Anaspaz: Package Insert / Prescribing Info
Package insert / product label
Generic name: hyoscyamine sulfate
Dosage form: tablet, orally disintegrating
Drug class: Anticholinergics / antispasmodics
Medically reviewed by Drugs.com. Last updated on Jul 29, 2024.
On This Page
Anaspaz Description
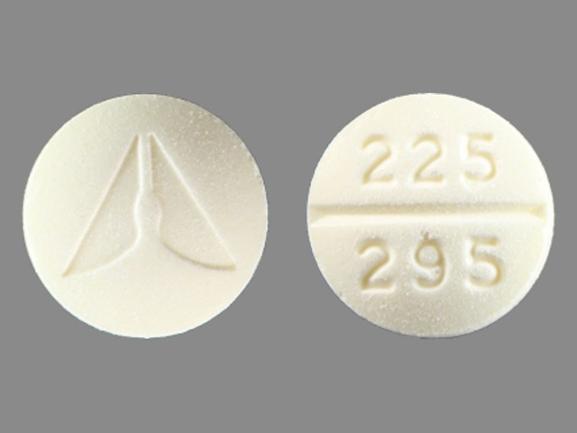
Each ANASPAZ orally disintegrating tablet contains 1-hyoscyamine sulfate 0.125 mg. ANASPAZ may be taken orally (swallowed or chewed) or sublingually. ANASPAZ tablets are compressed, light yellow and scored with the Ascher logo on one side and 225/295 on the other. Inactive ingredients: DC Yellow #10, FDC yellow #6, lactose NF, magnesium stearate NF, mannitol USP, sorbitol NF, pre-gelatinized starch NF, stearic acid NF.
ANASPAZ is chemically pure 1-hyoscyamine sulfate, one of the principal anticholinergic/antispasmodic components of belladonna alkaloids. Chemically, it is benzeneacetic acid, α-(hydroxymethyl)-, 8-methyl-8-azabicyclo[3.2.1.]oct-3-yl ester, [3(S)-endo]-, sulfate (2:1), dihydrate with the chemical formula (C17H23NO3)2•H2SO4•2H2O
Anaspaz - Clinical Pharmacology
ANASPAZ inhibits specifically the actions of acetylcholine on structures innervated by postganglionic cholinergic nerves and on smooth muscles that respond to acetylcholine but lack cholinergic innervation. These peripheral cholinergic receptors are present in the autonomic effector cells of smooth muscle, cardiac muscle, the sino-atrial node, the atrioventricular node and exocrine glands. At therapeutic doses, it is completely devoid of any action in the autonomic ganglia. ANASPAZ inhibits gastrointestinal propulsive motility and decreases gastric acid secretion. ANASPAZ also controls excessive pharyngeal, tracheal and bronchial secretions. ANASPAZ is absorbed totally and completely by sublingual administration as well as oral administration. Once absorbed, ANASPAZ disappears rapidly from the blood and is distributed throughout the entire body. The half-life of ANASPAZ is 3.5 hours and the majority of drug is excreted in the urine unchanged within the first 12 hours, with a small amount hydrolyzed to tropic acid and tropine. Only traces of this drug are found in breast milk. ANASPAZ passes the blood-brain and placental barriers.
Indications and Usage for Anaspaz
ANASPAZ is effective as adjunctive therapy in the treatment of peptic ulcer and irritable bowel syndrome (irritable colon, spastic colon, mucous colitis), acute entercolitis and other functional gastrointestinal disorders. It can also be used to control gastric secretion, visceral spasm and hypermotility in cystitis, pylorospasm and associated abdominal cramps. May be used in functional intestinal disorders to reduce symptoms such as those seen in mild dysenteries and diverticulitis. ANASPAZ is indicated (along with appropriate analgesics) in symptomatic relief of biliary and renal colic and as a drying agent in the relief of symptoms of acute rhinitis.
Contraindications
Glaucoma, obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis); paralytic ileus; intestinal atony of elderly or debilitated patients; unstable cardiovascular status; severe ulcerative colitis; toxic megacolon; myasthenia gravis; myocardial ischemia.
Warnings
In the presence of high environmental temperature, heat prostration can occur with drug use (fever and heat stroke due to decreased sweating). Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance, treatment with this drug would be inappropriate and possibly harmful. Like other anticholinergic agents, ANASPAZ may produce drowsiness or blurred vision. In this event, the patient should be warned not to engage in activities requiring mental alertness such as operating a motor vehicle or other machinery or to perform hazardous work while taking this drug.
Anticholinergic psychosis has been reported in sensitive individuals given anticholinergic drugs. CNS signs and symptoms include confusion, disorientation, short term memory loss, hallucinations, dysarthria, ataxia, coma, euphoria, decreased anxiety, fatigue, insomnia, agitation and mannerisms, and inappropriate affect. These CNS signs and symptoms usually resolve 12 to 48 hours after drug discontinuation.
Elderly patients may react with excitement, agitation, drowsiness, and other unfavorable manifestations to even small doses of ANASPAZ.
Precautions
General:
Use with caution and only when clearly indicated in patients with autonomic neuropathy, hyperthyroidism, coronary heart disease, congestive heart failure, cardiac arrhythmias, hypertension, and renal disease. Investigate any tachycardia before giving any anticholinergic drug since they may increase the heart rate. Use with caution in patients with hiatal hernia associated with reflux esophagitis.
Information for Patients:
ANASPAZ may produce drowsiness, dizziness or blurred vision. Patients should observe caution before operating a motor vehicle or other machinery or performing other tasks requiring mental alertness.
Use of ANASPAZ may decrease sweating resulting in heat prostration, fever or heat stroke; febrile patients or those who may be exposed to elevated environmental temperatures should use caution.
Drug Interactions:
Additive adverse effects resulting from cholinergic blockade may occur when ANASPAZ is administered concomitantly with other anti-muscarinics, amantadine, haloperidol, phenothiazines, monoamine oxidase (MAO) inhibitors, tricyclic antidepressants or some antihistamines.
Antacids may interfere with the absorption of ANASPAZ; take ANASPAZ before meals and antacids after meals.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
No long-term studies in animals have been performed to evaluate the carcinogenic, mutagenic or impairment of fertility potential of ANASPAZ in either males or females.
Pregnancy-Pregnancy Category C:
Animal reproduction studies have not been conducted with ANASPAZ. It is also not known whether ANASPAZ can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. ANASPAZ should be given to a pregnant woman only if clearly needed.
Nursing Mothers:
ANASPAZ is excreted in human milk. Caution should be exercised when ANASPAZ is administered to a nursing woman.
Pediatric Use:
Safety and effectiveness in pediatric patients below the age of 2 have not been established.
Geriatric use:
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Sedating drugs may cause confusion and over-sedation in the elderly; elderly patients generally should be started on low doses of ANASPAZ and observed closely.
Adverse Reactions/Side Effects
Not all the following adverse reactions have been associated with ANASPAZ, but have been reported for drugs in the same pharmacological class, with anticholinergic / antispasmodic action. Adverse reactions may include dryness of the mouth, urinary hesitancy, urinary retention, tachycardia, palpitations, blurred vision, mydriasis, cycloplegia, increased intraocular pressure, dry eyes, headache, nervousness, drowsiness, dizziness, weakness, mental confusion and/or excitement (especially in geriatric patients), flushing, insomnia, fever, altered sense of taste, nausea, vomiting, dysphagia, heartburn, constipation, bloated feeling, suppression of lactation, impotence, and decreased sweating. Allergic reactions or drug idiosyncrasies such as anaphylaxis, urticaria and other dermal manifestations may also occur.
Related/similar drugs
Overdosage
The signs and symptoms of overdose include headache, nausea, vomiting, dizziness, dry mouth, difficulty in swallowing, dilated pupils, blurred vision, urinary retention, hot dry and flushed skin, tachycardia, hypertension, hypotension, respiratory depression, CNS stimulation, fever, ataxia, excitation, lethargy, stupor, coma, and paralysis (with large overdoses). General measures such as emesis or gastric lavage and administration of activated charcoal should be undertaken immediately. Supportive therapy is given as needed, including artificial respiration if required. Physostigmine may be given by intravenous injection to reverse severe anticholinergic symptoms.
The LD50 for hyoscyamine in rats is 375 mg/kg.
Hyoscyamine sulfate is dialyzable.
Anaspaz Dosage and Administration
Adults and children 12 years of age and older: 1 or 2 tablets every four hours or as needed. Do not exceed 12 tablets in 24 hours. Children 2 to under 12 years of age: 1/2 to 1 tablet every four hours or as needed. Do not exceed 6 tablets in 24 hours.
ANASPAZ may be taken orally (swallowed or chewed) or sublingually. The dosage of ANASPAZ should be adjusted to the needs of the individual patient to assure symptomatic control with a minimum of adverse effects.
How is Anaspaz supplied
ANASPAZ (l-hyoscyamine sulfate orally disintegrating tablets) 0.125mg is available as a compressed, light yellow, scored tablet, imprinted with the Ascher logo and 225/295 in bottles of 100 tablets (NDC 0225-0295-15) and 500 tablets (NDC 0225-0295-20).
| ANASPAZ
hyoscyamine sulfate tablet, orally disintegrating |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - BF ASCHER AND CO INC (003854403) |
More about Anaspaz (hyoscyamine)
- Check interactions
- Compare alternatives
- Reviews (1)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: anticholinergics/antispasmodics
- En español
Professional resources
Other brands
Levsin, Hyosyne, Levsin SL, Levbid, ... +5 more