Revertor (Canada)
This treatment applies to the following species: Company: Modern Veterinary Therapeutics
Company: Modern Veterinary Therapeutics
(Atipamezole hydrochloride injection - 5 mg/mL)
DIN 02337207
Injectable medetomidine reversing agent for use in dogs only - Sterile solution
For veterinary use only.
Active ingredient - Each mL contains:
|
Atipamezole hydrochloride |
5 mg |
Non-medicinal ingredients - Each mL contains:
|
Methylparahydroxybenzoate (preservative) |
1 mg |
|
Sodium chloride |
8.5 mg |
|
Water for injection |
q.s |
Description
REVERTOR™ (atipamezole hydrochloride) is a synthetic α2-adrenergic antagonist which reverses the effects of dexmedetomidine and medetomidine hydrochloride in dogs. The chemical name is 4-(2-ethyl-2,3-dihydro-1H-inden-2-yl)-1H-imidazole hydrochloride. The molecular formula is C14H16N2 • HCl and the structural formula is:
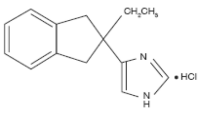
Revertor Indications
REVERTOR™ is indicated for the reversal of the clinical effects of the sedative and analgesic agents, dexmedetomidine hydrochloride and medetomidine hydrochloride, in dogs.
Dosage and Administration
REVERTOR™ is administered intramuscularly regardless of the route used for dexmedetomidine or medetomidine hydrochloride. The concentration of REVERTOR™ has been formulated such that the volume of injection is the same (mL for mL) as the recommended dose volume of dexmedetomidine or medetomidine hydrochloride, and may be given at any time following dexmedetomidine or medetomidine hydrochloride administration.Although injection volumes are the same, the concentration of REVERTOR™ (5.0 mg/mL) is 10 times that of dexmedetomidine hydrochloride (0.5 mg/mL) and 5 times that of medetomidine hydrochloride (1.0 mg/mL). Dogs that are sedated but ambulatory may be treated with REVERTOR™, if warranted.
The dosage of REVERTOR™ is calculated based upon body surface area. Use the table below to determine the proper injection volume based on body weight:
|
Body weight (kg) if dexmedetomidine or medetomidine is given IV |
REVERTOR™ injection Volume (mL) IM |
Body weight (kg) if dexmedetomidine or medetomidine is given IM |
|
2 - 2.9 |
0.1 |
|
|
3 - 3.9 |
0.15 |
2 - 2.9 |
|
4 - 4.9 |
0.2 |
3 - 3.9 |
|
5 - 9.9 |
0.3 |
4 - 4.9 |
|
10 - 14.9 |
0.4 |
5 - 9.9 |
|
15 - 19.9 |
0.5 |
10 - 12.9 |
|
20 - 24.9 |
0.6 |
13 - 14.9 |
|
25 - 29.9 |
0.7 |
15 - 19.9 |
|
30 - 36.9 |
0.8 |
20 - 24.9 |
|
37 - 44.9 |
0.9 |
25 - 29.9 |
|
45 - 49.9 |
1.0 |
30 - 32.9 |
|
50 - 59.9 |
1.1 |
33 - 36.9 |
|
60 - 64.9 |
1.2 |
37 - 44.9 |
|
65 - 74.9 |
1.3 |
45 - 49.9 |
|
75 - 79.9 |
1.4 |
50 - 54.9 |
|
>80 |
1.5 |
55 - 59.9 |
|
|
1.6 |
60 - 64.9 |
|
|
1.7 |
65 - 69.9 |
Revertor Caution
REVERTOR™ can produce an abrupt reversal of sedation and, presumably, analgesia. The potential for apprehensive or aggressive behaviour should be considered in the handling of dogs emerging from sedation, especially those individuals who are likely to be in pain.
Information on use of atipamezole with concurrent drugs is inadequate, therefore caution should be exercised when administering multiple drugs. Animals should be monitored closely, particularly for persistent hypothermia, bradycardia, and depressed respiration, until the animal has recovered completely. Caution should be used in administration of anesthetic agents to elderly or debilitated animals.
While atipamezole does reverse the clinical signs associated with dexmedetomidine or medetomidine sedation, complete physiological return to pretreatment status may not be immediate and should be monitored. REVERTOR™ has not been evaluated in breeding animals; therefore, the drug is not recommended for use in pregnant or lactating animals, or in animals intended for breeding.
NOTE TO USERS: Care should be taken to assure that REVERTOR™ is not inadvertently ingested as safety studies have indicated that the drug is absorbed when administered orally. As with all injectable drugs causing profound physiological effects, routine precautions should be employed when handling and using filled syringes, including washing eye and skin areas affected by accidental spillage. In case of accidental human exposure, a physician should be contacted.
Warning
Keep out of reach of children.Adverse Reactions
Occasional vomiting may occur. Rarely, a brief state of excitement or apprehensiveness may be seen in treated dogs. Other potential side effects of α2-antagonists include hypersalivation, diarrhea, and tremors.Over Dosage: Atipamezole was tolerated in healthy dogs receiving doses at 1-, 3-, and 5-fold doses, in the absence of an α2-antagonist. Signs of overdose were dose-related and consistent with those expected in nonsedated dogs having received a stimulant. Signs seen at elevated doses included excitement, panting, trembling, vomiting, soft or liquid feces or vasodilation (injection) of the sclera. Some localized skeletal muscle injury was seen at the injection site; but no associated clinical signs or complications were observed. Dogs receiving the proper dose in the absence of dexmedetomidine or medetomidine, or 3-fold overdose after dexmedetomidine or medetomidine sedation, exhibited no significant clinical signs.
Clinical Pharmacology
Activation of peripheral and central α2-adrenergic receptors is known to induce a pattern of pharmacological responses including sedation, reduction of anxiety, analgesia, bradycardia, and transient hypertension with a subsequently reduced blood pressure. Atipamezole is a potent α2-antagonist which selectively and competitively inhibits α2-adrenergic receptors. The result of atipamezole administration in the dog is the rapid recovery from the sedation and other clinical effects produced by the α2-adrenergic agonist, dexmedetomidine and medetomidine. Atipamezole is not expected to reverse the effects of other classes of sedatives, anesthetics, or analgesics. Rapid absorption occurs following intramuscular injection, with a maximum serum concentration reached in approximately 10 minutes. Onset of arousal is usually apparent within 5 to 10 minutes of injection, depending on the depth and duration of dexmedetomidine-or medetomidine-induced sedation. Elimination half-life from serum is less than 3 hours. Atipamezole undergoes extensive hepatic biotransformation, with excretion of metabolites primarily in urine.A transient, approximately 10%, decrease in systolic blood pressure occurs immediately after administration of atipamezole to dexmedetomidine- or medetomidine-sedated dogs, followed by an increase in pressure within 10 minutes to the pre-atipamezole level. This is the opposite of the response to α2-agonist therapy, and is probably due to peripheral vasodilation.
Atipamezole will produce a rapid improvement in dexmedetomidine- or medetomidine-induced bradycardia. An increase in heart rate is usually apparent within approximately 3 minutes of injection, but approximately 40% of dogs are not expected to immediately return to presedative rate. Some dogs may experience brief heart rate elevations above baseline. Respiratory rate also increases following atipamezole injection.
Storage
Store at controlled room temperature 15-30°C. Protect from light.Presentation: REVERTOR™ is supplied in 10-mL multidose vials containing 5.0 mg of atipamezole hydrochloride per mL.
Manufactured for:
Modern Veterinary Therapeutics, LLC, Sunrise, Florida 33326 USA
Tel. (888) 590 9839
info@modernveterinarytherapeutics.com
www.modernveterinarytherapeutics.com
Imported by:
Modern Veterinary Therapeutics Inc., 261065 Wagon Wheel Way, Bay 3, Balzac (Rocky View County), AB T4A 0T5
Orders & Product information: call 1 888 590 9839
Made in The Netherlands
Revision date: 20 May 2024
E3525-5.0624
CPN: 1354004.6
261065 WAGON WHEEL WAY, ROCKY VIEW COUNTY, AB, T4A 0T5
| Telephone: | 407-852-8039 | |
| Toll-Free: | 888-590-9839 | |
| Website: | www.modernveterinarytherapeutics.com | |
| Email: | info@modernveterinarytherapeutics.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
