Ralgro Implants for Beef Cattle
This treatment applies to the following species:(zeranol implants)
IMPLANTS IN CARTRIDGES for Beef Cattle
RALGRO® (zeranol implants) provide 36 mg of zeranol per implant (three 12-mg pellets per implant).
INDICATIONS FOR USE: For increased rate of weight gain and improved feed efficiency in growing beef steers and heifers fed in confinement for slaughter. For increased rate of weight gain in beef calves 2 months of age or older, and in growing beef steers and heifers on pasture (stocker, feeder, and slaughter), and in growing beef steers and heifers in a dry lot. Not approved for repeated implantation (reimplantation) with this or any other cattle ear implant within a single production phase as safety and effectiveness have not been evaluated.
Do not use in beef calves less than 2 months of age, dairy calves, and veal calves because effectiveness and safety have not been evaluated.
Do not use in replacement beef heifers after weaning or in bulls, dairy cows or replacement dairy heifers.
 |
WITHDRAWAL PERIODS AND RESIDUE WARNINGS No withdrawal period is required when used according to labeling. Do not use in beef calves less than 2 months of age, dairy calves, and veal calves. A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in replacement beef heifers after weaning or in dairy cows or replacement dairy heifers. Use in these cattle may cause drug residues in milk and/or calves born to these cows. Implant pellets subcutaneously in ear only. Any other location is a violation of Federal law. Do not attempt salvage of implanted site for human or animal food. |
 |
Manufactured by a non-sterilizing process.
Restricted Drug (California), Use Only as Directed.
USER SAFETY WARNINGS: Not for use in humans. Keep out of reach of children.
ANIMAL SAFETY WARNINGS: Edema of the vulva and udder, teat elongation, rectal and vaginal prolapse, and signs of estrus may occur when heifers are implanted. Delayed testicular development may occur in young males. To avoid difficulty in castration, young males should be castrated at the time of implanting.
Directions For Use
A single pull of the RALOGUN® Pellet Injector trigger delivers one dose (36-mg provided as three 12-mg pellets per chamber).
STEP 1

Each chamber of the plastic RALGRO cartridge contains a full dose of RALGRO implants. Insert the cartridge in the magazine of the RALOGUN pellet injector with the hole in the axle of the cartridge pointed toward the handle of the RALOGUN pellet injector. After snapping into place, rotate cartridge clockwise to ensure proper seating, then check window at top to ensure that pellet chamber is aligned with plunger.
STEP 2

Always use sharp needle. A dull needle tears tissue and makes proper implanting difficult. Be sure that needle is fully seated in RALOGUN pellet injector. This ensures that pellets will be expelled from the needle as RALOGUN plunger is extended. Failure to set the needle could cause the last pellet to be left in the needle and not implanted.
STEP 3
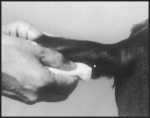
After appropriately restraining the animal to allow access to the ear, cleanse the skin at the implant needle puncture site.
STEP 4

The implant site is subcutaneous, between the skin and cartilage on the back side of the ear and below the midline of the ear. The implant must not be placed closer to the head than the edge of the auricular cartilage ring farthest from the head. The location for insertion of the needle is a point toward the tip of the ear and at least a needle length away from the intended deposition site. Care should be taken to avoid injuring the major blood vessels or cartilage of the ear. Squeeze trigger of the RALOGUN pellet injector to deliver a full dose of RALGRO. Keep trigger depressed while withdrawing the needle to be sure that RALGRO implants stay in place.
STEP 5

Wipe needle with cotton or gauze moistened with alcohol or other suitable disinfectant. Do not dip needle in solution because solution clinging to inside of needle will cause plugging of needle.
STEP 6

Rotate cartridge in direction of arrow until a new dose is visible and centered in window. The RALOGUN pellet injector is now ready for next implantation.
Storage
Store between 2° and 25° C (36° and 77° F).QUESTIONS/COMMENTS? For a copy of the Safety Data Sheet or to report side effects, contact Merck Animal Health Technical Services at 1-800-211-3573. For additional information about reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae
Made in Austria
This product may not be marketed or used in the EU.
Approved by FDA under NADA # 038-233
Distributed by: Intervet Inc d/b/a Merck Animal Health, Madison, NJ 07940
Copyright © 2022 Merck & Co., Inc., Rahway, NJ, USA and its affiliates.
All rights reserved.
Rev. 03/2023
167206 R2
|
|
NDC |
|
|
1 cartridge of 24 doses |
0061-5026-02 |
167171 R3 |
|
Ten 24-dose cartridges. |
0061-5026-03 |
611084 R2 |
CPN: 1047168.6
Intervet Inc.
126 E. LINCOLN AVENUE, PO BOX 2000, Rahway, NJ, 07065
| Customer Service: | 800-521-5767 | |
| Technical Service (Companion Animal): | 800-224-5318 | |
| Technical Service (Livestock): | 800-211-3573 | |
| Website: | www.merck-animal-health-usa.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
