Uptravi Dosage
Generic name: SELEXIPAG 200ug
Dosage form: tablet, coated
Drug class: Agents for pulmonary hypertension
Medically reviewed by Drugs.com. Last updated on Jul 8, 2024.
Recommended Dosage
UPTRAVI Film-coated Tablets
The recommended starting dosage of UPTRAVI tablets is 200 micrograms (mcg) given twice daily. Tolerability may be improved when taken with food .
Increase the dose in increments of 200 mcg twice daily, usually at weekly intervals, to the highest tolerated dose up to
1600 mcg twice daily. If a patient reaches a dose that cannot be tolerated, the dose should be reduced to the previous tolerated dose.
Do not split, crush, or chew tablets.
UPTRAVI for Injection
Use UPTRAVI for injection in patients who are temporarily unable to take oral therapy.
Administer UPTRAVI for injection twice daily by intravenous infusion at a dose that corresponds to the patient's current dose of UPTRAVI tablets (see Table 1). Administer UPTRAVI for injection as an 80-minute intravenous infusion.
Preparation Instructions
Reconstitute and further dilute UPTRAVI for injection prior to intravenous infusion following aseptic procedures.
Determine the dose and total volume of reconstituted UPTRAVI solution required (see Table 1).
Reconstitution
- Remove the carton of UPTRAVI for injection from the refrigerator and allow to stand for approximately 30 to 60 minutes to reach room temperature (20°C to 25°C [68°F to 77°F]).
- The vial needs to be protected from light at all times. Ensure the protective wrap around label is covering the entire vial.
- Peel back light protective wrap on vial to inspect the contents in the vial. It should appear white to almost white broken cake or powdered material. Immediately close the light protective wrap on the vial.
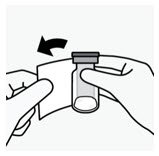
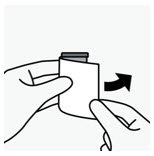
- Reconstitute UPTRAVI for injection using a polypropylene syringe with 8.6 mL of 0.9% Sodium Chloride Injection, USP and slowly inject into the UPTRAVI vial with the stream directed toward the inside wall of the vial to obtain a concentration of 225 mcg/mL of selexipag.
- Document date and time of first puncture. Complete infusion within 4 hours of first puncture.
- Gently invert the vial and repeat until powder is completely dissolved. Do not shake.
- Inspect the vial by peeling back the light protective wrap around label for discoloration. The reconstituted solution should appear clear, colorless and free from foreign material. Do not use if the reconstituted solution is discolored, cloudy, or contains visible particles.
Dilution
- UPTRAVI for injection must be diluted in glass containers only.
- Withdraw 100 mL of 0.9% Sodium Chloride Injection, USP and transfer into an empty sterile glass container.
- Withdraw the required volume of reconstituted solution (see Table 1 for reconstituted transfer volume) from the UPTRAVI vial using a single, appropriately sized polypropylene syringe and dilute into the glass container containing 100 mL 0.9% Sodium Chloride Injection, USP to obtain the desired final dose.
- Mix the diluted UPTRAVI infusion solution by gentle inversion of the glass container 5 times. Do not shake.
- Protect diluted UPTRAVI infusion solution from light at all times. Assign a 4-hour expiry from the time of first vial puncture and wrap the glass container completely with light protective cover.
- The UPTRAVI infusion solution should be kept at room temperature (20°C–25°C [68°F–77°F]) and must be infused within 4 hours from the first puncture of the vial stopper (including infusion time).
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. The diluted UPTRAVI infusion solution should be clear and colorless. Discard if particulate matter is observed.
UPTRAVI for injection vials are single-dose, for single administration. All remaining reconstituted product must be discarded.
| UPTRAVI tablets dose (mcg) for twice daily dosing | Corresponding IV UPTRAVI Dose (mcg) for twice daily dosing | Reconstituted transfer volume (mL) for dilution |
|---|---|---|
| 200 | 225 | 1.0 |
| 400 | 450 | 2.0 |
| 600 | 675 | 3.0 |
| 800 | 900 | 4.0 |
| 1000 | 1125 | 5.0 |
| 1200 | 1350 | 6.0 |
| 1400 | 1575 | 7.0 |
| 1600 | 1800 | 8.0 |
Administration Instructions
Administer by intravenous infusion over 80 minutes using an infusion set made of DEHP-free polyvinyl chloride (PVC), natural latex rubber-free microbore tubing protected from light.
Do not use a filter for administration.
Once the solution for infusion glass container is empty, continue the infusion at the same rate with 0.9% saline to empty the remaining solution for infusion in the IV line, to ensure that the entire solution for infusion has been administered.
Interruptions and Discontinuations
If a dose of UPTRAVI is missed, patients should take a missed dose as soon as possible unless the next dose is within the next 6 hours.
If treatment is missed for 3 days or more, restart UPTRAVI at a lower dose and then retitrate.
Dosage Adjustment in Patients with Hepatic Impairment
No dose adjustment of UPTRAVI is necessary for patients with mild hepatic impairment (Child-Pugh class A).
For patients with moderate hepatic impairment (Child-Pugh class B), the starting dose of UPTRAVI tablets is 200 mcg once daily. Increase in increments of 200 mcg once daily at weekly intervals, as tolerated .
Avoid use of UPTRAVI in patients with severe hepatic impairment (Child-Pugh class C).
Frequently asked questions
More about Uptravi (selexipag)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (28)
- Drug images
- Side effects
- During pregnancy
- Generic availability
- FDA approval history
- Drug class: agents for pulmonary hypertension
- Breastfeeding
- En español
Patient resources
- Uptravi drug information
- Uptravi (Selexipag Intravenous) (Advanced Reading)
- Uptravi (Selexipag Oral) (Advanced Reading)
Professional resources
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.




