Esidrix: Package Insert / Prescribing Info
Package insert / product label
Generic name: hydrochlorothiazide
Dosage form: Tablets
Drug class: Thiazide diuretics
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
Esidrix Description
Esidrix, hydrochlorothiazide USP, is a diuretic and antihypertensive available as 25-mg and 50-mg tablets for oral administration. Its chemical name is 6-chloro-3,4-dihydro-2H-1,2,4- benzothiadiazine-7-sulfonamide 1,1-dioxide, and its structural formula is
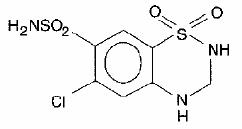
Hydrochlorothiazide USP is a white, or practically white, practically odorless, crystalline powder. It is slightly soluble in water, freely soluble in sodium hydroxide solution, in n - butylamine and in dimethylformamide, sparingly soluble in methanol, and insoluble in ether, in chloroform, and in dilute mineral acids. Its molecular weight is 297.73.
Inactive Ingredients. Colloidal silicon dioxide, D&C Yellow No. 10 (50-mg tablets), FD&C Red No. 40 and FD&C Yellow No. 6 (25- mg tablets), lactose, starch, stearic acid, and sucrose.
Esidrix - Clinical Pharmacology
Thiazides affect the renal tubular mechanisms of electrolyte reabsorption. At maximal therapeutic dosage all thiazides are approximately equal in their diuretic potency. Thiazides increase excretion of sodium and chloride in approximately equivalent amounts. Natriuresis causes a secondary loss of potassium.
The mechanism of the antihypertensive effect of thiazides is unknown. Thiazides do not affect normal blood pressure.
Onset of action of thiazides occurs in 2 hours and the peak effect at about 4 hours. Its action persists for approximately 6 to 12 hours. Thiazides are eliminated rapidly by the kidney.
Indications and Usage for Esidrix
Hypertension
In the management of hypertension either as the sole therapeutic agent or to enhance the effect of other antihypertensive drugs in the more severe forms of hypertension.
Edema
As adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
Esidrix has also been found useful in edema due to various forms of renal dysfunction, such as the nephrotic syndrome, acute glomerulonephritis, and chronic renal failure.
Usage in Pregnancy: The routine use of diuretics in an otherwise healthy woman is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy, and there is no satisfactory evidence that they are useful in the treatment of developed toxemia.
Edema during pregnancy may arise from pathological causes or from the physiologic and mechanical consequences of pregnancy. Thiazides are indicated in pregnancy when edema is due to pathologic causes, just as they are in the absence of pregnancy (however, see PRECAUTIONS, Pregnancy). Dependent edema in pregnancy, resulting from restriction of venous return by the expanded uterus, is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy which is not harmful to either the fetus or the mother (in the absence of cardiovascular disease) but which is associated with edema, including generalized edema, in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances, this edema may cause extreme discomfort which is not relieved by rest. In these cases, a short course of diuretics may provide relief and may be appropriate.
Warnings
Use with caution in severe renal disease. In patients with renal disease, thiazides may precipitate azotemia. Cumulative effects of the drug may develop in patients with impaired renal function.
Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor alterations of fluid and electrolyte imbalance may precipitate hepatic coma.
Thiazides may add to or potentiate the action of other antihypertensive drugs. Potentiation occurs with ganglionic or peripheral adrenergic blocking drugs.
Sensitivity reactions are more likely to occur in patients with a history of allergy or bronchial asthma.
The possibility of exacerbation or activation of systemic lupus erythematosus has been reported.
Precautions
General
All patients receiving thiazide therapy should be observed for clinical signs of fluid or electrolyte imbalance: namely, hyponatremia, hypochloremic alkalosis, and hypokalemia (see Laboratory Tests and Drug/Drug Interactions). Warning signs are dryness of mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbance such as nausea or vomiting.
Hypokalemia may develop, especially with brisk diuresis or when severe cirrhosis is present.
Interference with adequate oral intake of electrolytes will also contribute to hypokalemia. Hypokalemia may be avoided or treated by use of potassium supplements or foods with a high potassium content.
Any chloride deficit is generally mild and usually does not require specific treatment except under extraordinary circumstances (as in liver disease or renal disease). Dilutional hyponatremia may occur in edematous patients in hot weather; appropriate therapy is water restriction rather than administration of salt, except in rare instances when the hyponatremia is life-threatening. In actual salt depletion, appropriate replacement is the therapy of choice.
Hyperuricemia may occur or frank gout may be precipitated in certain patients receiving thiazide therapy.
Latent diabetes may become manifest during thiazide administration (see Drug/Drug Interactions).
The antihypertensive effects of the drug may be enhanced in the postsympathectomy patient.
If progressive renal impairment becomes evident, withholding or discontinuing diuretic therapy should be considered.
Calcium excretion is decreased by thiazides. Pathological changes in the parathyroid gland with hypercalcemia and hypophosphatemia have been observed in a few patients on prolonged thiazide therapy. The common complications of hyperparathyroidism such as renal lithiasis, bone resorption, and peptic ulceration have not been seen.
Thiazide diuretics have been shown to increase the urinary excretion of magnesium; this may result in hypomagnesemia.
Information for Patients
Patients should be informed of possible side effects and advised to take the medication regularly and continuously as directed.
Laboratory Tests
Initial and periodic determinations of serum electrolytes to detect possible electrolyte imbalance should be performed at appropriate intervals.
Serum and urine electrolyte determinations are particularly important when the patient is vomiting excessively or receiving parenteral fluids.
Drug/Drug Interactions
Hypokalemia can sensitize or exaggerate the response of the heart to the toxic effects of digitalis (e.g., increased ventricular irritability).
Hypokalemia may develop during concomitant use of steroids or ACTH.
Insulin requirements in diabetic patients may be increased, decreased, or unchanged.
Thiazides may decrease arterial responsiveness to norepinephrine. This diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use.
Thiazide drugs may increase the responsiveness to tubocurarine.
Lithium renal clearance is reduced by thiazides, increasing the risk of lithium toxicity.
There have been rare reports in the literature of hemolytic anemia occurring with the concomitant use of hydrochlorothiazide and methyldopa.
Concurrent administration of some nonsteroidal anti-inflammatory agents may reduce the diuretic, natriurefic and anti-hypertensive effects of thiazide diuretics.
Cholestyramine and colestipol resins: Absorption of hydrochlorothiazide is impaired in the presence of anionic exchange resins. Single doses of either cholestyramine or colestipol resins bind the hydrochlorothiazide and reduce its absorption from the gastrointestinal tract by up to 85% and 43%, respectively.
Drug/Laboratory Test Interactions
Thiazides may decrease serum PBI levels without signs of thyroid disturbance.
Thiazides should be discontinued before carrying out tests for parathyroid function (see PRECAUTIONS, General, calcium excretion).
Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year feeding studies in mice and rats conducted under the auspices of the National Toxicology Program (NTP) uncovered no evidence of a carcinogenic potential of hydrochlorothiazide in female mice (at doses of up to approximately 600 mg/kg/day) or in male and female rats (at doses of up to approximately 100 mg/kg/day). The NTP, however, found equivocal evidence for hepatocarcinogenicity in male mice.
Hydrochlorothiazide was not genotoxic in in vitro assays using strains TA 98, TA 100, TA 1535, TA 1537, and TA 1538 of Salmonella typhimurium (Ames assay) and in the Chinese Hamster Ovary (CHO) test for chromosomal aberrations, or in in vivo assays using mouse germinal cell chromosomes, Chinese hamster bone marrow chromosomes, and the Drosophila sex-linked recessive lethal trait gene. Positive test results were obtained only in the in vitro CHO Sister Chromatid Exchange (clastogenicity) and in the Mouse Lymphoma Cell (mutagenicity) assays, using concentrations of hydrochlorothiazide from 43 to 1300 µg/mL, and in the Aspergillus nidulans nondisjunction assay at an unspecified concentration.
Hydrochlorothiazide had no adverse effects on the fertility of mice and rats of either sex in studies wherein these species were exposed, via their diet, to doses of up to 100 and 4 mg/kg/day, respectively, prior to mating and throughout gestation.
Pregnancy
Teratogenic Effects. Pregnancy Category B
Studies in which hydrochlorothiazide was orally administered to pregnant mice and rats during their respective periods of major organogenesis at doses up to 3000 and 1000 mg/kg/day, respectively, provided no evidence of harm to the fetus. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Adverse Reactions/Side Effects
Adverse reactions are usually reversible upon reduction of dosage or discontinuation of Esidrix. Whenever adverse reactions are moderate or severe, it may be necessary to discontinue the drug.
The following adverse reactions have been observed, but there has not been enough systematic collection of data to support an estimate of their frequency. Consequently the reactions are categorized by organ systems and are listed in decreasing order of severity and not frequency.
Digestive: Pancreatitis, jaundice (intrahepatic cholestatic), sialadenitis, vomiting, diarrhea, cramping, nausea, gastric irritation, constipation, anorexia.
Cardiovascular: Orthostatic hypotension (may be potentiated by alcohol, barbiturates, or narcotics).
Neurologic: Vertigo, dizziness, transient blurred vision, headache, paresthesia, xanthopsia, weakness, restlessness.
Musculoskeletal: Muscle spasm.
Hematologic: Aplastic anemia, agranulocytosis, leukopenia, thrombocytopenia.
Metabolic: Hyperglycemia, glycosuria, hyperuricemia.
Hypersensitive Reactions: Necrotizing angiitis, Stevens- Johnson syndrome, respiratory distress including pneumonitis and pulmonary edema, purpura, urticaria, rash, photosensitivity.
Related/similar drugs
Overdosage
Acute Toxicity
No deaths due to acute poisoning with Esidrix have been reported.
Highest known doses ingested: children, 500 mg (14-year-old girl); young children, 125 mg (2 1/2-year-old child).
Oral LD50 in rats: >2750 mg/kg.
Signs and Symptoms
The most prominent feature of poisoning with Esidrix is acute loss of fluid and electrolytes.
Cardiovascular: Tachycardia, hypotension, shock.
Neuromuscular: Weakness, confusion, dizziness, cramps of the calf muscles, paresthesia, fatigue, impairment of consciousness.
Gastrointestinal: Nausea, vomiting, thirst.
Renal: Polyuria, oliguria or anuria (due to hemoconcentration).
Laboratory Findings: Hypokalemia, hyponatremia, hypochloremia, alkalosis, increased BUN (especially in patients with renal insufficiency).
Combined Poisoning: Signs and symptoms may be aggravated or modified by concomitant intake of antihypertensive medication, barbiturates, curare, digitalis (hypokalemia), corticosteroids, narcotics, or alcohol.
Treatment
There is no specific antidote.
Elimination of the drug: Induction of vomiting, gastric lavage.
Measures to reduce absorption: Activated charcoal.
Hypotension, shock: The patient’s legs should be kept raised, and lost fluid and electrolytes (potassium, sodium) should be replaced.
Surveillance: Fluid and electrolyte balance (especially serum potassium) and renal function should be monitored until conditions become normal.
Esidrix Dosage and Administration
Therapy should be individualized according to patient response.
Dosage should be titrated to gain maximal therapeutic response as well as the minimal dose possible to maintain that therapeutic response.
ADULTS
Hypertension
To Initiate Therapy: Usual dosage is 50-100 mg daily. May be given as a single dose every morning.
Maintenance: After a week dosage may be adjusted downward to as little as 25 mg a day, or upward. Rarely patients may require up to 200 mg daily in divided doses.
Combined Therapy: When necessary, other antihypertensive agents may be added cautiously. Since this drug potentiates the antihypertensive effect of other agents, such additions should be gradual. Dosages of ganglionic blockers in particular should be halved initially.
INFANTS AND CHILDREN
The usual pediatric dosage is administered twice daily.
The total daily dosage for infants up to 2 years of age: 12.5 to 37.5 mg; for children 2 to12 years of age: 37.5 to100 mg. Dosages should be based on body weight at the rate of 1 mg per pound, but infants below 6 months of age may require 1.5 mg per pound.
How is Esidrix supplied
Tablets 25 mg – round, pink, scored (imprinted CIBA 22)
Bottles of 100 ..... NDC 0083-0022-30
Tablets 50 mg – round, yellow, scored (imprinted CIBA 46)
Bottles of 100 ..... NDC 0083-0046-30
Do not store above 86ºF (30ºC).
Dispense in tight, light-resistant container (USP).
667903
C96-27 (Rev. 3/96)
C I B A
Ciba-Geigy Corporation
Pharmaceuticals Division
Summit, New Jersey 07901
| ESIDRIX
hydrochlorothiazide tablet |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| ESIDRIX
hydrochlorothiazide tablet |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Ciba-Geigy Corporation |
Frequently asked questions
- What is the best time of day to take blood pressure medication?
- How long does hydrochlorothiazide stay in your system?
- Should you use a diuretic with diazoxide?
More about Esidrix (hydrochlorothiazide)
- Check interactions
- Compare alternatives
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: thiazide diuretics
- Breastfeeding

