Panacur Powerpac Equine Dewormer (57 g)
This treatment applies to the following species:(fenbendazole)
Paste 10% (100 mg/g)
Controls Encysted EL3 Small Strongyle Larvae
Controls both larvae & adult parasites
Panacur® (fenbendazole) is indicated for the control of:
● EL3 - encysted (hypobiotic) early 3rd stage small strongyle (cyathostome) larvae
● LL3/L4 - encysted late 3rd stage and 4th stage mucosal cyathostome larvae
● Small strongyles (cyathostomes)
● Large strongyles (Strongylus edentatus, S. equinus, S. vulgaris)
● 4th stage S. vulgaris larvae
● Ascarids (Parascaris equorum)
● Pinworms (Oxyuris equi)
Description
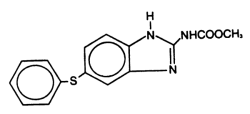
Panacur® (fenbendazole) Paste 10% contains the active anthelmintic fenbendazole. The chemical name of fenbendazole is methyl 5-(phenylthio)-2-benzimidazole carbamate. The chemical structure is:
Each gram of Panacur® Paste 10% contains 100 mg of fenbendazole and is flavored with artificial apple-cinnamon liquid.
ACTIONS:
The antiparasitic action of Panacur® Paste 10% is believed to be due to the inhibition of energy metabolism in the parasite.
Panacur Powerpac Equine Dewormer (57 g) Indications
Panacur® Paste 10% is indicated for the treatment and control of large strongyles (Strongylus edentatus, S. equinus, S. vulgaris), encysted early 3rd stage (hypobiotic), late 3rd stage and 4th stage cyathostome larvae, small strongyles, pinworms (Oxyuris equi), ascarids (Parascaris equorum), and control of arteritis caused by 4th stage larvae of Strongylus vulgaris in horses.
Precautions
Side effects associated with Panacur® Paste 10% could not be established in
well-controlled safety studies in horses with single doses as high as 454 mg/lb (1,000 mg/kg) and 15 consecutive daily doses of 22.7 mg/lb (50 mg/kg). Particularly with higher doses, the lethal action of fenbendazole may cause the release of antigens by the dying parasites. This phenomenon may result in either a local or systemic hypersensitivity reaction. As with any drug, these reactions should be treated symptomatically.
Panacur® Paste 10% has been evaluated for safety in pregnant mares during all stages of gestation with doses as high as 11.4 mg/lb (25 mg/kg) and in stallions with doses as high as 11.4 mg/lb (25 mg/kg). No adverse effects on reproduction were detected. The recommended dose for control of 4th stage Strongylus vulgaris larvae, 4.6 mg/lb (10 mg/kg) daily for 5 consecutive days, has not been evaluated for safety in stallions or pregnant mares.
Warnings
NOT FOR USE IN HUMANS. KEEP OUT OF REACH OF CHILDREN. The Safety Data Sheet (SDS) contains more detailed occupational safety information. For customer service, adverse effects reporting, and/or a copy of the SDS, call 1-800-211-3573. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDAVETS, or http://www.fda.gov/reportanimalae.OTHER WARNINGS: Do not use in horses intended for human consumption.
Parasite resistance may develop to any dewormer, and has been reported for most classes of dewormers. Treatment with a dewormer used in conjunction with parasite management practices appropriate to the geographic area and the animal(s) to be treated may slow the development of parasite resistance. Fecal examinations or other diagnostic tests and parasite management history should be used to determine if the product is appropriate for the herd, prior to the use of any dewormer. Following the use of any dewormer, effectiveness of treatment should be monitored (for example, with the use of a fecal egg count reduction test or another appropriate method). A decrease in a drug’s effectiveness over time as calculated by fecal egg count reduction tests may indicate the development of resistance to the dewormer administered. Your parasite management plan should be adjusted accordingly based on regular monitoring.
DOSAGE: Panacur® Paste 10% is administered orally at a rate of 2.3 mg/lb (5 mg/ kg) for the control of large strongyles, small strongyles, and pinworms. One syringe will deworm two 1,250 lb (568 kg) horses at a dose of 5 mg/kg. For foals and weanlings (less than 18 months of age) where ascarids are a common problem, the recommended dose is 4.6 mg/lb (10 mg/kg); one syringe will deworm a 1,250 lb horse.
For control of hypobiotic (encysted early 3rd stage), late 3rd stage, and 4th stage cyathostome larvae, as well as 4th stage Strongylus vulgaris larvae, the recommended dose is 4.6 mg/lb (10 mg/kg) daily for 5 consecutive days; administer one syringe for each 1,250 lb body weight per day.
Do not underdose. Ensure each animal receives a complete dose based on a current body weight. Underdosing may result in ineffective treatment, and encourage the development of parasite resistance.
Directions For Use
1. Determine the weight of the horse.
2. Remove syringe tip.
3. Turn the dial ring until the edge of the ring nearest the tip lines up with zero.
4. Depress plunger to advance paste to tip.
5. Now set the dial ring at the graduation nearest the weight of the horse.
6. Horse’s mouth should be free of food.
7. Insert nozzle of syringe through the interdental space and deposit the paste on the back of the tongue by depressing the plunger.
CONSULT YOUR VETERINARIAN FOR ASSISTANCE IN THE DIAGNOSIS, TREATMENT AND CONTROL OF PARASITISM.
How Supplied
Panacur® Paste 10% Equine Dewormer is supplied in 57 gram syringes, 5 per carton.Store at or below 25°C (77°F).
Fenbendazole (active ingred.) made in: see imprint. Formulated in France.
Distributed by:
Intervet Inc. (d/b/a Merck Animal Health), Rahway, NJ 07065
Approved by FDA under NADA # 120-648
©2023 Merck & Co., Inc., Rahway, NJ, USA and its affiliates. All rights reserved.
Rev. 07/23
798933 R1
CPN: 1047342.4
Intervet Inc.
126 E. LINCOLN AVENUE, PO BOX 2000, Rahway, NJ, 07065
| Customer Service: | 800-521-5767 | |
| Technical Service (Companion Animal): | 800-224-5318 | |
| Technical Service (Livestock): | 800-211-3573 | |
| Website: | www.merck-animal-health-usa.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
