Quest Plus Gel (Canada)
This treatment applies to the following species:moxidectin/praziquantel
VETERINARY USE ONLY
DIN 02276763
Description
QUEST PLUS® Gel contains moxidectin and praziquantel as its active ingredients. Moxidectin is a second generation endectocide of the milbemycin class of compounds that is highly effective, when administered at the recommended dosage of 0.4 mg per kg of body weight, against a broad range of economically important endoparasites of horses. Praziquantel is highly effective, when administered at the recommended dose of 2.5 mg/kg of body weight, against tapeworms.
Mode of action studies indicate that moxidectin eliminates parasites by interfering with chloride channel-mediated neurotransmission in the parasite, resulting in paralysis and elimination. The structural characteristics of the mammalian nervous system insure the safe use of moxidectin in horses and ponies. Praziquantel increases the tapeworm’s membrane permeability to calcium and other cations to cause severe contraction and paralysis of a tapeworm’s muscles. This spastic paralysis results in the inability of tapeworms to attach to the host’s intestinal wall. Detached tapeworms are either destroyed by the host’s immune system or passed out from the host in the feces.
Active Ingredients
QUEST PLUS Gel has been formulated in a highly palatable gel base for ease of administration.
Each mL contains 20 mg of moxidectin (2% w/v) and 125 mg of praziquantel (12.5% w/v).
Preservative
benzyl alcohol (22.59% w/v)
Quest Plus Gel Indications
QUEST PLUS Gel is indicated for the treatment of internal parasitic infections caused by the following parasites in healthy horses and ponies, 4 months of age or older:
Large Strongyles
Strongylus vulgaris (adults)
S. edentatus (adults and visceral, migratory stages)
Triodontophorus brevicauda (adults)
T. serratus (adults)
QUEST PLUS Gel is also indicated as an aid in the treatment of parasitic infections caused by the arterial stages of S. vulgaris.
Small Strongyles
Adult and luminal, larval (L4) stages of Cyathostomum spp., including Cyathostomum catinatum and C. pateratum;
Adult and luminal, larval (L4) stages of Cylicocyclus spp., including Cylicocyclus brevicapsulatus, C. elongatus, C. insigne, C. leptostomum, C. nassatus and C. radiatus;
Adult and luminal, larval (L4) stages of Cylicostephanus spp., including Cylicostephanus calicatus, C. goldi, C. longibursatus and C. minutus;
Adult and luminal, larval (L4) stages of Cylicodontophorus spp.;
Adult and luminal, larval (L4) stages of Gyalocephalus capitatus;
Adult stage of Coronocyclus spp., including Coronocyclus coronatus, C. labiatus and C. labratus.
Adult stage of Petrovinema poculatus;
Adult stage of Poteriostomum imparidentatum;
QUEST PLUS Gel is also indicated as an aid in the treatment of parasitic infections caused by the developing, encysted mucosal larvae (LL3 and L4) of cyathostomes. It is expected that the administration of QUEST PLUS Gel as per label directions can eliminate approximately 80% of the developing, encysted cyathostomes.
Ascarids (adults And L4)
Parascaris equorum
Pinworms (adults And L4)
Oxyuris equi
Hairworms (adults)
Trichostrongylus axei
Stomach Bots (2nd And 3rd Instars)
Gasterophilus nasalis
G. intestinalis
Integumentary Microfilariae
Onchocerca cervicalis
Tapeworms (adults)
Anoplocephala perfoliata
Large-mouthed Stomach Worms (adults And Gastric L4)
Habronema muscae
QUEST PLUS Gel, when used at the recommended dose, suppresses strongyle egg production for up to 84 days following a single administration, thereby reducing pasture contamination and providing a period of protection from reinfection for equines grazing that pasture.
Quest Plus Gel Dosage And Administration
QUEST PLUS Gel is specifically formulated as a palatable gel for administration which is easily administered to horses and ponies.
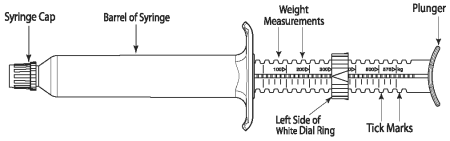
QUEST PLUS Gel is packaged in ready-to-use SURE-DIAL® syringes (see diagram of the SURE-DIAL syringe below). The syringe is calibrated in 25 kg increments, up to 575 kg. This enables administration of the recommended dose level of 0.4 mg moxidectin and 2.5 mg praziquantel/kg body weight by choosing a setting consistent with the animal’s weight.
How to set the dose
Since the dose is based on the weight of the animal, the administrator needs to use a scale or weight tape to find each animal’s weight before treating with QUEST PLUS Gel. Once the weight is known, set the dose of each horse or pony as follows.
1. Hold the syringe with the capped end pointing to the left so that you can see the weight measurements and tick marks (small black lines) as shown in the diagram below. Each tick mark relates to 25 kg body weight.
2. Turn the white dial ring until the left side of the ring lines up with the weight of the animal. In the diagram below the dial ring is set to dose a 300 kg animal.
How to give QUEST PLUS Gel to a horse or pony
1. Make sure there is no feed in the animal’s mouth.
2. Remove the cap from the end of the syringe. Save the cap for reuse.
3. Place the tip of the syringe inside the animal’s mouth at the space between the teeth.
4. Gently push the plunger until it stops, depositing the gel on the back of the tongue.
5. Remove the syringe from the animal’s mouth and raise the animal’s head slightly to make sure it swallows the gel.
6. Replace the syringe cap.
SURE-DIAL Syringe
Treating a second horse or pony with the same syringe
If the animal you treat weighs less than 575 kg, there will be gel left in the syringe. The administrator can use this gel to treat other horses or ponies. To set the next dose, add the weight of the animal to be treated to the dose setting already on the syringe. For example, if the syringe was fi rst used to treat a 100 kg animal, the white dial ring is set on 100 kg. To treat a 250 kg animal next, move the white dial ring to the 350 kg marking (100+250=350). More than one syringe are needed to treat horses weighing more than 575 Kg (1260 lb).
Each syringe of QUEST PLUS Gel may be used to treat more than one animal especially when dosing foals, ponies and growing and lighter breeds of horses. The table below will help to estimate the number of horses or ponies the contents of each syringe will treat.
|
Age |
Ponies |
Light Horses |
Heavy Horses |
|||
|
Approximate Weight |
No. ponies treated per syringe |
Approximate Weight |
No. horses treated per syringe |
Approximate Weight |
No. horses treated per syringe |
|
|
4 months |
75 (165) |
7 |
125 (275) |
4 |
175 (385) |
3 |
|
8 months |
100 (220) |
5 |
200 (440) |
2 |
250 (550) |
2 |
|
Mature |
200 (440) |
2 |
400 (880) |
1 |
500+ (1100+) |
1 |
If the animals are likely to be reinfected, a strategic parasite control program should be established: see Parasite Control Program below.
Parasite control program
For the best control of parasites, all horses and ponies should be included in a strategic treatment program, with particular care given to high performance animals, brood mares, stallions and foals. In foals, initial treatment is recommended at 4 months of age, after which they should be included in a routine retreatment program. Since QUEST PLUS Gel provides an aid in the control of developing, encysted cyathostomes (small strongyles), it may be particularly useful in reducing the frequency of dosing in a strategic treatment program. QUEST PLUS Gel, when used at the recommended dose, suppresses strongyle egg production for up to 84 days following a single administration, thereby reducing pasture contamination and providing a period of protection from reinfection for equines grazing that pasture. Consult your veterinarian for the diagnosis, prevention and treatment of parasitic infections in horses and ponies.
CAUTIONS
1. Extra care should be taken with foals to ensure that the correct dose is administered. Do not use in foals less than 4 months of age or in sick, debilitated and underweight horses.
2. QUEST PLUS Gel has been formulated specifically for use in horses and ponies only. This product should not be used in other animal species as serious adverse reactions may result, such as death in dogs.
3. Overdoses, should they occur, may manifest themselves as transient depression, drowsiness and ataxia 8 to 24 hours post administration. Supportive treatment is not generally necessary and recovery is usually complete within 24 to 72 hours. There is no specific antidote for overdosing with moxidectin.
4. Do not mix this product with other veterinary medicinal preparations before administration.
5. Dispose of any containers and any residual content safely (e.g., bury or incinerate). Bury away from water courses, as free moxidectin may adversely affect fish and certain water borne organisms.
6. Some horses have experienced reactions with swelling and itching shortly after treatment. In most such cases, the horses have been diagnosed as carrying heavy infections of Onchocerca microfilariae, and it is assumed the reactions were the result of microfilariae dying in large numbers. Although the signs have resolved spontaneously in a few days, symptomatic treatment may be advisable. Consult your veterinarian should these symptoms persist.
7. QUEST PLUS Gel has not been tested in breeding stallions.
WARNINGS
1. This drug is not to be administered to horses and ponies that are to be slaughtered for use in food.
2. Avoid oral exposure and direct contact with skin and eyes. Wash hands after handling. If accidental contact with eyes occurs, flush repeatedly with water. If irritation or any other symptom attributable to exposure to this product persists, consult a physician.
3. Keep out of reach of children.
Adverse Reactions
Although all adverse reactions are not reported, the following information is based on voluntary post-approval drug experience reporting. It is generally recognized that this results in significant under-reporting. The adverse events listed here reflect reporting and not necessarily causality. Adverse events are listed by body system, in decreasing order of frequency:
- Digestive tract disorders: colic, diarrhea,
- Systemic disorders: lethargy, anorexia,
- Immune system disorders: urticaria, allergic edema, hypersensitivity reaction,
- Neurologic disorders: ataxia.
Animal Safety
QUEST PLUS Gel can be safely administered at the recommended dose of 0.4 mg moxidectin and 2.5 mg praziquantel per kg of body weight to horses and ponies of all breeds, 4 months of age or older. Transient depression and/or ataxia has been observed occasionally in a small number of animals at three times the recommended dose level; however, it has been demonstrated that these effects diminish over time with minimal or no supportive therapy and result in no permanent cellular damage in the animal. The product has not been fully tested in young horses less than four months of age where multiple overdoses may result in reactions consistent with the known neurological mode of action for this class of compounds.
QUEST PLUS Gel has been tested in breeding, pregnant, and lactating mares. In a reproductive safety study, 10 mares received QUEST PLUS Gel at three times the recommended dosage, and 10 mares received tap water (control). Mares were dosed at least once a month from breeding through 30 days post-foaling, with increased frequency (every 2 weeks) around breeding. Nine out of 10 QUEST PLUS Gel-treated mares became pregnant, and gave birth to live foals. Ten control mares became pregnant, and 8 out of the 10 mares gave birth to live foals. One QUEST PLUS Gel-treated mare had episodes of depression, decreased appetite and diarrhea shortly after receiving 5 of the 17 (29%) doses. The same mare had one episode of colic shortly after one administration of QUEST PLUS Gel. This mare’s colic, depression, decreased appetite, and diarrhea all resolved without treatment. The foal born from this mare was observed shortly after birth with reduced menace and repetitive tongue rolling which resolved within a few days and the foal was normal at Day 30 post-foaling. In addition, a mare in QUEST PLUS Gel-treated group was reported with anxiety, nervousness accompanied with diarrhea throughout the trial when left alone. She gave birth to a foal which had a mild depression, chronic diarrhea and died from infectious colitis at Day 8 of age. Another QUEST PLUS Gel-treated mare had an abnormal reproductive cycle (prolonged time to ovulation) following foal heat (at least 39 days). This mare became pregnant on the first breeding and gave birth to a female foal that was normal at birth; however, the foal developed an enlarged clitoris by Day 30 post-foaling. One more observation in foals from QUEST PLUS Gel-treated group was a case of angular limb deformities (carpal valgus). This condition was resolved within a few days and the foal was normal at Day 30 post-foaling.
Storage
Store between 15 and 30°C. Keep from freezing.
Environmental Safety
Care should be taken to avoid release of significant volumes of moxidectin into either ground water or free-running water. Free moxidectin may adversely affect fish and certain water borne organisms. Moxidectin is readily and tightly bound to soil and rendered inactive over time. QUEST PLUS Gel syringes and their residual contents should be disposed of in an approved manner.
Presentation
QUEST PLUS Gel, is supplied as an individual, ready-to-use syringe which contains sufficient product to treat a 575 kg (1250+ lb) animal at the recommended dose of 0.4 mg moxidectin and 2.5 mg praziquantel per kg of body weight.
Zoetis is a trademark and Quest Plus and SURE-DIAL are registered trademarks of Zoetis or its licensors, used under license by Zoetis Canada Inc.
Zoetis Canada Inc., Kirkland QC H9H 4M7
2698-11-2
40026525
CPN: 1198432.3
16,740 TRANS-CANADA HIGHWAY, KIRKLAND, QC, H9H 4M7
| Order Desk: | 800-663-8888 | |
| Technical Services Canada: | 800-461-0917 | |
| Technical Services USA: | 800-366-5288 | |
| Website: | www.zoetis.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
