AErrane (isoflurane, USP) (Canada)
This treatment applies to the following species: Company: Baxter
Company: Baxter
(isoflurane, USP)
For Veterinary Use Only
Inhalation Anaesthetic
Action
AErrane (isoflurane, USP) is an inhalation anaesthetic. Induction and particularly recovery from anaesthesia with isoflurane are rapid.1 The level of anaesthesia may be changed rapidly with isoflurane. Isoflurane is a profound respiratory depressant. RESPIRATION MUST BE MONITORED CLOSELY IN THE HORSE AND DOG AND SUPPORTED WHEN NECESSARY. As anaesthetic dose is increased, both tidal volume and respiratory rate decrease.2 This depression is partially recovered by surgical stimulation, even at deeper levels of anaesthesia. No convulsive activity occurs with isoflurane, even with hypocapnia deep levels of anaesthesia or auditory stimulation. Changes with isoflurane anaesthesia are predictable.
Blood pressure decreases with induction of anaesthesia but returns toward normal with surgical stimulation. Progressive increases in depth of anaesthesia produce corresponding decreases in blood pressure; however, heart rhythm is stable and cardiac output is maintained with controlled ventilation and normal PaCO2 despite increasing depth of anaesthesia. The hypercapnia which attends spontaneous ventilation during isoflurane anaesthesia increases heart rate and raises cardiac output above levels observed with controlled ventilation.2 Isoflurane does not sensitize the myocardium to exogenously administered epinephrine in the dog. Limited data in humans indicate that a subcutaneous injection of 0.25 mg of epinephrine (50 mL of 1:200,000 solution) does not produce an increase in ventricular arrhythmias when anaesthetized with isoflurane.
Muscle relaxation may be adequate for intra-abdominal operations at normal levels of anaesthesia. However, if muscle relaxants are used to achieve greater relaxation, it should be noted that: ALL COMMONLY USED MUSCLE RELAXANTS ARE MARKEDLY POTENTIATED WITH ISOFLURANE, THE EFFECT BEING MOST PROFOUND WITH THE NONDEPOLARIZING TYPE. Neostigmine reverses the effect of nondepolarizing muscle relaxants in the presence of isoflurane but does not reverse the direct neuromuscular depression of isoflurane.
AErrane (isoflurane, USP) Indications
AErrane (isoflurane, USP) may be used for induction and maintenance of general anaesthesia in horses and dogs.
Contraindications
AErrane (isoflurane, USP) is contraindicated in horses and dogs with a known history of sensitivity to isoflurane or to other halogenated agents including signs of malignant hyperthermia.
Usage in Pregnancy: Reproduction studies have been performed in mice and rats with no evidence of foetal malformation attributable to isoflurane. Adequate data concerning the use of isoflurane in pregnant and breeding horses and dogs have not been obtained to establish its safe use during pregnancy.
Warning
This drug is not to be administered to horses that are to be slaughtered for use in food.
AErrane (isoflurane, USP) Cautions
The action of nondepolarizing relaxants is augmented by AErrane (isoflurane, USP) Inhalation Anaesthetic. Less than the usual amounts of these drugs should be used. If the usual amounts of nondepolarizing relaxants are given, the time for recovery from myoneural blockade will be longer in the presence of isoflurane than for other commonly used anaesthetics.
Increasing depth of anaesthesia with isoflurane may increase hypotension and respiratory depression and the possibility of overdosing is increased with controlled ventilation.
The electroencephalographic pattern associated with deep anaesthesia is characterized by burst suppression, spiking, and isoelectric periods.3
Care should be exercised to ensure maintenance of the airway during general anaesthesia. Other factors such as: age, pre-existing disease and surgical sites should also be considered when using isoflurane.
Isoflurane, like other halogenated agents, should be used with care in animals with an anaemic condition.
Extreme caution should be used with horses suffering from chronic obstructive pulmonary disease. Isoflurane is as suitable as any other inhalation anaesthetic but blood/gas analysis should be used to evaluate the adequacy of ventilation.
Since levels of anaesthesia may be altered easily and rapidly, only vaporizers producing predictable percentage concentrations of isoflurane should be used (see DOSAGE AND ADMINISTRATION).
No surgical stimulation investigations have been done with isoflurane in horses or in dogs. The following have been extrapolated from human data: surgical stimulation partially reverses respiratory depression produced by anaesthetic depth. Blood pressure decreases with induction of anaesthesia but returns toward normal when surgically stimulated.
Extrapolating from human data, patients suffering from hepatic or renal dysfunction may have impaired ability to deal with muscle relaxants. During isoflurane anaesthesia the amount of muscle relaxant is reduced, thereby decreasing the risk of residual muscle relaxant affecting the postoperative period. Similar considerations should be observed when administering muscle relaxant in the horse and dog. It can also be extrapolated from human data that when using isoflurane to minimize changes in intracranial pressure, under controlled ventilation, isoflurane is the best currently available choice of inhalation anaesthetic.
Adverse Reactions
Hypotension, respiratory depression, and arrhythmias have been reported.
In sensitive animals, isoflurane anaesthesia can induce a hypermetabolic state in the skeletal muscles, which leads to a high oxygen consumption and a clinical syndrome known as malignant hyperthermia (see CONTRAINDICATIONS).
Overdose
Overdose with isoflurane produces marked hypotension and respiratory depression and hypercapnia. In the event of an overdose, or an adverse event, stop drug administration, establish a clear airway and initiate assisted or controlled ventilation with pure oxygen. Administration of fluids may be beneficial.
AErrane (isoflurane, USP) Dosage And Administration
Premedication: A premedication regimen, which may be employed depending upon the patient status, to avert excitement during induction, might include an anticholinergic, a tranquilizer, a muscle relaxant, and a short-acting barbiturate.
Inspired Concentration: The delivered concentration of AErrane (isoflurane, USP) should be known. Isoflurane may be vaporized from a flow-through vaporizer specifically calibrated for isoflurane. Vaporizers delivering a saturated vapour which then is diluted (e.g., Vernitrol® vaporizer) also may be used. The delivered concentration from such a vaporizer may be calculated using the formula:
|
% isoflurane = |
100 PV FV _________ FT (PA-PV) |
where:
PA = Pressure of atmosphere
PV = Vapour pressure of isoflurane
FV = Flow of gas through vaporizer (mL/min)
FT = Total gas flow used (mL/min)
Isoflurane contains no stabilizer. Nothing in the agent alters calibration or operation of these vaporizers.
Induction: Inspired concentration of 3 to 5% isoflurane alone with oxygen are usually employed to induce surgical anaesthesia in the horse. Inspired concentrations of 2.0 to 2.5% isoflurane alone with oxygen following a barbiturate anaesthetic induction are usually employed to induce surgical anaesthesia in the dog. These concentrations can be expected to produce surgical anaesthesia in 5 to 10 minutes.
Although mask inductions have been employed, horses over 136 kilograms should not be mask induced. Isoflurane is compatible with injectable anaesthetics and sedatives and these drugs may be used as indicated in larger horses.
Body temperature should be monitored during anaesthesia and anaesthesia flow adjusted to compensate for changes in anaesthetic requirements associated with increased or decreased body temperatures.
Maintenance: The concentration of vapour necessary to maintain anaesthesia is much less than that required to induce it. Surgical levels of anaesthesia in the horse may be sustained with a 1.5 to 2.0% concentration of isoflurane in oxygen and surgical levels of anaesthesia in the dog may be sustained with a 1.5 to 1.8% concentration of isoflurane in oxygen.
The level of blood pressure during maintenance is an inverse function of isoflurane concentration in the absence of the other complicating problems. Excessive decreases, unless related to hypovolaemia, may be due to depth of anaesthesia and in such instances may be corrected by lightening the level of anaesthesia.
Following isoflurane anaesthesia, variability exists in the time until standing. Older horses appear to be slower to achieve sternal recumbency than younger horses. However, in comparison with other general anaesthetics, recovery is quiet and horses have excellent coordination upon standing.
KEYED BOTTLE COLLAR (FOR USE WITH KEY-FIL VAPORIZER) DIRECTIONS FOR USE:
● To attach a keyed bottle adaptor, remove cap and seal from anaesthetic bottle.
● Check that the anaesthetic bottle neck is not chipped or damaged.
● Match keyed bottle adaptor to keyed bottle collar and screw together until tight.
● Now connect the bottle to the vaporizer filler receptacle.
NOTE THAT COLOUR OF KEYED BOTTLE COLLAR WILL MATCH THE COLOUR OF THE ADAPTOR.
PHYSICAL PROPERTIES
|
Molecular weight |
184.5 |
|
|
Boiling point at 760 mm Hg |
48.5°C |
|
|
Refractive index n20D |
1.2990-1.3005 |
|
|
Specific gravity 25°C |
1.496 |
|
|
Vapour pressure in mm Hg** |
20°C |
238 |
|
25°C |
295 |
|
|
30°C |
367 |
|
|
35°C |
450 |
|
**Equation for vapour pressure calculation:
log10 Pvap = A + B / T
where:
A = 8.056
B = -1664.58
T = °C + 273.16 (Kelvin)
Partition coefficients at 37°C
|
Water / gas |
0.61 |
|
Blood / gas |
1.43 |
|
Oil / gas |
90.8 |
Partition coefficients at 25°C-rubber and plastic
|
Conductive rubber / gas |
62.0 |
|
Butyl rubber / gas |
75.0 |
|
Polyvinyl chloride / gas |
110.0 |
|
Polyethylene / gas |
~2.0 |
|
Polyurethane / gas |
~1.4 |
|
Polyolefin / gas |
~1.1 |
|
Butyl acetate / gas |
~2.5 |
|
Purity by gas chromatography |
>99.9% |
|
Lower limit of flammability in oxygen or nitrous oxide at 9 joules / sec. and 23°C |
None |
|
Lower limit of flammability in oxygen or nitrous oxide at 900 joules / sec. and 23°C |
Greater than useful concentration in anaesthesia. |
MAC (Minimum Alveolar Concentration) in horses (100% oxygen) is 1.31%.4
MAC (Minimum Alveolar Concentration) in dogs (100% oxygen) is 1.28%.5
AErrane (isoflurane, USP) is a clear colourless, stable liquid containing no additives or chemical stabilizers. Isoflurane has a mildly pungent, musty, ethereal odour. Samples stored in indirect sunlight in clear, colourless glass for five years, as well as samples directly exposed for 30 hours to a 2 amp, 115 volt, 60 cycle long wave U.V. light were unchanged in composition as determined by gas chromatography. Isoflurane in one normal sodium methoxidemethanol solution, a strong base, for over six months consumed essentially no alkali, indicative of strong base stability. Isoflurane does not decompose in the presence of soda lime, and does not attack aluminum, tin, brass, iron or copper.
Availability
AErrane (isoflurane, USP) Inhalation Anaesthetic is available in 250 mL amber-coloured bottle.
Storage
Store at room temperature 15°-30°C.
Conditions Of Use
A vaporizer which will allow adequate flows of anaesthetic that can be easily and readily calculated will be required in order to satisfactorily anaesthetize horses and dogs.
Pharmacology
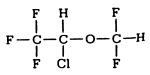
AErrane (isoflurane, USP) is a halogenated methyl ethyl ether with the chemical name 1-chloro-2,2,2-trifluoroethyl difluoromethyl ether, and the structural formula:
Isoflurane produces general anaesthesia on inhalation in horses, mice, dogs, monkeys, rabbits, and rats. The low blood/gas partition coefficient (1.4 for isoflurane compared to 1.9 for enflurane and 2.4 for halothane) permits a rapid induction of and recovery from anaesthesia. Recovery is free from nausea, vomiting, or evidence of malaise.
MAC in horses, the minimum alveolar concentration at which 50% of the animals move in response to pain stimulation, is 1.31%, and in dogs is 1.28%. The anaesthetic index in horses (dose at apnea divided by MAC) is 2.33, and in dogs is 2.5. The anaesthetic index for other anaesthetics is: enflurane 2.26 and halothane 2.60. The anaesthetic index in dogs for other anaesthetics is: enflurane 2.6 and halothane 2.9.
In rats, isoflurane is favored in either of two anaesthetic indices indicating a margin of safety (see Table: Anaesthetic Indices). The respiratory anaesthetic index is the ratio of the brain anaesthetic concentration at respiratory arrest/minimum brain anaesthetic concentration producing anaesthesia. The cardiac arrest index is the ratio of the myocardial anaesthetic concentration at circulatory collapse/minimum heart anaesthetic concentration at which anaesthesia is produced.
Isoflurane has a wide cardiovascular margin of safety.
TABLE: Anaesthetic Indices (Rats)
|
|
Respiratory |
Cardiac |
|
ISOFLURANE |
3.1 |
5.7 |
|
HALOTHANE |
2.3 |
3.0 |
|
METHOXYFLURANE |
2.2 |
3.7 |
|
ENFLURANE |
1.8 |
3.3 |
Like other halogenated agents, isoflurane causes respiratory depression and respiratory acidosis in dogs, rats, rabbits, monkeys, and horses.
The arrhythmic doses of epinephrine in dogs anaesthetized with isoflurane or fluroxene do not differ from those which produce arrhythmias in the awake animal. Significantly lower doses are found for halothane anaesthetized animals. Similarly, the dose of epinephrine producing ventricular extrasystoles in 50 percent of humans anaesthetized with 1.25 MAC isoflurane is 6.7 µg per kg when the epinephrine is injected submucosally. This would equal 47 mL of a 1:100,000 epinephrine containing solution in a 70 kg man.
Isoflurane does not cause convulsive activity either at high, normal or low levels of arterial PaCO2. Twitching or other muscular movement suggesting increased central nervous system hyperactivity is not provoked by isoflurane.
Metabolism
Several metabolic studies were conducted in both animals and man.
In Fischer 344 rats, the peaks of inorganic fluoride ion occurred during the second 24 hours post-anaesthesia; all values had returned to baseline 2-3 days later (organic fluoride to inorganic fluoride ratio 0.53:1). Pre-treatment with phenobarbital did not change the fluoride values. Also in mice and rats, little or no skeletal deposition of fluoride ions did occur, suggesting low or no metabolism for AErrane (isoflurane, USP).
In miniature swine treated with subanaesthetic doses, values indicated little or no metabolism of isoflurane.
In vitro studies confirmed the findings using livers from both untreated and phenobarbital treated mice and rats. When the liver homogenates were exposed to isoflurane, inorganic fluoride production was very low.
In three human volunteers given 0.9% (0.8% MAC) isoflurane for an average of 2.8 hours, it was also found that the metabolism of isoflurane was low. The peaks occurred during the first postanaesthetic day; by the third day all values had returned to pre-anaesthetic levels. Tonic fluoride levels in urine rose from around 100 µM/day pre-anaesthesia to a peak of around 400 µM and had returned to baseline levels on the fourth post-anaesthetic day.
Toxicology
Acute: A single acute study in 4 week old female mice given AErrane (isoflurane, USP) intraperitoneally in olive oil yielded an LD50 of 6.74 µg/kg (5.87-7.77). The animals showed disorientation and hypnosis; at the higher doses, some showed convulsions.
Subacute and Chronic: Groups of five mature beagle dogs were exposed to 1.5 MAC isoflurane, enflurane, halothane, methoxyflurane, nitrous oxide, or thiopental. There was a control group of five animals. Each group was exposed for four hours per day every other day, for a total of 16 hours of anaesthesia. Histopathological examination of liver and kidney tissues showed no cellular damage related to the anaesthetics. Serum creatinine, BUN and SGPT were normal. Urine lysozyme measurements revealed no consistent change in the lysozyme excretion, with no differences among the anaesthetics. It was concluded from these results that there was no renal or hepatic toxic effect from isoflurane or any other anaesthetic.
Five groups of five Rhesus monkeys were exposed for four hours per day on alternate days for a total of 16 hours of anaesthesia. The anaesthetic agents used were isoflurane (1.5-2.5%), enflurane (1.5-2%), methoxyflurane (0.5-0.7%), and halothane (1.0-1.25%). No other drugs were administered. No significant changes in serum creatinine, BUN or SGPT were found. The minimal changes found in liver and kidney tissues did not indicate that isoflurane was nephrotoxic or hepatotoxic.
The chronic toxicity of isoflurane was compared to that of halothane and diethyl ether in mice, rats, and guinea pigs. Animals were exposed continuously for 35 days to 0.05% or 1/30th MAC isoflurane. At the end of this period, all species showed no effect other than a slightly slower weight gain compared to controls. By comparison, halothane animals tolerated only 1/200th MAC, or 0.005% concentration. At concentrations higher than this, halothane exhibited focal hepatic necrosis and lipoidosis which was not seen in the animals exposed to isoflurane.
A 15 month chronic toxicity study was also carried out in Swiss ICR mice. Enflurane, halothane, methoxyflurane and nitrous oxide were used; air and oxygen were given to groups of control mice. The anaesthetics were given at 1/2, 1/8, and 1/32 MAC for four exposures to the mothers during pregnancy, and for 24 exposures to the pups post-partum. The pups were followed to 15 months, at which time they were sacrificed and autopsied. No significant differences were found between isoflurane treated animals and those exposed to halothane, enflurane, methoxyflurane or nitrous oxide.
No incidence of toxic manifestations produced by isoflurane which would be evidenced by imbalances in haematology and serum chemistry values was found in older horses with repeated exposure to isoflurane nor was it found in foals and weanlings with single exposure to the drug.
Reproduction And Teratology
Reproduction studies, including fertility, general reproductive performance, embryotoxicity, teratogenicity, and lactation, did not show any abnormality in mice and rats using AErrane (isoflurane, USP) at 0.1 to 0.6%.
NOTES
1. Auer, J.A., Garner, H.E., Amend, J.F., Hutcheson, D.P. and Salem, C.A.: Recovery from Anesthesia in Ponies: A Comparative Study of the Effects of Isoflurane, Enflurane, Methoxyflurane and Halothane. Equine Vet. J. 10(1): 18-23, 1978.
2. Steffey, E.P., and Howland, D. Jr.: Comparison of Circulatory and Respiratory Effects of Isoflurane and Halothane Anesthesia in Horses. Am. J. Vet. Res. 41(5): 821-825, 1980.
3. Auer, J.A., Amend, J.F., Garner, H.E., Hutcheson, D.P. and Salem, C.A.: Electroencephalographic Responses During Volatile Anesthesia in Domestic Ponies: A Comparative Study of Isoflurane, Enflurane, Methoxyflurane, and Halothane. Equine Practice 3: 130-134, 1979.
4. Steffey, E.P., Howland, D. Jr., Giri, S. and Eger, E.I. II.: Enflurane, Halothane and Isoflurane Potency in Horses. Am. J. Vet. Res. 38(7): 1037-1039, 1977.
5. Steffey, E.P., Howland, D.: Isoflurane Potency in the Dog and Cat. Am. J. Vet. Res. 38, No. 11:1833-1836, 1977.
BIBLIOGRAPHY
1. Stevens, W.C., Dolan, W.M., Gibbons, R.T., White, A., Eger, E.I. II, Miller, R.D., DeJong, R.H., and Elashoff, R.M.: Minimum alveolar concentrations (MAC) of isoflurane with and without nitrous oxide in patients of various ages. Anesthesiology 42(2): 197-200, 1975.
2. Dobkin, A.B., Byles, P.H., Ghanooni, S., and Valbuena, D.A.: Clinical and laboratory evaluation of a new inhalation anaesthetic, Forane® (Compound 469) CHF2-O-CHClCF3. Canad. Anaesth. Soc. J. 18(3): 264-271, 1971.
3. Homi, J., Eckenhoff, J.E., Konchigeri, H.N., and Linde, H.W.: A new anesthetic - Forane®: Preliminary observations in man. Anesth. and Analg. 51(3): 439-447, 1972.
4. Pauca, A.L. and Dripps, R.D.: Clinical experience with isoflurane (Forane®): Preliminary communication. Br. J. Anaesth. 45: 697-703, 1973.
5. Joas, T.A. and Stevens, W.C.: Comparison of the arrhythmic doses of epinephrine during Forane®, halothane and fluroxene anesthesia in dogs. Anesthesiology 35(1): 48-53, 1971.
6. Johnston, R.R., Eger, E.I. II, and Wilson, C.: A comparative interaction of epinephrine with enflurane, isoflurane, and halothane in man. Anesth. and Analg. 55(5): 709-712, 1976.
7. Halsey, M.J., Sawyer, D.C., Eger, E.I. II, Bahlman, S.H., and Impelman, D.M.K.: Hepatic metabolism of halothane, methoxyflurane, cyclopropane, Ethrane®, and Forane® in miniature swine. Anesthesiology 35(1): 43-47, 1971.
8. Fiserova-Bergerova, V.: Changes of fluoride content in bone: An index of drug defluorination in Vivo. Anesthesiology 38(4): 345-351, 1973.
9. Cousins, M.J., Mazze, R.I., Barr, G.A., and Kosek, J.C.: A comparison of the renal effects of isoflurane and methoxyflurane in Fischer 344 rats. Anesthesiology 38(6): 557-563, 1973.
10. Stevens, W.C., Eger, E.I. II, White, A., Halsey, M.J., Munger, W., Gibbons, R.D., Dolan, W., and Shargel, R.: Comparative toxicities of halothane, isoflurane and diethyl ether at subanesthetic concentrations in laboratory animals. Anesthesiology 42(4): 408-419, 1975.
11. Byles, P.H., Dobkin, A.B., and Jones, D.B.: Forane® (Compound 469): 3. Comparative effects of prolonged anaesthesia on mature beagle dogs and young Rhesus monkeys. Canad. Anaesth. Soc. J. 18(4): 397-407, 1971.
12. Eger, E.I. II, White, A.E., Brown, C.L., Biava, C.G., Corbett, T.H., and Stevens, W.C.: A test of the carcinogenicity of enflurane, isoflurane, halothane, methoxyflurane, and nitrous oxide in mice. Anesth. and Analg. 57: 678-694, 1978.
13. Holaday, D.A., Fiserova-Bergerova, V., Latto, I.P., et al.: Resistance of isoflurane to biotransformation in man. Anesthesiology 43: 325-332, 1975.
14. Stevens, W.C., Eger, E.I. II, Joas, T.A., et al.: Comparative toxicity of isoflurane, halothane, fluroxene, and diethyl ether in human volunteers. Canad. Anaesth. Soc. J. 20: 357-368, 1973.
15. Wolfson, B., Hetrick, W.D., Lake, C.L., and Siker, E.S.: Anesthetic indices - further data. Anesthesiology 48: 187-190, 1978.
Baxter and Aerrane are trademarks of Baxter International Inc.
Baxter Corporation, Mississauga, Ontario L5N 0C2
DIN 02225875
November 16, 2016
CPN: 3700000.4
Distributed by AVENTIX ANIMAL HEALTH CORP.
4350 MAINWAY, BURLINGTON, ON, L7L 5R7
| Telephone: | 905-332-4744 | |
| Toll-Free: | 877-909-2242 | |
| Website: | www.aventix.ca | |
| Email: | customerservice@aventix.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
