PrevEquine Tablets for Horses (Canada)
This treatment applies to the following species: Company: Boehringer Ingelheim Animal Health
Company: Boehringer Ingelheim Animal Health
(firocoxib) Tablets for Horses
DIN 02501228
VETERINARY USE ONLY
Description
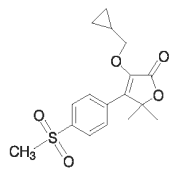
PREVEQUINE™ (firocoxib) tablets are round, beige to tan, double-scored tablets containing 57 mg of firocoxib. Firocoxib belongs to the coxib class of non-narcotic, non-steroidal anti-inflammatory drugs (NSAIDs). Firocoxib is a white crystalline compound described chemically as 3 (cyclopropylmethoxy)-4-(4-methylsulfonyl)phenyl)-5,5-dimethylfuranone. The empirical formula is C17H20O5S, and the molecular weight is 336.4 g/mol. The structural formula is shown below:
PrevEquine Tablets for Horses Indications
PREVEQUINE™ tablets are indicated for the control of pain and inflammation associated with osteoarthritis in horses.
Dosage and Administration
(Always provide the Client Information Sheet with the prescription). The recommended dosage of PREVEQUINE™ tablets is one 57 mg tablet administered orally to horses weighing 360 - 600 kg, once daily for up to 14 days. For ease of administration, PREVEQUINE™ tablets may be given with food.
Contraindications
Horses with hypersensitivity to firocoxib should not receive PREVEQUINE™ tablets.
Cautions:
Horses should undergo a thorough history and physical examination before initiation of NSAID therapy. Appropriate laboratory tests should be conducted to establish hematological and serum biochemical baseline data before and periodically during administration of any NSAID. Clients should be advised to observe for signs of potential drug toxicity and should be given a Client Information Sheet with each prescription. See information for Owner or Person Treating Horse section of the package insert.
Treatment with PREVEQUINE™ tablets should be terminated if signs such as inappetence, colic, abnormal feces, or lethargy are observed.
As a class, cyclooxygenase inhibitory NSAIDs may be associated with gastrointestinal, renal, and hepatic toxicity. Sensitivity to drug-associated adverse events varies with the individual patient. Horses that have experienced adverse reactions from one NSAID may experience adverse reactions from another NSAID. Patients at greatest risk for adverse events are those that are dehydrated, on diuretic therapy, or those with existing renal, cardiovascular, and/or hepatic dysfunction. Concurrent administration of potentially nephrotoxic drugs should be carefully approached or avoided. NSAIDs may inhibit the prostaglandins that maintain normal homeostatic function. Such anti-prostaglandin effects may result in clinically significant disease in patients with underlying or pre-existing disease that has not been previously diagnosed. Since many NSAIDs possess the potential to produce gastrointestinal ulcerations and/or gastrointestinal perforation, concomitant use of PREVEQUINE™ tablets with other anti-inflammatory drugs, such as NSAIDs or corticosteroids, should be avoided. Consider appropriate washout times when switching from one NSAID to another NSAID or corticosteroid.
The concomitant use of protein bound drugs with PREVEQUINE™ tablets has not been studied in horses. The influence of concomitant drugs that may inhibit the metabolism of PREVEQUINE™ tablets has not been evaluated. Drug compatibility should be monitored in patients requiring adjunctive therapy.
The safe use of PREVEQUINE™ tablets in horses less than one year in age, horses used for breeding, or in pregnant or lactating mares has not been evaluated.
Warnings
Keep out of reach of children. Do not use in horses intended for human consumption. Consult a physician in case of accidental ingestion by humans.
Adverse Reactions
Adverse reactions may include erosions and ulcers of the gums, tongue, lips and face, colic, diarrhea or pruritus.
Serious adverse reactions associated with this drug class can occur with or without warning. If adverse reactions occur, NSAID therapy should be discontinued and veterinary advice sought.
Pharmacovigilance data confirms adverse events following firocoxib administration are very rarely reported. The majority are non-serious and resolve once treatment is discontinued.
Information for owner or person treating horse:
A Client Information Sheet should be provided to the person treating the horse. Treatment administrators and caretakers should be aware of the potential for adverse reactions and the clinical signs associated with NSAID intolerance. Adverse reactions may include erosions and ulcers of the gums, tongue, lips and face, weight loss, colic, diarrhea, or icterus. Serious adverse reactions associated with this drug class can occur without warning, and in some situations result in death. Clients should be advised to discontinue NSAID therapy and contact their veterinarian immediately if any of these signs of intolerance are observed. The majority of patients with drug-related adverse reactions recover when the signs are recognized, drug administration is stopped and veterinary care is initiated.
Clinical Pharmacology
Pharmacokinetics
Following oral administration in horses at the recommended dose of 1 tablet per 360-600 kg horse, firocoxib is rapidly absorbed, and the average time to maximal concentration (Tmax) is 2.43 (± 2.17) hours. The average peak concentration (Cmax) is 75.3 (± 21.5) ng/ml, and the average area under the curve (AUCinf) is 3480 (± 1150) ng x h/ml. The elimination half-life (t1/2) after a single dose is 38.7 (± 7.8) hours. Firocoxib is approximately 97% bound to plasma proteins. Following multiple oral administrations, the steady state is reached by approximately the eighth daily dose. Firocoxib is metabolised predominantly by dealkylation and glucuronidation in the liver. Elimination is principally in the excreta (primarily the urine), with some biliary excretion also observed.
Efficacy:
The efficacy of PREVEQUINE™ tablets was established in a relative bioavailability study comparing firocoxib tablets and firocoxib oral paste. Therefore, additional field studies were not performed to support the efficacy of PREVEQUINE™ tablets.
Two hundred and fifty-three client-owned horses of various breeds, ranging in age from 2 to 37 years and weighing from 270 - 743 kg, were randomly administered firocoxib oral paste or an active control drug in multi-center field studies. Two hundred and forty horses were evaluated for efficacy and 252 horses were evaluated for safety. Horses were assessed for lameness, pain on manipulation, range of motion, joint swelling, and overall clinical improvement in a non-inferiority evaluation of firocoxib oral paste compared to an active control. At study’s end, 84.4% of horses treated with firocoxib oral paste were judged improved on veterinarians’ clinical assessment, and 73.8% were also rated improved by owners. Horses treated with firocoxib oral paste showed improvement in veterinarian-assessed lameness, pain on manipulation, range of motion, and joint swelling that was comparable to the active control.
Animal Safety:
The safety of PREVEQUINE™ tablets was supported by a relative bioavailability study comparing firocoxib tablets to firocoxib oral paste. No additional studies were conducted to support the safety of PREVEQUINE™ tablets.
In two target animal safety studies, healthy adult horses were administered firocoxib paste orally at 0.1X, 3X or 5X the recommended 0.1 mg/kg dose for varying lengths of time (30 and 42 days). Administration of firocoxib at 0.1, 0.3 and 0.5 mg/kg body weight was associated with an increased incidence of oral ulcers as compared to the control group, as well as a delayed healing of pre-existing oral (lip, tongue, gingival) ulcers. Clinical chemistry and coagulation abnormalities were seen in several horses receiving 5X the recommended dose. Elevated BUN and creatinine were noted in some horses receiving the 5X dose, as well as prolonged buccal mucosal bleeding time (BMBT) and renal pathology (dilated pelvis, tubulo-interstitial nephropathy, papillary necrosis).
In another target animal safety study, healthy adult horses were administered 0, 2.5, 7.5 or 12.5 mg/kg body weight of firocoxib for 92 days. Some horses were monitored until 147-149 days. All groups receiving the oral firocoxib experienced treatment-related adverse events. These consisted of ulcers of the lips, gingiva and tongue and erosions of the skin of the mandible and head. Gross and microscopic lesions of the kidneys consistent with tubulo-interstitial nephropathy were seen in all treated groups. Papillary necrosis was seen in the 2.5X and 12.5X groups. In addition, several 12.5X horses had elevated liver enzymes (GGT, SDH, AST and ALT). One 2.5X horse had increased urine GGT and urine protein levels, which was due to renal hemorrhage and nephropathy. Gastric ulcers in the margo plicatus and glandular area were more prevalent in the 2.5X and 7.5X groups, but not seen in the 12.5X group. The group of horses that were monitored until Days 147-149 showed partial to full recovery from oral and skin ulcers, but no recovery from tubulo-interstitial nephropathy.
In controlled field studies, 127 horses (aged 3 to 37 years) were evaluated for safety when given firocoxib oral paste at a dose of 0.1 mg/kg orally once daily for up to 14 days. In these field trials, firocoxib oral paste was safely used concomitantly with other therapies, including vaccines, anthelmintics, and antibiotics. Therefore, based on relative bioavailability of firocoxib formulations, concomitant use of PREVEQUINE™ tablets with other therapies is expected to have the same level of safety.
In a two-period crossover study conducted to evaluate the relative bioavailability of the tablet to the paste formulation, 30 horses were observed daily for adverse reactions, including oral cavity examinations. Observations were conducted at specified intervals during each treatment period to assess the effects of firocoxib on the oral mucosa. Varying degrees of oral ulcerations, lesions or other minor abnormalities were noted during the study. However, they were consistent with observations seen in horses fed a diet of hay and grain and are not likely to be related to the use of firocoxib.
Storage Information:
Store at controlled room temperature, between 15° - 30°C. Brief excursions up to 40°C are permitted, as long as they do not exceed 24 hours.
How Supplied
PREVEQUINE™ tablets are supplied in 60 and in 180 count bottles. Not all pack sizes may be marketed.
Boehringer Ingelheim Animal Health Canada Inc., 5180 South Service Road, Burlington ON L7L 5H4
PREVEQUINE™ is a trademark of Boehringer Ingelheim Animal Health France, used under license.
926931
CPN: 1182165.1
5180 SOUTH SERVICE ROAD, BURLINGTON, ON, L7L 5H4
| Customer Care No.: | 1-800-567-1885 | |
| Technical Services No.: | 1-877-565-5501 | |
| Website: | www.boehringer-ingelheim.ca |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". Animalytix assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the Animalytix service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
