Revalor-XH (Canada)
This treatment applies to the following species:Trenbolone acetate and Estradiol Implant
Trenbolone acetate 200 mg and Estradiol 20 mg
VETERINARY USE ONLY
FOR FEEDLOT HEIFERS
DIN 02469219
Description
Revalor®-XH is an extended-release implant containing 200 mg of trenbolone acetate and 20 mg estradiol. Each implant consists of 10 small yellow pellets, four uncoated and six coated with a polymer for extended release.
Each pellet contains:
Active Ingredients
20 mg trenbolone acetate and 2 mg estradiol.
Non-Medicinal Ingredients: 4.25 mg cholesterol, 0.5 mg magnesium stearate, 0.25 mg ethylcellulose N200, 1.69 mg poly-(DL-lactide-co-glycolide) 65:35, 0.08 mg colloidal silica anhydrous.
THERAPEUTIC CLASSIFICATION
Growth promotant.
Revalor-XH Indications
To increase rate of weight gain and improve feed efficiency for up to 200 days after implantation in feedlot heifers.
NOTE
The administration of Revalor-XH can result in decreased marbling compared to non-implanted heifers. Efficacy was confirmed in growth promotion trials conducted in heifers weighing 238 - 360 kg at the time of implantation.
Revalor-XH Dosage And Administration
Dosage Form:
One implant containing 200 mg trenbolone acetate and 20 mg estradiol is administered to each animal. The 10 pellets which make up the dosage of Revalor®-XH are contained in one division of the multiple dose cartridge. Each cartridge contains 10 doses. The cartridge is designed to be used with the Revalor® implant gun.
Administration
a) Site of Administration:
The implant is placed under the skin on the posterior aspect of the ear by means of the Revalor® implanter available from Intervet Canada Corp. After properly restraining the animal to allow access to the ear, cleanse the skin at the implant needle puncture site. Improperly cleansed skin may result in abscess formation and the subsequent loss of the implant. The implant is placed subcutaneously between the skin and cartilage on the back, middle third of the ear below the midline (see Figure 1). The implant must not be placed closer to the head than the outer edge of the cartilage ring. The location of needle insertion is at a point corresponding to the centre of the ear with the needle directed towards the head, and at least a needle length away from the intended deposition site (see Figure 2). Care should be taken to avoid injuring the major blood vessels or cartilage of the ear.
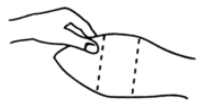
Figure 1 - Ear of Bovine Ready for Implantation
b) Method of Administration:
1. Do not remove the cap (C) of the cartridge containing the implants.
2. Place the cartridge (D) (with the capped end to the front) into slot at the top of the implanter magazine (labelled A on the diagram).
3. Gently push the cartridge into the slot until it clicks into place.
4. The implanter is now ready for use.
5. Take the ear of the animal firmly with the free hand (in the manner shown in Figure 1) and insert the needle into the subcutaneous tissue at the point indicated (Figure 2). Press the trigger (E on Figure 3).
6. The retraction bar will automatically withdraw the needle from the ear aiding in depositing the pellets of the implant in a single row.
7. The gun automatically advances the cartridge to the next implant.
8. When all the implants have been administered, the cartridge will fall out of the bottom of the magazine and may be replaced by a new one.
9. To change the needle, loosen the needle locking nut (labelled F in Figure 3) and replace the needle. Tighten the nut finger tight and the implanter is ready for use.
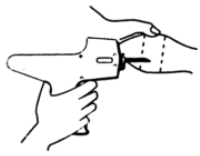
Figure 2 - Rear view of the bovine ear showing the site for insertion of the implanter needle.
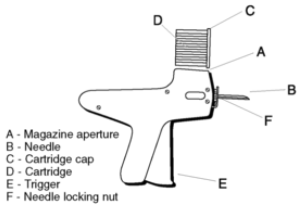
Figure 3 - Diagram of the implanter and cartridge
Revalor-XH Cautions
Do not use in animals intended for subsequent breeding.
Warnings
No withdrawal period in cattle is required when this product is used according to the label directions. Do not use in calves to be processed for veal. Do not use in dairy cattle. Implant pellets in the ear only. Do not attempt salvage of implant site for human or animal food. Keep out of reach of children.
Storage
Store the unopened product below 25°C in the original packaging. Do not freeze. Store the opened product cartridges in the foil pouch protected from light in a refrigerator (2-8°C). Do not use the opened product after 6 months.
HOW SUPPLIED
Box of 10 cartridges x 10 implants (100 implants).
Intervet Canada Corp., 16750, route Transcanadienne, Kirkland, QC H9H 4M7
1 866 683-7838
Intervet Canada Corp. is a subsidiary of Merck & Co., Inc.
® Registered trademark of Intervet International B.V.
Used under license.
Version: 06OCT2017
CPN: 1208294.0
Intervet Canada Corp.
16750 ROUTE TRANSCANADIENNE, KIRKLAND, QC, H9H 4M7
| Order Desk: | 514-428-7013 | |
| Toll-Free: | 866-683-7838 | |
| Fax: | Toll-free 888-498-4444; local 514-428-7014 | |
| Website: | www.merck-animal-health.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
