Rimadyl Injectable Solution (Canada)
This treatment applies to the following species: Company: Zoetis
Company: Zoetis
carprofen injectable solution
Veterinary Use Only
Non-steroidal anti-inflammatory
For subcutaneous use in dogs only.
DIN 02255693
Description
Rimadyl injectable solution is a sterile solution containing carprofen, a non-steroidal anti-inflammatory drug (NSAID) of the propionic acid class that includes ibuprofen, naproxen, and ketoprofen. The chemical name for carprofen, a substituted carbazole, is (±)-6-chloro-α-methylcarbazole-2-acetic acid. The empirical formula is C15H12NO2CI and molecular weight 273.72. The chemical structure of carprofen is:
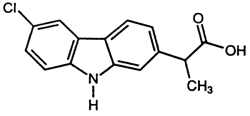
Each mL of Rimadyl injectable solution contains 50.0 mg of carprofen as the medicinal ingredient and 10.0 mg of benzyl alcohol as the preservative.
Clinical Pharmacology
Carprofen is a non-narcotic, non-steroidal anti-inflammatory agent with characteristic analgesic and antipyretic activity approximately equipotent to indomethacin in animal models.1The mechanism of action of carprofen, like that of other NSAIDs, is believed to be associated with the inhibition of cyclooxygenase activity. Two unique cyclooxygenases have been described in mammals.2 The constitutive cyclooxygenase, COX-1, synthesizes prostaglandins necessary for normal gastrointestinal and renal function. The inducible cyclooxygenase, COX-2, generates prostaglandins involved in inflammation. Inhibition of COX-1 is thought to be associated with gastrointestinal and renal toxicity while inhibition of COX-2 provides anti-inflammatory activity. The specificity of a particular NSAID for COX-2 versus COX-1 may vary from species to species.3 In an in vitro study using canine cell cultures, carprofen demonstrated selective inhibition of COX-2 versus COX-1.4 Clinical relevance of these data has not been shown. Carprofen has also been shown to inhibit the release of several prostaglandins in two inflammatory cell systems: rat polymorphonuclear leukocytes (PMN) and human rheumatoid synovial cells, indicating inhibition of acute (PMN system) and chronic (synovial cell system) inflammatory reactions.1 Several studies have demonstrated that carprofen has modulatory effects on both humoral and cellular immune responses.5-9 Data also indicate that carprofen inhibits the production of osteoclast-activating factor (OAF), PGE1, and PGE2 by its inhibitory effects on prostaglandin biosynthesis.1 Based upon comparison with data obtained from intravenous administration, carprofen is rapidly and nearly completely absorbed (more than 90% bioavailable) when administered orally.10 Peak blood plasma concentrations are achieved in 1-3 hours after oral administration of 1, 5, and 25 mg/kg to dogs. The mean terminal half-life of carprofen is approximately 8 hours after single oral doses varying from 1-35 mg/kg of body weight. After a 100 mg single intravenous bolus dose, the mean elimination half-life was approximately 11.7 hours in the dog. Rimadyl is more than 99% bound to plasma protein and exhibits a very small volume of distribution. Comparison of subcutaneous and oral administration of a single dose of 25 mg in beagle dogs demonstrated similar bioavailability with respect to total drug exposure (area under the curve).
Comparative pharmacokinetic studies in beagle dogs revealed that the subcutaneous administration of Rimadyl results in a slower rate of drug absorption (lower peak concentrations) compared to the oral administration of Rimadyl in a caplet formulation.
Carprofen is eliminated in the dog primarily by biotransformation in the liver followed by rapid excretion of the resulting metabolites (the ester glucuronide of carprofen and the ether glucuronides of 2 phenolic metabolites, 7-hydroxy carprofen and 8-hydroxy carprofen) in the feces (70% - 80%) and urine (10-20%). Some enterohepatic circulation of the drug is observed.
Rimadyl Injectable Solution Indications
Rimadyl injectable solution is indicated for the control of postoperative pain associated with soft tissue and orthopedic surgeries in dogs.
Dosage and Administration
The recommended dosage for subcutaneous administration to dogs is 4.4 mg carprofen per kg body weight (1.0 mL/11.4 kg) once daily. Administer approximately 2 hours prior to surgery and once daily thereafter, as needed, for a maximum of 3 consecutive days postoperatively. If necessary, treatment may be continued with orally administered Rimadyl chewable tablets at a dose of 4.4 mg/kg once daily, or 2.2 mg/kg twice daily.Owners should be advised when their dog has received a carprofen injection, and be informed of the potential for adverse reactions and clinical signs associated with NSAID intolerance. Always provide client information sheet with prescription. Owners should be advised of the importance of periodic follow up for all dogs during administration of any NSAID.
SAFETY: Laboratory studies in unanesthetized dogs and clinical field trials have demonstrated that Rimadyl is well tolerated in dogs after oral and subcutaneous administration.
In target animal safety studies, Rimadyl was administered orally to dogs at 1, 3, and 5 times the recommended dose for 42 consecutive days with no significant adverse reactions. Serum albumin for a single female dog receiving 5 times the recommended dose decreased to 2.1 g/dL after 2 weeks of treatment, returned to the pre-treatment value (2.6 g/dL) after 4 weeks of treatment, and was 2.3 g/dL at the final 6-week evaluation. Over the 6 week treatment period, black or bloody stools were observed in 1 dog (1 incident) treated with the recommended dose and in 1 dog (2 incidents) treated with 3 times the recommended dose. Redness of the colonic mucosa was observed in 1 male that received 3 times the recommended dose.
Two of 8 dogs receiving 10 times the recommended dose (22 mg/kg twice daily) orally for 14 days exhibited hypoalbuminemia. The mean albumin level in the dogs receiving this dose was lower (2.38 g/dL) than each of 2 placebo control groups (2.88 and 2.93 g/dL, respectively). Three incidents of black or bloody stool were observed in 1 dog. Five of 8 dogs exhibited reddened areas of duodenal mucosa on gross pathologic examination. Histologic examination of these areas revealed no evidence of ulceration, but did show minimal congestion of the lamina propria in 2 of the 5 dogs.
In separate safety studies lasting 13 and 52 weeks, respectively, dogs were administered up to 25.1 mg/kg/day (5.7 times the recommended total daily dose) of carprofen orally. In both studies, the drug was well tolerated clinically by all of the animals. No gross or histologic changes were seen in any of the treated animals. In both studies, dogs receiving the highest doses had average increases in serum L-alanine aminotransferase (ALT) of approximately 20 IU.
In the 52 week study, minor dermatologic changes occurred in dogs in each of the treatment groups but not in the control dogs. The changes were described as slight redness or rash and were diagnosed as non-specific dermatitis. The possibility exists that these mild lesions were treatment related, but no dose relationship was observed.
Clinical field studies were conducted with 549 dogs of different breeds at the recommended oral dose for 14 days. The drug was clinically well tolerated and the incidence of clinical adverse reactions for Rimadyl-treated animals was no higher than placebo-treated animals (placebo contained inactive ingredients found in Rimadyl). Mean post-treatment serum ALT values were 11 IU greater and 9 IU less than pre-treatment values for dogs receiving Rimadyl and placebo, respectively. Differences were not statistically significant. For animals receiving 4.4 mg/kg orally once daily, the mean post-treatment serum ALT values were 5 IU greater and 1 IU less than pre-treatment values for dogs receiving Rimadyl and placebo, respectively. Changes in clinical laboratory values (hematology and clinical chemistry) were not considered clinically significant nor reported as adverse reactions. The 2.2 mg/kg twice daily course of oral therapy was repeated as needed at 2 week intervals in 244 dogs, some for as long as 5 years.
An injection site toleration study demonstrated that Rimadyl injectable solution is clinically well tolerated.
Clinical studies on the use of Rimadyl injectable solution were conducted on 331 dogs undergoing orthopedic or soft tissue surgery. Dogs were administered 4.4 mg/kg of Rimadyl subcutaneously two hours prior to surgery and once daily thereafter, as needed, for 2 days (soft tissue surgery) or 3 days (orthopedic surgery). Rimadyl was well tolerated when used in conjunction with a variety of anesthetic-related drugs. The type and severity of abnormal health observations in Rimadyl- and placebo-treated animals were approximately equal and few in number (see Adverse Reactions). The most frequent health observation was vomiting and was observed at approximately the same frequency in Rimadyl- and placebo-treated animals. Changes in clinicopathologic indices of hematopoietic, renal, hepatic, and clotting function were not clinically significant. The mean post-treatment serum ALT values were 8.4 IU and 7.0 IU less than pre-treatment values for dogs receiving Rimadyl and placebo, respectively. The mean post-treatment AST values were 1.5 IU and 0.7 IU greater for dogs receiving Rimadyl and placebo, respectively.
Swelling and warmth were associated with the injection site after subcutaneous administration of Rimadyl injectable solution. These findings were not clinically significant. Long term use of the injectable has not been studied.
Contraindications
Rimadyl should not be used in dogs exhibiting previous hypersensitivity to carprofen.
CAUTIONS: As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal and renal toxicity. NSAIDs decrease prostaglandin production and inhibit the enzyme cyclo-oxygenase which leads to formation of prostaglandins from arachidonic acid.11-14 When NSAIDs inhibit prostaglandins that promote inflammation, they may also inhibit prostaglandins which maintain normal homeostatic function. These anti-prostaglandin side-effects may result in clinically significant disease in patients with underlying or pre-existing disease more often than in healthy patients. NSAID therapy could therefore reveal the presence of disease which has been previously undiagnosed due to the absence of clinical signs. Patients with underlying renal disease for example, may experience exacerbation or decompensation of their renal disease while on NSAID therapy.11-14
Carprofen is an NSAID and, as with others in that class, side effects may occur with its use. The most frequently reported effects have been gastrointestinal signs. Events involving suspected renal, hematologic, neurologic, dermatologic, and hepatic effects have also been reported. Patients at greatest risk for renal toxicity are those that are dehydrated, on concomitant diuretic therapy, or those with renal, cardiovascular, and/or hepatic dysfunction. Since many NSAIDs possess the potential to induce gastrointestinal ulceration, concomitant use of Rimadyl with other anti-inflammatory drugs, such as corticosteroids and NSAIDs, should be avoided or very closely monitored. Although the drug class has been associated with renal toxicity and gastrointestinal ulceration, Rimadyl treatment did not produce these effects in well-controlled safety studies of up to ten times the dose in dogs.
All dogs should undergo a thorough history and physical examination before initiation of NSAID therapy. Appropriate laboratory tests to establish hematological and serum biochemical baseline data prior to, and periodically during, administration of any NSAID should be considered in some patients. Dogs receiving Rimadyl should be observed for signs of drug intolerance, such as inappetence, vomiting, diarrhea, melena, polyuria/ polydipsia, anemia, jaundice, lethargy, ataxia, seizure, or behavioral changes. Stop administration of Rimadyl immediately if decreased appetite, vomiting, lethargy, diarrhea or other suspected adverse reactions occur, and seek the advice of a veterinarian (see ADVERSE REACTIONS).
Susceptibility to drug-associated adverse effects varies with the individual patient. Recognition of possible drug-related clinical signs followed by cessation of drug therapy, and by supportive therapy if appropriate, will improve patient recovery. The side effects of this drug class, in rare situations, may be serious, and if corrective action is not taken may result in hospitalization or even fatal outcomes.
The safe use of Rimadyl in animal less than 6 weeks of age, pregnant dogs, dogs used for breeding purposes or in lactating bitches has not been established. Studies to determine the activity of Rimadyl when administered concomitantly with other protein-bound drugs have not been conducted. Drug compatibility should be monitored closely in patients requiring additional therapy. Rimadyl is not recommended for use in dogs with bleeding disorders (e.g., Von Willebrand’s Disease), as safety has not been established in dogs with these disorders. It has been suggested that treatment with carprofen may reduce the level of inhalant anesthetics needed.15 It is suggested to use different sites for additional injections. If additional pain medication is warranted after administration of the total daily dose of Rimadyl, alternative analgesia should be considered. The use of another NSAID is not recommended.
Do not use in cats.
Information for Dog Owners:
Rimadyl, like other drugs of its class, is not free from adverse reactions. Owners should be advised of the potential for adverse reactions and be informed of the clinical signs associated with drug intolerance. Adverse reactions may include decreased appetite, vomiting, diarrhea, dark or tarry stools, increased water consumption, increased urination, pale gums due to anemia, yellowing of gums, skin or white of the eye due to jaundice, lethargy, incoordination, seizure, or behavioral changes. Serious adverse reactions associated with this drug class can occur without warning and in rare situations result in death (see ADVERSE REACTIONS). Discontinue Rimadyl therapy and contact your veterinarian immediately if signs of intolerance are observed. The vast majority of patients with drug related adverse reactions have recovered when the signs are recognized, the drug is withdrawn, and veterinary care, if appropriate, is initiated. Owners should be advised of the importance of periodic follow up for all dogs during administration of any NSAID.
Warnings
Keep out of reach of children.Adverse Reactions
During investigational studies for a caplet formulation with twice daily administration of 2.2 mg/kg, no clinically significant adverse reactions were reported. Some clinical signs were observed during field studies (n=297) which were similar for carprofen caplet- and placebo-treated dogs. Incidences of the following were observed in both groups: vomiting (4%), diarrhea (4%), changes in appetite (3%), lethargy (1.4%), behavioral changes (1%), and constipation (0.3%).There were no serious adverse events reported during clinical field studies with once daily oral administration of 4.4 mg/kg. The following categories of abnormal health observations were reported. The product vehicle served as control.
Percentage of Dogs with Abnormal Health Observations Reported in Clinical Field Study (4.4 mg/kg once daily)
|
Observation |
Rimadyl (n=129) |
Placebo (n=132) |
|
Inappetance |
1.6 |
1.5 |
|
Vomiting |
3.1 |
3.8 |
|
Diarrhea/Soft stool |
3.1 |
4.5 |
|
Behavior change |
0.8 |
0.8 |
|
Dermatitis |
0.8 |
0.8 |
|
PU/PD |
0.8 |
- |
|
SAP increase |
7.8 |
8.3 |
|
ALT increase |
5.4 |
4.5 |
|
AST increase |
2.3 |
0.8 |
|
BUN increase |
3.1 |
1.5 |
|
Bilirubinuria |
16.3 |
12.1 |
|
Ketonuria |
14.7 |
9.1 |
Clinical pathology parameters listed represent reports of increases from pre-treatment values; the use of clinical judgment is necessary to determine clinical relevance (refers also to table below).
During investigational studies for the chewable tablet formulation, gastrointestinal signs were observed in some dogs. These signs included vomiting and soft stools.
There were no serious adverse events reported during clinical field studies for the injectable formulation. The following categories of abnormal health observations were reported. The product vehicle served as control.
Percentage of Dogs with Abnormal Health Observations Reported in Clinical Field Studies with the Injectable Formulation
|
Observation* |
Rimadyl (n=168) |
Placebo (n=163) |
|
Vomiting |
10.1 |
9.2 |
|
Diarrhea/soft stool |
2.4 |
3.7 |
|
Dermatitis |
0.6 |
1.2 |
|
Dysrhythmia |
0.6 |
0.6 |
|
Swelling |
0 |
1.2 |
|
Dehiscence |
1.2 |
0 |
|
WBC increase |
13.7 |
6.7 |
* A single dog may have experienced more than one occurrence of an event.
Post-approval Experience
Although all adverse reactions are not reported, the following adverse reactions are based on voluntary post-approval adverse drug experience reporting to the Center for Veterinary Medicine in the United States (for both oral and injectable formulations). The categories of adverse reactions are listed in decreasing order of frequency by body system.
Gastrointestinal: Vomiting, diarrhea, constipation, inappetance, melena, hematemesis, gastrointestinal ulceration, gastrointestinal bleeding, pancreatitis.
Hepatic: Inappetence, vomiting, jaundice, acute hepatic toxicity, hepatic enzyme elevation, abnormal liver function test(s), hyperbilirubinemia, bilirubinuria, hypoalbuminemia. Approximately one-fourth of hepatic reports were in Labrador Retrievers.
Neurologic: Ataxia, paresis, paralysis, seizures, vestibular signs, disorientation.
Urinary: Hematuria, polyuria, polydipsia, urinary incontinence, urinary tract infection, azotemia, acute renal failure, tubular abnormalities including acute tubular necrosis, renal tubular acidosis, glucosuria.
Behavioral: Sedation, lethargy, hyperactivity, restlessness, aggressiveness.
Hematologic: Immune-mediated hemolytic anemia, immune-mediated thrombocytopenia, blood loss anemia, epistaxis.
Dermatologic: Pruritus, increased shedding, alopecia, pyotraumatic moist dermatitis (hot spots), necrotizing panniculitis/vasculitis, ventral ecchymosis.
Immunologic or hypersensitivity: Facial swelling, hives, erythema.
In rare situations, death has been associated with some of the adverse reactions listed above.
Storage
Store under refrigeration between 2 and 8°C. Once broached, product may be stored at temperatures up to 25°C for 28 days.PRESENTATION: Rimadyl injectable solution is supplied in 20 mL amber glass sterile multi-dose vials.
References
1. Baruth H, et al: In Anti-Inflammatory and Anti-Rheumatic Drugs, Vol. II, Newer Anti-Inflammatory Drugs, Rainsford KD, ed. CRC Press, Boca Raton, pp. 33-47, 1986.
2. Vane JR, Botting RM: Mechanism of action of anti-inflammatory drugs. Scand J Rheumatol 25:102, pp. 9-21.
3. Grossman CJ, Wiseman J, Lucas FS, et al: Inhibition of constitutive and inducible cyclooxygenase activity in human platelets and mononuclear cells by NSAIDs and COX-2 inhibitors. Inflammation Research 44:253- 257, 1995.
4. Ricketts AP, Lundy KM, Seibel SB: Evaluation of selective inhibition of canine cyclooxygenase 1 and 2 by carprofen and other nonsteroidal anti-inflammatory drugs. AM J Vet Res 59:11, pp. 1441-1446, November 1998.
5. Ceuppens JL, et al: Non-steroidal anti-inflammatory agents inhibit the synthesis of IgM rheumatoid factor in vitro. Lancet 1:528, 1982.
6. Ceuppens JL, et al: Endogenous prostaglandin E2 enhances polyclonal immunoglobulin production by ionically inhibiting T suppressor cell activity. Cell Immunol 70:41, 1982.
7. Schleimer RP, et al: The effects of prostaglandin synthesis inhibition on the immune response. Immunopharmacology 3:205, 1981.
8. Leung KH, et al: Modulation of the development of cell mediated immunity: possible roles of the products of cyclo-oxygenase and lipoxygenase pathways of arachidonic acid metabolism. Int J Immunopharmacology 4:195, 1982.
9. Veit BC: Immunoregulatory activity of culture-induced suppressor macrophages. Cell Immunol 72:14, 1982.
10. Schmitt M, et al: Biopharmaceutical evaluation of carprofen following single intravenous, oral, and rectal doses in dogs. Biopharm Drug Dispos 11(7): 585-94, 1990.
11. Kore AM: Toxicology of nonsteroidal anti-inflammatory drugs. Veterinary Clinics of North America, Small Animal Practice 20, March 1990.
12. Binns SH: Pathogenesis and pathophysiology of isochemic injury in cases of acute renal failure. Compend for Cont Ed 16:1, January 1994.
13. Boothe DM: Prostaglandins: Physiology and clinical implications. Compend for Cont Ed 6:11, November 1984.
14. Rubin SI: Nonsteroidal anti-inflammatory drugs, prostaglandins, and the kidney. JAVMA 188:9, May 1986.
15. Ko CH, Lange DN, Mandsager RE, et al: Effects of butorphanol and carprofen on the minimal alveola concentration of isoflurane in dogs. JAVMA 217:1025-1028, 2000.
Zoetis® and Rimadyl are registered trademarks of Zoetis or its licensors.
Zoetis Canada Inc., Kirkland QC H9H 4M7
10023017-11-1
40045328
August 2023
CPN: 1198315.6
16,740 TRANS-CANADA HIGHWAY, KIRKLAND, QC, H9H 4M7
| Order Desk: | 800-663-8888 | |
| Technical Services Canada: | 800-461-0917 | |
| Technical Services USA: | 800-366-5288 | |
| Website: | www.zoetis.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
