Reconcile (fluoxetine hydrochloride) Chewable Tablets (32 mg) (Canada)
This treatment applies to the following species: Company: Virbac
Company: Virbac
fluoxetine (as fluoxetine hydrochloride)
For Veterinary Use Only
Description
RECONCILE is a chewable, flavoured tablet that contains fluoxetine hydrochloride. RECONCILE chewable tablets are available in 8, 16, 32, and 64 mg tablet strengths for oral administration to dogs. The active ingredient in RECONCILE is fluoxetine hydrochloride, a selective serotonin reuptake inhibitor (SSRI).
Reconcile (fluoxetine hydrochloride) Chewable Tablets (32 mg) Indications
RECONCILE chewable tablets are indicated for the treatment of canine separation anxiety in conjunction with a behaviour modification plan.
Dosage and Administration
The recommended dose of RECONCILE is 1-2 mg/kg administered once daily, in conjunction with a behaviour modification plan. A typical behaviour modification plan consists of the pet owner implementing standard training techniques based on principles such as rewarding appropriate behaviour, coming and going in a manner that does not elicit inappropriate responses from the dog, and teaching the dog to be content while alone.Table 1: Recommended Dose of RECONCILE
|
Dog Weight |
No. of Tablets/day |
Tablet Strength (mg) |
|
|
(lb) |
(kg) |
||
|
8.8 - 17.6 |
4.0 - 8.0 |
1 |
8 |
|
17.7 - 35.2 |
8.1 - 16.0 |
1 |
16 |
|
35.3 - 70.4 |
16.1 - 32.0 |
1 |
32 |
|
70.5 - 140.8 |
32.1 - 64.0 |
1 |
64 |
The patient’s response to therapy should be monitored. If no improvement is noted within 8 weeks, case management should be reevaluated.
Professional judgment should be used in monitoring the patient’s response to therapy to determine the need to continue treatment with RECONCILE beyond 8 weeks. To discontinue therapy, it is not necessary to taper or reduce doses because of the long half-life of this product. Continued behavioural modification is recommended to prevent recurrence of the clinical signs.
RECONCILE is readily consumed by dogs or can be administered like other tablet medications, and can be given with or without food.
Professional discretion should be used in determining the need for dose reduction in the event of a possible adverse reaction. Approximately half of patients tolerate a return to the previous dose after 1-2 weeks on a reduced schedule (see ADVERSE REACTIONS).
If a dose is missed, the next scheduled dose should be administered as prescribed. Do not increase or double the dose.
Contraindications
RECONCILE is contraindicated for use in dogs with epilepsy or a history of seizures. RECONCILE should not be given concomitantly with drugs that lower the seizure threshold (e.g., phenothiazines such as acepromazine or chlorpromazine).
RECONCILE should not be given in combination with a monoamine oxidase inhibitor (MAOI) [e.g., selegiline hydrochloride (L-deprenyl) or amitraz], or within a minimum of 14 days of discontinuing therapy with an MAOI.
RECONCILE is contraindicated in dogs with a known hypersensitivity to fluoxetine HCl or other SSRIs.
Because fluoxetine and its major metabolite, norfluoxetine, have long half-lives, a 6-week washout interval should be observed following discontinuation of therapy with RECONCILE prior to the administration of any drug that may adversely interact with fluoxetine or norfluoxetine.
Cautions: RECONCILE is not recommended for the treatment of aggression. RECONCILE has not been clinically tested for the treatment of other behavioural disorders. Studies to determine the effects of RECONCILE in breeding, pregnant, or lactating dogs and in patients less than 6 months of age have not been conducted. Seizures may occur in dogs treated with RECONCILE, even in dogs without a history of epilepsy or seizures (see ADVERSE REACTIONS). Before prescribing RECONCILE, a comprehensive physical examination should be conducted to rule out causes of inappropriate behaviour unrelated to separation anxiety. The examination should include a thorough history and assessment of the patient’s household environment and standard practice laboratory tests as appropriate for the patient’s age and health status. Veterinarians should be familiar with the risks and benefits of the treatment of behavioural disorders in dogs before initiating therapy. Inappropriate use of RECONCILE, i.e. in the absence of a diagnosis or without concurrent behaviour modification, may expose the animal to unnecessary adverse reactions and may not provide any lasting benefit of therapy.
RECONCILE has not been evaluated with drugs that affect the cytochrome P450 enzyme system. RECONCILE should be used with caution when co-administered with any drug that affects the cytochrome P450 enzyme system (for example, ketoconazole). Studies to assess the interaction of RECONCILE with tricyclic antidepressants (TCAs) (for example, amitriptyline and clomipramine) have not been conducted. The minimum washout period to transition dogs from TCAs to RECONCILE has not been evaluated. Published pharmacokinetic data demonstrates that TCAs are cleared 4 days following discontinuation.1,2
1 Plumb DC. Amitriptyline. Veterinary Drug Handbook 5th Edition (Pocket Edition). Iowa State Press. Ames, IA. Page 39, 2002.
2 Hewson CJ, et al. The pharmacokinetics of clomipramine and desmethylclomipramine in dogs: parameter estimates following a single oral dose and 28 consecutive daily doses of clomipramine. J Vet Pharmacol Therap 21:214-222, 1998.
Warnings
Not for use in humans. Keep out of reach of children. In case of accidental ingestion seek medical attention immediately. In humans, the most common symptoms associated with over dosage include seizures, somnolence, nausea, tachycardia, and vomiting. In case of ingestion by a human, contact a physician immediately.Adverse Reactions: Post Market Experience: The following adverse events are based on post-approval adverse drug experience reporting with RECONCILE. Not all adverse reactions are reported. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using this data. The following categories of adverse reactions are listed in decreasing order of frequency by body system:
Systemic disorders: Decreased appetite, lethargy, weight loss
Behavioural disorders: Anxiety, restlessness, vocalization, disorientation, aggression
Digestive tract disorders: Vomiting, diarrhea, hypersalivation
Neurological disorders: Depression, shivering, muscle tremor, seizure, mydriasis, incoordination
Respiratory tract disorders: Panting
In two North American multi-site field studies, which included a total of 427 dogs, the following adverse reactions were observed:
Seizures: In one study, with behaviour modification, one of 112 dogs in the control group and three of 117 dogs that received RECONCILE experienced the serious adverse reaction of seizures. One of the three dogs treated with RECONCILE experienced two seizures 10 days after the end of therapy. Despite escalating phenobarbital doses, the seizures continued and this dog died in status epilepticus approximately six months after the first seizure. Another of the three dogs treated with RECONCILE had experienced one seizure approximately 1 1/2 years prior to study enrollment immediately after receiving head trauma. No additional seizures were reported to have occurred until 45 days after concluding treatment with RECONCILE. During the 1 1/2-year period since the second seizure, this dog’s seizure activity increased from single seizures to cluster seizures despite increasing doses of phenobarbital and the addition of oral potassium bromide and rectal diazepam. The third dog treated with RECONCILE and the control dog experienced one seizure 24 days and 35 days, respectively, after the start of therapy; no anticonvulsant therapy was initiated and no further seizures were reported in either dog.
In the second study, without behaviour modification, one of 99 dogs treated with RECONCILE and one of 99 dogs treated with the control tablet experienced the serious adverse reaction of seizures 9 and 27 days, respectively, after initiation of therapy. The dog treated with RECONCILE was subsequently diagnosed with vestibular disease and the control dog had a history of recurrent hind leg weakness.
In a European multi-site study, 234 dogs were treated with daily doses of fluoxetine chewable tablets ranging from 0.25 mg/kg to 4 mg/kg. One dog treated with a daily dose of 0.4 mg/kg for one month experienced one seizure one week after discontinuing therapy. No anticonvulsant therapy was initiated and no further seizures were reported.
Weight loss: Of the dogs in the two North American field studies with body weight measurements throughout the study (n=196 and n=185 in the RECONCILE and control group, respectively), a 5% or greater weight loss (when compared to initial, pre-study body weight) was observed in 58 (29.6%) of dogs treated with RECONCILE and 24 (13.0%) of dogs in the control group. No dogs were withdrawn from clinical studies due to weight loss alone. The following table shows the number of dogs with weight loss, stratified by percent weight loss relative to initial body weight.
Table 2: Dogs with Weight Loss (stratified by percent loss relative to initial body weight)
|
Treatment Group |
≥ 5% to < 10% |
≥ 10 to < 15% |
≥ 15% |
|
RECONCILE chewable tablets |
44 (22.5%) |
13 (6.6%) |
1a (0.5%) |
|
Control |
20 (10.8%) |
4 (2.2%) |
0 (0%) |
aThis dog lost 20% of its initial body weight and was the same dog that died in status epilepticus.
Other adverse reactions:
Additional adverse reactions observed in dogs treated with RECONCILE at a rate of 1% or greater were:
Table 3: Adverse Reactions Reported in the North American Field Studies
|
Adverse Reaction |
RECONCILE Chewable Tablets, N=216 |
Control,* N=211 |
||
|
n |
% |
n |
% |
|
|
Calm/Lethargy/Depression |
71 |
32.9 |
22 |
10.4 |
|
Decreased Appetite |
58 |
26.9 |
13 |
6.2 |
|
Vomiting |
37 |
17.1 |
28 |
13.3 |
|
Shaking/Shivering/Tremor |
24 |
11.1 |
4 |
1.9 |
|
Diarrhea |
21 |
9.7 |
17 |
8.1 |
|
Restlessness |
16 |
7.4 |
8 |
3.8 |
|
Excessive Vocalization (Including Whining) |
13 |
6.0 |
7 |
3.3 |
|
Aggression |
9 |
4.2 |
13 |
6.2 |
|
Otitis Externa |
6 |
2.8 |
2 |
0.9 |
|
Disorientation |
5 |
2.3 |
1 |
0.5 |
|
Incoordination |
5 |
2.3 |
0 |
0.0 |
|
Constipation |
3 |
1.4 |
0 |
0.0 |
|
Excessive Salivation |
3 |
1.4 |
4 |
1.9 |
* The control group received the tablet formulation without fluoxetine.
Dose Reduction:
Twenty dogs in the RECONCILE group and five dogs in the control group required a reduction in dose due to unacceptable adverse reactions, generally anorexia, vomiting, shaking and depression. Lowering the dose eliminated or reduced the severity of these adverse reactions in the RECONCILE group only. Resumption of the full dose of RECONCILE resulted in a return of the initial adverse reactions in approximately half of the affected dogs. The majority of these adverse reactions were intermittent and mild. However, one dog experienced recurrence of severe adverse reactions, which necessitated withdrawal from the study for that dog. Additionally, two dogs required a second dose reduction of RECONCILE. Effectiveness was maintained in a majority of those dogs in which a dose reduction was necessary.
Clinical Pharmacology
The molecular weight of fluoxetine is 345.79. The structural formula is depicted below.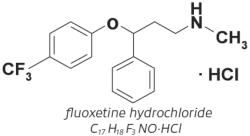
Fluoxetine exerts its effect by inhibiting the reuptake of serotonin at the pre-synaptic neuron. Fluoxetine does not act as a sedative. Fluoxetine is well absorbed after oral administration (~72%). It is largely metabolized in the liver by cytochrome P450 enzyme system to norfluoxetine, an equipotent SSRI that contributes to the efficacy of RECONCILE.
After a single dose, and also at steady state, calculations were made as follows:
Table 4: Single Dose* Pharmacokinetic Parameters of Fluoxetine Hydrochloride (mean ± standard error).
|
|
AUC0-∞ (µg•hr/mL) |
Cmax (ng/mL) |
Tmax (hr) |
T1/2 (hr) |
T1/2 Range (hr) |
|
Fluoxetine |
1.388 (±0.137) |
126.6 (±12.3) |
1.8 (±0.2) |
6.2 (±0.8) |
3.0-12.9 |
|
Norfluoxetine |
11.44 (±0.74) |
138.3 (±9.6) |
12.8 (±1.7) |
49 (±3) |
33.0-64.0 |
* approximately 2 mg/kg body weight
In a 21-day study, fluoxetine was administered daily at a dose of 0.75, 1.5 and 3.0 mg/kg to laboratory Beagles. The maximum plasma concentration (Cmax) and area under the plasma concentration time curve (AUC) for fluoxetine were approximately dose proportional between 0.75 and 1.5 mg/kg, with a greater than dose proportional increase at 3 mg/kg.
Norfluoxetine Cmax and AUC were generally dose proportional.
Although steady state appeared to be reached within 10 days in the 21-day study, a continuous increase in trough concentrations was observed in a one year, multiple-dose laboratory safety study. In this study, dogs administered a 1 mg/kg dose of fluoxetine had plasma fluoxetine concentrations that continued to increase over the one-year dosing period. A similar increase in concentrations was observed with norfluoxetine.
This phenomenon was not observed at higher doses. During the one-year dosing interval and the subsequent two-month recovery period, there were no changes in the nature and frequency of adverse reactions observed as compared to those seen by Day 28 of fluoxetine administration.
Effectiveness
In one randomized multi-centered, double-blinded, vehicle-controlled study of 8 weeks duration, 229 dogs were evaluated at 34 investigative sites in the United States and Canada. One hundred seventeen dogs were randomized to 1-2 mg/kg/day of RECONCILE and 112 dogs were randomized to the control group. Both groups underwent concurrent behaviour modification. In seven of the eight weeks, the percentage of dogs with improved overall separation anxiety scores was significantly higher (p < 0.05) among dogs treated with RECONCILE compared to dogs that received the control tablet. At the end of the study, 73% of dogs treated with RECONCILE showed significant improvement (p=0.010) as compared to 51% of dogs treated with behaviour modification alone.Dogs treated with RECONCILE also showed improvement in destructive behaviour, excessive vocalization, and restlessness over dogs that received the control tablet. In addition, dogs in both groups experienced improvement in inappropriate urination, inappropriate defecation, excessive salivation, excessive licking/grooming, shaking/shivering and depression.
Overall separation anxiety severity scores improved more rapidly for dogs taking RECONCILE than those dogs receiving the control tablet. The same effect was also noted for the individual scores for excessive vocalization and depression. Crating dogs did not affect the results.
The effectiveness and safety of RECONCILE was demonstrated in a field study in client-owned dogs (see EFFECTIVENESS and ADVERSE REACTIONS). At the end of the 8-week study, 73% of dogs treated with RECONCILE showed significant improvement (p=0.010), as compared to behaviour modification alone (51%). During the course of therapy, 42% of dogs showed improvement within the first week, which was significantly greater (p=0.005) than with behaviour modification alone (18%).
Animal Safety: In a one-year laboratory safety study, dogs were dosed daily at 1, 4.5, and 20 mg/kg/day of a gelatin capsule filled with fluoxetine powder. Based upon the results of a relative bioavailability study comparing the fluoxetine-filled capsule versus the RECONCILE chewable tablets, the corresponding equivalent doses were 0.87, 3.9 and 17.4 mg/kg/day of RECONCILE chewable tablets (where the average ratio of fluoxetine AUC values for RECONCILE chewable tablets/fluoxetine-filled capsule = 1.15). Three of five female dogs in the 20 mg/kg group died or were euthanatized during the first six months of the study. The high dose was decreased to 10 mg/kg/day (equivalent to 8.7 mg/kg/day of RECONCILE) for the last six months of the treatment, and all remaining dogs completed the study. One dog in the 1 mg/kg group (equivalent to 0.87 mg/kg/day of RECONCILE) and two dogs in the 20 mg/kg group (equivalent to 17.4 mg/kg/day of RECONCILE) experienced a seizure. Aggressive behaviour, ataxia, salivation at dosing, hyperesthesia, nystagmus, thin body condition, weakness, lethargy, diarrhea and head tilt were also noted in the high dose group. Anorexia, tremors, decreased pupillary light response, mydriasis, vomiting, and decreased weight gain were observed in all treatment groups, but occurred more frequently in the high dose group. With the exception of decreased weight gain, all abnormal observations resolved by the end of a two-month recovery period. Evidence of phospholipidosis was noted in the lung, liver, adrenal glands, lymph nodes, spleen, retina and white blood cells of all groups, which resolved during the recovery period. Fluoxetine caused no marked or consistent effects on hematology, blood chemistries or urinalysis. Bradycardia was absent on the electrocardiogram in the control and lowest dose groups, but was mildly present in a dose-dependent manner in the two higher dose groups. There were no effects noted on gross organ examination.
The effectiveness and clinical safety of RECONCILE for long-term use (i.e. for more than 8 weeks) has not been evaluated. RECONCILE was evaluated at the recommended label dose for one year in a laboratory safety study in dogs (see ANIMAL SAFETY).
Storage Information: Store at 20-25°C. Excursions permitted between 15-30°C. Do not remove desiccant canister from the bottle.
Completely close bottle between uses.
How Supplied
RECONCILE is available in four tablet sizes: 8, 16, 32, and 64 mg, 30 tablets per bottle.10460 DIN 02307952
10461 DIN 02307960
10462 DIN 02307979
10463 DIN 02307987
Pegasus Laboratories, Inc., 8809 Ely Road, Pensacola, FL USA 32514
Imported and distributed by: Virbac Canada, Inc., 231-209 Shearson Crescent, Cambridge, ON, N1T 1J5
ca.virbac.com
® Pegasus Laboratories, Inc.
To report an adverse event, call Pegasus Laboratories at 1-800-874-9764.
CPN: 1177098.1
209-231 SHEARSON CRESCENT, CAMBRIDGE, ON, N1T 1J5
| Toll-Free: | 866-458-3350 | |
| Fax: | 844-458-4004 | |
| Website: | https://ca.virbac.com/ |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
