The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
Vista 5 VL5 SQ (Canada)
This treatment applies to the following species:BOVINE RHINOTRACHEITIS-VIRUS DIARRHEA-PARAINFLUENZA-3-RESPIRATORY SYNCYTIAL VIRUS VACCINE, Modified Live Virus-CAMPYLOBACTER FETUS-LEPTOSPIRA CANICOLA-GRIPPOTYPHOSA-HARDJO-ICTEROHAEMORRHAGIAE- POMONA Bacterin
Cattle Vaccine
Description
The reconstituted vaccine-bacterin product contains modified-live cultures of infectious bovine rhinotracheitis (IBR) virus, bovine virus diarrhea (BVD) viruses Types 1 & 2, parainfluenza-3 (PI-3) virus, bovine respiratory syncytial virus (BRSV) and inactivated cultures of Campylobacter fetus and Leptospira canicola, L. grippotyphosa, L. hardjo, L. icterohaemorrhagiae and L. pomona with a proprietary adjuvant.
Vista 5 VL5 SQ Indications
For use in healthy cows and heifers 6 months of age or older, prior to breeding, as an aid in the reduction of abortion caused by IBR virus and as an aid in the prevention of fetal infection, including persistently infected calves caused by BVD viruses Types 1 & 2. In addition, it can be used as an aid in the prevention of disease caused by IBR, BVD Type 2 and BRSV viruses, as an aid in the control of disease caused by BVD Type 1 and PI-3 viruses, as an aid in the reduction of infertility (reproductive disease caused by Campylobacter fetus), as an aid in the prevention of leptospirosis (caused by Leptospira canicola, L. grippotyphosa, L. hardjo-including the L. borgpetersenii serovar hardjo bovis, L. icterohaemorrhagiae and L. pomona) and as an aid in the prevention of urinary shedding of L. hardjo organisms.
Respiratory duration of immunity (DOI) has been demonstrated to be at least 182 days for IBR virus, at least 206 days for BVD virus Type 1 and at least 200 days for BVD virus Type 2.
Reproductive DOI has been demonstrated to be at least 217 days for IBR virus and at least 206 days for BVD viruses Types 1 & 2.
May be used in pregnant cattle provided they were vaccinated prior to breeding according to label directions, with any of the modified live IBR and BVD containing vaccine(s) in this product line.
Prior to use of this vaccine during pregnancy and consistent with good vaccination practices, it is recommended that cows and heifers receive for primary vaccination at least 2 doses of a modified live IBR and BVD containing vaccine(s) in this product line, with the second dose given at or about 30 days prior to breeding.
May be used in calves nursing pregnant cows provided their dams were vaccinated prior to breeding according to label directions.
Use Directions:
General Directions:
10 Doses - Rehydrate freeze dried vial of Vista® 5 SQ with the accompanying vial of VL5 SQ. Mix reconstituted vial well and administer 2 mL subcutaneously to healthy cattle of 6 months of age or older.
50 Doses - Rehydrate freeze dried vial of Vista® 5 SQ with part of the accompanying vial of VL5 SQ using the transfer needle provided (see below for pictorial directions). Mix reconstituted vial well and transfer rehydrated vaccine into VL5 SQ vial using transfer needle. Remove transfer needle from VL5 SQ vial and mix rehydrated vial well. Peel label from bottle of Vista® 5 SQ and place on VL5 SQ vial containing the rehydrated vaccine. Administer 2 mL subcutaneously to healthy cattle of 6 months of age or older.
Primary Vaccination: Administer a single 2-mL dose to all breeding cows and heifers 14-60 days prior to breeding or being added to the herd.
Revaccination: Annual revaccination is recommended. A revaccination dose can be administered at more frequent intervals based upon individual farm disease risk assessment or any time epidemic conditions exist or are reported. Consult your veterinarian.
Mixing Instructions
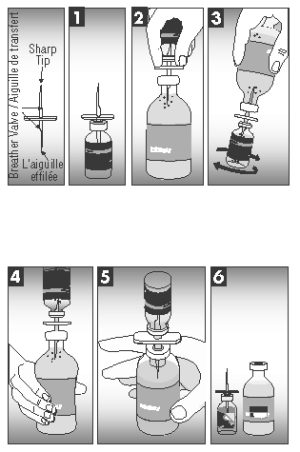
CAUTION: THE TRANSFER NEEDLE, INCLUDED IN THE CARTON PACKAGING (50 DOSES), IS SHARP AND MAY CAUSE INJURY TO SELF OR ANIMALS IF NOT HANDLED OR DISPOSED OF PROPERLY.
1. Insert needle beyond breather valve into the vial of lyophilized vaccine to remove vacuum.
2. Turn the vial of lyophilized vaccine upside down and fully insert needle into bacterin diluent bottle. Be sure to clear cap with breather valve.
3. Turn attached vial and bottle so the vial of lyophilized vaccine is underneath the bacterin diluent bottle and squeeze enough bacterin diluent into the vial of lyophilized vaccine to rehydrate the vaccine. Mix well.
4. Turn attached bottles so rehydrated vaccine is above bacterin diluent bottle. Squeeze air into rehydrated vaccine vial.
5. Squeeze and release bacterin diluent bottle until all the solution in the rehydrated vaccine vial drains into the bacterin diluent bottle.
6. Separate the vial of lyophilized vaccine and needle from the bacterin diluent bottle. Using the tab, remove the back of the vaccine vial label. Place the separated label over the bacterin diluent bottle label to accurately identify the newly created solution.
Cautions: Store at 2°-7°C (35°-45°F). Do not freeze. Use entire contents immediately after rehydration. Dispose of containers and all unused vaccine according to local biohazardous waste disposal regulations. Use only in healthy cattle. Do not vaccinate within 21 days before slaughter. Fetal health risks associated with vaccination of pregnant animals with modified live vaccines cannot be unequivocally determined by clinical trials conducted for licensure. Vaccination of pregnant animals with modified live vaccines should be discussed with your veterinarian. If allergic reaction occurs, treat with epinephrine. Contains penicillin, streptomycin & thimerosal as preservatives.
FOR ANIMAL USE ONLY
Intervet Inc., Omaha, NE 68103 USA
U.S. Veterinary License No. 165A
1 866 683-7838 (Canada)
|
|
|
Code |
|
|
10 Doses |
20 mL |
105397 |
141502-02 |
|
50 Doses |
100 mL |
107197 |
129615-02 |
CPN: 1208204.4
Intervet Canada Corp.
16750 ROUTE TRANSCANADIENNE, KIRKLAND, QC, H9H 4M7
| Order Desk: | 514-428-7013 | |
| Toll-Free: | 866-683-7838 | |
| Fax: | Toll-free 888-498-4444; local 514-428-7014 | |
| Website: | www.merck-animal-health.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-05-29
