ThyroKare Equine Powder
This treatment applies to the following species: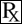 Company: Neogen
Company: Neogen
Levothyroxine Sodium USP (0.22%)
ThyroKare Equine Powder Caution
Federal law restricts this drug to use by or on the order of a licensed veterinarian.
FOR ANIMAL USE ONLY
KEEP OUT OF REACH OF CHILDREN
ThyroKare Equine Powder Indications
For use in horses for correction of conditions associated with low circulating thyroid hormone (hypothyroidism).
Dosage:
The suggested initial dose is 0.5 to 3.0 mg levothyroxine sodium (T-4) per 100 lbs of body weight (1 to 6 mg per 100 kg) once per day or in divided doses. Response to the administration of ThyroKare Equine Powder should be evaluated clinically until an adequate maintenance dose is established. In most horses, the total daily dose of T-4 is in the range of 6 to 36 mg. Serum T-3 and T-4 values can vary greatly among individual horses on thyroid supplementation. Dosages should be individualized and animals should be monitored daily for clinical signs of hyperthyroidism or hypersensitivity.
Administration:
ThyroKare Equine Powder can be administered by mixing the daily dose in the concentrate or by top dressing on the grain, preferably rolled or ground. To facilitate the proper adhesion of ThyroKare Equine Powder to the ration, slightly moisten the grain with water or a liquid supplement.
Active Ingredients
Each pound (453.6 g) contains:
|
Levothyroxine Sodium USP |
0.22% (1.0 g) |
|
One teaspoon contains: |
12 mg of T-4 |
|
One tablespoon contains: |
36 mg of T-4 |
In a palatable limestone base with vanilla flavoring.
Precaution:
Store at 20°-25°C (68°-77°F) with excursions between 15°-30°C (59°-86°F) and protect from light.
Warning
Administer with caution to animals with clinically significant heart disease, hypertension, or other complications for which a sharply increased metabolic rate might prove hazardous. Use in pregnant mares has not been evaluated.
NEOGEN® Vet
Manufactured by: Neogen Corporation, Lexington, KY 40511
859-254-1221 or 800-621-8829
animalsafety.neogen.com
Made in USA
|
Net Contents: |
NDC: |
Item No. |
|
|
1 lb (453.6 g) |
59051-9127-4 |
09127 |
L3382-0716 |
CPN: 1491013.6
944 NANDINO BLVD, LEXINGTON, KY, 40511-1205
| Telephone: | 859-254-1221 | |
| Order Desk: | 800-525-2022 | |
| Fax: | 859-255-5532 | |
| Website: | http://animalsafety.neogen.com | |
| Email: | inform@neogen.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-05-29
