The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
VetScan Canine Anaplasma Antibody Test Kit
This treatment applies to the following species:FOR THE QUALITATIVE DETECTION OF ANTIBODIES TO A. PHAGOCYTOPHILUM AND/OR A. PLATYS IN CANINE WHOLE BLOOD, SERUM, OR PLASMA
For Veterinary Use Only
Intended Use
The VetScan Anaplasma Antibody Test Kit is a visual rapid test for the qualitative detection of antibodies to A. phagocytophilum and/or A. platys in canine whole blood, serum or plasma. This test is for veterinary use only. Gram negative bacteria belonging to the above Anaplasma spp. are obligatory intracellular parasites and cause the disease known as Anaplasmosis in man, dogs and some other animals. This disease is transmitted by a variety of tick vectors and has a world-wide distribution. Clinical signs of Anaplasmosis differ depending on the species of bacteria causing disease. A. phagocytophilum causes granulocytic Anaplasmosis causing hematologic abnormalities and formation of Morulae in granulocytes. Common hematological changes include thrombocytopenia, leukopenia and anemia. Increases in serum alkaline phosphatase, alanine transferase and total bilirubin but reduction in albumin levels has been noted in granulocytic Anaplasmosis. A. platys infection in dogs causes bacteria to accumulate in platelets. The appearance of Morulae in platelets is cyclical in nature. Apart from thrombocytopenia, A. platys infection is characterized by bleeding disorders.
The VetScan Anaplasma Antibody Test Kit uses synthetic peptides that bind antibodies produced in dogs in response to certain dominant antigens of the above two Anaplasma spp. Co-polymers of synthetic peptides with bovine serum albumin are coated on colloidal gold particles and used in a double antigen sandwich assay to visualize the presence of antibodies binding to these peptides. The antibodies bound to antigen-coated gold particles flows through the strip and are captured by immobilized antigen on the test strip. The accumulation of the captured gold particle/antibody complex causes a color to become visible on the Test Line (T). The intensity of the colored line is further enhanced by an amplification mechanism. A procedural Control Line (C) will always appear whether the sample is positive or negative.
INSTRUCTION FOR USE
● This Test is for the detection of Anaplasma spp. antibodies in canine samples.
● Refrigerated or frozen samples must be at room temperature 15° to 27°C (59° to 80°F) before running the assay.
● DO NOT HEAT.
● Whole canine blood collected in any type of EDTA, heparin, or citrate tubes may be used within one day of collection, provided no visual clotting has occurred.
● Do not freeze whole blood or use whole blood that has been frozen.
● If whole blood is not used within two hours of draw, store refrigerated.
● Serum or plasma, either fresh or previously frozen, or stored at 2° to 8°C (35° to 46°F), may be used in this test.
● Serum or plasma may be stored for use up to 7 days 2° to 8°C (35° to 46°F). For longer storage, sample should be frozen at -20°C (-4°F) or colder.
● Previously frozen or older serum or plasma samples must be centrifuged at > 1600g to remove any particulate material before use.
● Excessive hemolysis may obscure the results.
● EDTA, heparin, or ACD in plasma will not affect the results.
Precautions And Warnings
VetScan Canine Anaplasma Antibody Test Kit Caution
● Do not remove Test Device from the pouch until ready for use.
● Test Device must be used as soon as possible after removing from pouch and within a maximum of 15 minutes.
● For veterinary use only.
● Do not use components after expiration date.
● The Test Device should be used in a horizontal position on a flat surface while the test is performed.
● The Test Device should not be moved or tilted during the test procedure.
● Use a separate Transfer Pipette for each test.
● The Chase Buffer is not interchangeable from serial (lot) to serial (lot).
● Do not use a Test Device from a pouch that is obviously torn or damaged.
● Do not use a Test Device if it appears cracked, broken, or otherwise damaged.
● The Kit Components must not be frozen.
● Contains Proclin™ 300 and sodium azide as preservatives.
STORAGE
● The Test Devices and Chase Buffer must be stored at 2° to 27°C (35° to 80°F) and never frozen.
● Test Devices and Chase Buffer are stable until expiration when stored at recommended temperatures.
KIT COMPONENTS
1. Test Devices
2. Chase Buffer Bottle
3. Transfer Pipettes
4. Instruction for Use
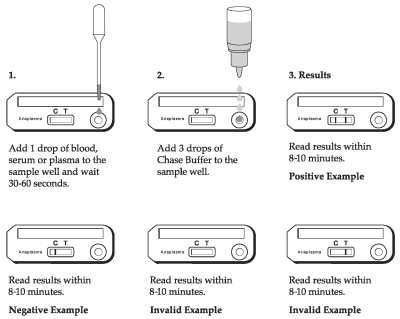
TEST PROCEDURE
1. Remove the Test Device from the protective pouch and place on a flat surface. Label the Test Device with the subject I.D. or control identification.
2. Gently mix the sample by inverting.
3. Using the Transfer Pipette provided, transfer one drop of sample (whole blood, serum or plasma) in to the sample well.
4. Let the sample absorb for 30-60 seconds.
5. Holding the Chase Buffer Bottle vertically, add 3 drops of the chase buffer into the sample well.
Read the results within 8-10 minutes. High positive results may appear as soon as 1 minute, and low positive results may take up to 8-10 minutes to appear. Do not read results after 15 minutes. Colored lines which appear after 15 minutes are not diagnostic and should be ignored.
INTERPRETATION OF TEST RESULTS
Positive Results
The test is positive if two colored lines appear. One colored line will appear at the Test line (T) area and other in the Control line (C) area. Any intensity of the Test line (T) should be considered positive. Colored lines may be lighter or darker than each other.
Negative Results
The result is negative if only one line appears at the Control line (C) area.
Invalid Results
The test is invalid if no colored line appears at the Control line (C) area even if a colored line appears at the Test line (T) area. Colored lines that appear after 15 minutes are not diagnostic and should be ignored.
ANAPLASMA TEST PROCEDURE
REFERENCES
1. Michael J. Day (2011) The Immunopathology of Canine Vector-Borne Diseases. Parasites & Vectors 2011, 4:48

For Technical Assistance Call: 800-822-2947
READ ALL INSTRUCTIONS BEFORE BEGINNING THE ASSAY
Manufacturer
SA Scientific, 4919 Golden Quail, San Antonio, TX 78240 USA
U.S. Vet. License No. 373
Distributed by: Abaxis, Inc., 3240 Whipple Road, Union City, CA 94587
+1 800 822 2947
www.abaxis.com
ABAXIS Europe GmbH, Bunsenstr. 9-11, 64347 Griesheim, Germany
+49 6155 780 210
For patent information, see www.abaxis.com/about_us/patents
510-7021-1 Rev. C
Presentation: 25 test kit.
CPN: 1740007.2
Distributed by ZOETIS INC.
333 PORTAGE STREET, KALAMAZOO, MI, 49007
| Telephone: | 269-359-4414 | |
| Customer Service: | 888-963-8471 | |
| Website: | www.zoetis.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-05-29
Elsewhere on our site
Qulipta
Qulipta is used to help prevent episodic or chronic migraine headaches in adults. Qulipta is an ...
Aimovig
Learn about Aimovig (erenumab-aooe) a once-monthly, injectable medication that can be ...
Dupixent
Dupixent is used to treat eczema, eosinophilic or oral-corticosteroid-dependent asthma, chronic ...
Ubrelvy
Ubrelvy (ubrogepant) tablets are used for the acute treatment of migraine. Includes Ubrelvy side ...
Nurtec ODT
Nurtec ODT (rimegepant) is used to treat acute migraines and prevent episodic migraines, by ...
Xeomin
Xeomin (incobotulinumtoxinA) is used to treat cervical dystonia, blepharospasm, upper facial lines ...
Dysport
Dysport (abobotulinumtoxinA) is used to treat cervical dystonia, glabellar lines and limb ...
Botox Cosmetic
Botox Cosmetic is a prescription treatment for fine lines and wrinkles. It temporarily improves the ...
