removing from pouch.
● The Test Device should be in a horizontal position on a flat surface while the test is performed.
● Use a separate Transfer Pipette for each test.
● The FeLV Chase Buffer is not interchangeable from serial (lot) to serial (lot).
● The FIV Chase Buffer is not interchangeable from serial (lot) to serial (lot).
● Contains sodium azide as a preservative.
● Contains Proclin™ 300 and sodium azide as preservatives.
STORAGE
● Test Devices and Chase Buffers must be stored at room temperature 15-27 °C (59-80 °F).
● Test Devices and Chase Buffers are stable until the expiration date when stored at 15-27 °C (59-80 °F).
KIT COMPONENTS
1. Test Devices
2. FeLV Chase Buffer Bottle
3. FIV Chase Buffer Bottle
4. Transfer Pipettes
5. Instructions for Use
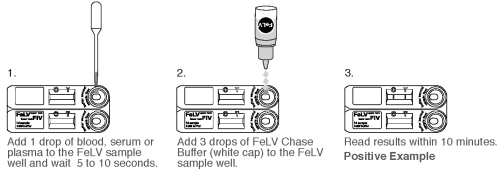
TEST PROCEDURE FOR FeLV TEST
1. Remove the Test Device from the protective pouch and place on a flat surface. Label the Test Device with the subject I.D. or control identification.
2. Gently mix the sample by inverting.
3. Using the Transfer Pipette provided, transfer 1 drop of sample (whole blood, serum, or plasma) in to the top FeLV sample well. Wait for the sample to be absorbed (5-10 seconds).
4. Holding the FeLV Chase Buffer Bottle (white cap) vertically, promptly add 3 drops of the FeLV Chase Buffer to the FeLV sample well.
5. Read the results within 10 minutes. Do not read results after 10 minutes. Colored lines which appear after 10 minutes are not diagnostic and should be ignored.
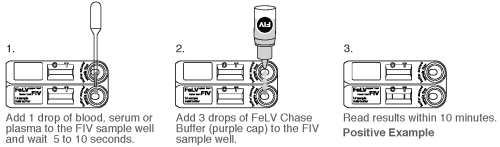
TEST PROCEDURE FOR FIV TEST
1. Gently mix the sample by inverting.
2. Using the Transfer Pipette provided, dispense 1 drop of sample (whole blood, serum, or plasma) into the bottom sample well. Wait for the sample to be absorbed (5-10 seconds).
3. Holding the FIV Chase Buffer Bottle (purple cap) vertically, promptly add 3 drops of the FIV Chase Buffer to the bottom FIV sample well.
4. Read the results within 10 minutes. High positives may appear as soon as 2 minutes, and low positive results may take up to 10 minutes to appear. Do not read results after 10 minutes. Colored lines which appear after 10 minutes are not diagnostic and should be ignored.
INTERPRETATION OF TEST RESULTS
Positive Results
The test is positive if two colored lines appear. One colored line will appear at the Test line (T) area and other in the Control line (C) area. Any intensity of the Test line (T) area should be considered positive. Colored lines may be lighter or darker than each other.
Negative Results
The test is negative if only one line appears at the Control line (C) area.
Invalid Results
The test is invalid if no colored line appears at the Control line (C) area even if a colored line appears in the Test line (T) area. If a colored line still does not appear in the C area, the test is invalid and should be repeated.
FeLV Test Procedure
FIV Test Procedure
REFERENCES
1.) Willett and Hosie. (2013). Feline leukaemia virus: Half a century since its discovery. The Veterinary Journal 195 (2013) 16-23.
2.) Courchamp F. and Pontier D. (1994). Immunodeficiency virus: an epidemiologic review. C R Acad Sci Ser III-Vie 317: 1123-1134.
3.) Hartmann. (2012) Clinical Aspects of Feline Retroviruses: A Review. Viruses 2012, 4, 2684-2710
4.) Journal of Feline Medicine and Surgery 2008 10: 300 Julie Levy, Cynda Crawford, Katrin Hartmann, Regina Hofmann-Lehmann, Susan Little, Eliza Sundahl and Vicki Thayer
5.) European Advisory Board on Cat Diseases (ABCD) Fact Sheet. April, 2009. http://www.abcd-vets.org/fact_sheets/English/EN_FIV_Feline_immunodeficiency.pdf
ABAXIS CUSTOMER CARE
In Americas: +1 800 822 2947. Outside Americas: +49 6155 780 210
Manufacturer
SA Scientific, 4919 Golden Quail, San Antonio, TX 78240 USA, U.S. Vet. License No. 373
Distributed by: Abaxis, Inc. 3240 Whipple Road, Union City, CA 94587 USA
+1 800 822 2947
www.abaxis.com
ABAXIS Europe GmbH, Bunsenstr. 9-11, 64347 Griesheim, Germany
+49 6155 780 210
For patent information, see www.abaxis.com/about_us/patents.
Learn more about all of our products and services at www.abaxis.com. Abaxis and VetScan are registered trademarks of Abaxis, Inc. © Abaxis 2017.

250-7002 Rev. D
Presentation: 10 and 25 test kits.
CPN: 1740006.2
Distributed by ZOETIS INC.
333 PORTAGE STREET, KALAMAZOO, MI, 49007
| Telephone: | 269-359-4414 | |
| Customer Service: | 888-963-8471 | |
| Website: | www.zoetis.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
